Abstract
