Abstract
Background and Objectives
Antihypertensive therapy is effective in reducing the risk of major adverse cardiovascular events. However, blood pressure (BP) control rate remains poor and the optimal combination therapy against hypertension is not established in China. The objective of this study was to evaluate the long-term efficacy and safety of two antihypertensive regimens, amlodipine plus telmisartan and amlodipine plus amiloride/hydrochlorothiazide, in patients with essential hypertension and at least one cardiovascular risk factor.
Methods
In a multicenter open-label clinical trial, eligible patients were randomized to receive treatment with amlodipine 2.5–5 mg plus amiloride/hydrochlorothiazide 1.25–2.5 mg/12.5–25 mg (Group A) or amlodipine 2.5–5 mg plus telmisartan 40–80 mg (Group T). If a target BP was not reached, other antihypertensive agents would be added. The target BP was <130/80mmHg for patients with diabetes mellitus or chronic kidney disease and <140/90mmHg for others. Efficacy variables were changes from baseline in systolic BP and diastolic BP at the endpoint of 96 weeks. Safety evaluations included monitoring of any adverse events (AEs).
Results
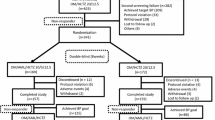
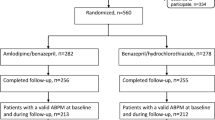
Of 13 542 patients randomized, 13 080 (96.6%) completed the study: 6529 in Group A and 6551 in Group T. At endpoint, the BP levels were reduced by 27.4/14.3 mmHg in Group A and 27.1/14.5 mmHg in Group T. The BP control rates were similar for the two therapeutic regimens (87.5% vs 86.1%). Less than 4% of patients in each group discontinued their drugs during follow-up. Peripheral edema was one of the most common AEs, and occurred in only 24 patients in Group A and 19 in Group T.
Conclusions
Long-term combination therapy with amlodipine plus telmisartan or amlodipine plus amiloride/hydrochlorothiazide was not only well tolerated but also efficacious in reducing BP levels with acceptable control rates in the majority of hypertensive patients.
Clinical Trials Registration
ClinicalTrials.gov number NCT01011660.



Similar content being viewed by others
References
Wang L. 2002 National Nutrition and Health Survey: Report I [in Chinese], general report. People’s Medical Publishing House: Beijing, 2005.
Chobanian AV, Bakris GL, Black HR, et al. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. JAMA 2003; 289 (19): 2560–72.
Lopatriello S, Berto P. Long-term social costs of hypertension. Expert Rev Pharmacoeconomics Outcomes Res 2003; 3 (1): 33–40.
Zhang XH, Lu ZL, Liu L. Coronary heart disease in China. Heart 2008; 94 (9): 1126–31.
Liu M, Wu B, Wang WZ, et al. Stroke in China: epidemiology, prevention, and management strategies. Lancet Neurol 2007; 6 (5): 456–64.
Liu L. Cardiovascular diseases in China. Biochem Cell Biol 2007; 85 (2): 157–63.
Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ 2009; 338: b1665.
Wolf-Maier K, Cooper RS, Kramer H, et al. Hypertension treatment and control in five European countries, Canada, and the United States. Hypertension 2004; 43 (1): 10–7.
Wu Y, Huxley R, Li L, et al., China NNHS Steering Committee; China NNHS Working Group. Prevalence, awareness, treatment, and control of hypertension in China: data from the China National Nutrition and Health Survey 2002. Circulation 2008; 118 (25): 2679–86.
ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002; 288 (23): 2981–97.
Dahlöf B, Sever PS, Poulter NR, et al., ASCOT Investigators. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): a multicentre randomised controlled trial. Lancet 2005; 366 (9489): 895–906.
Jamerson K, Weber MA, Bakris GL, et al., ACCOMPLISH Trial Investigators. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359 (23): 2417–28.
Mancia G, de Backer G, Dominiczak A, et al. 2007 Guidelines for the Management of Arterial Hypertension — The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 2007; 25 (6): 1105–87.
Gong L, Zhang W, Zhu Y, et al. Shanghai Trial of Nifedipine in the Elderly (STONE). J Hypertens 1996; 14 (10): 1237–45.
Wang JG, Staessen JA, Gong L, et al. Chinese trial on isolated systolic hypertension in the elderly. Systolic Hypertension in China (Syst-China) Collaborative Group. Arch Int Med 2000; 160 (2): 211–20.
Liu L, Zhang Y, Liu G, et al., FEVER Study Group. The Felodipine Event Reduction (FEVER) study: a randomized long-term placebo controlled trial in Chinese hypertensive patients. J Hypertens 2005; 23 (12): 2157–72.
Hashimoto J, Hirayama H, Hanasawa T, et al. Efficacy of combination antihypertensive therapy with low-dose indapamide: assessment by blood pressure self-monitoring at home. Clin Exp Hypertens 2005; 27 (4): 331–41.
Littlejohn 3rd TW, Majul CR, Olvera R, et al., Study Investigators. Results of treatment with telmisartan-amlodipine in hypertensive patients. J Clin Hypertens (Greenwich) 2009; 11 (4): 207–13.
Littlejohn 3rd TW, Majul CR, Olvera R, et al., Study investigators. Telmisartan plus amlodipine in patients with moderate or severe hypertension: results from a subgroup analysis of a randomized, placebo-controlled, parallelgroup, 4×4 factorial study. Postgrad Med 2009; 121 (2): 5–14.
White WB, Littlejohn TW, Majul CR, et al. Effects of telmisartan and amlodipine in combination on ambulatory blood pressure in stages 1–2 hypertension. Blood Press Monit 2010; 15 (4): 205–12.
Wang W, Ma L, Zhang Y, et al. The combination of amlodipine and angiotensin receptor blocker or diuretics in high-risk hypertensive patients: rationale, design and baseline characteristics. J Hum Hypertens 2011; 25 (4): 271–7.
Mancia G, Laurent S, Agabiti-Rosei E, et al. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension Task Force document. Blood Press 2009; 18 (6): 308–47.
Calhoun DA, Crikelair NA, Yen J, et al. Amlodipine/valsartan/hydrochlorothiazide triple combination therapy in moderate/severe hypertension: secondary analyses evaluating efficacy and safety. Adv Ther 2009; 26 (11): 1012–23.
Black HR, Elliott WJ, Grandits G, et al., CONVINCE Research Group. Principal results of the Controlled Onset Verapamil Investigation of Cardiovascular End Points (CONVINCE) trial. JAMA 2003; 289 (16): 2073–82.
Li H, Meng Q, Sun X, et al. Prevalence, awareness, treatment, and control of hypertension in rural China: results from Shandong Province. J Hypertens 2010; 28 (3): 432–8.
Ong KL, Cheung BM, Man YB, et al. Prevalence, awareness, treatment, and control of hypertension among United States adults 1999–2004. Hypertension 2007; 49 (1): 69–75.
Cooper-DeHoff RM, Gong Y, Handberg EM, et al. Tight blood pressure control and cardiovascular outcomes among hypertensive patients with diabetes and coronary artery disease. JAMA 2010; 304 (1): 61–8.
ACCORD Study Group, Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med 2010; 362 (17): 1575–85.
Smith TR, Glazer RD, Koren MJ, et al. Combination therapy with amlodipine/valsartan in essential hypertension: a 52-week, randomised, open-label, extension study. Int J Clin Pract 2010; 64 (10): 1367–74.
Philipp T, Smith TR, Glazer R, et al. Two multicenter, 8-week, randomized, double-blind, placebo-controlled, parallel-group studies evaluating the efficacy and tolerability of amlodipine and valsartan in combination and as monotherapy in adult patients with mild to moderate essential hypertension. Clin Ther 2007; 29 (4): 563–80.
Philipp T, Glazer RD, Zhao Y, et al. Long-term tolerability and efficacy of the combination of amlodipine/valsartan in hypertensive patients: a 54-week, open-label extension study. Curr Med Res Opin 2009; 25 (1): 187–93.
Fogari R, Malamani GD, Zoppi A, et al. Effect of benazepril addition to amlodipine on ankle oedema and subcutaneous tissue pressure in hypertensive patients. J Hum Hypertens 2003; 17 (3): 207–12.
Fogari R, Zoppi A, Derosa G, et al. Effect of valsartan addition to amlodipine on ankle oedema and subcutaneous tissue pressure in hypertensive patients. J Hum Hypertens 2007; 21 (3): 220–4.
Ogihara T, Saruta T, Rakugi H, et al., COLM study investigators. Rationale, study design and implementation of the COLM study: the combination of OLMesartan and calcium channel blocker or diuretic in high-risk elderly hypertensive patients. Hypertens Res 2009; 32 (2): 163–7.
Acknowledgments
This study was supported by the Ministry of Sciences and Technology of the People’s Republic of China (Grant No. 2006BAI01A03). We would like to express our gratitude to the doctors participating in the CHIEF study and thank Dawnrays Pharmaceutical (Holdings) Limited Company for providing the study drugs for free. —Liyuan Ma and Yong Zhao are co-first authors, and contributed equally to this work. —Conflicts of interest: The authors have no conflicts of interest directly relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, L., Wang, W., Zhao, Y. et al. Combination of Amlodipine plus Angiotensin Receptor Blocker or Diuretics in High-Risk Hypertensive Patients. Am J Cardiovasc Drugs 12, 137–142 (2012). https://doi.org/10.2165/11598110-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11598110-000000000-00000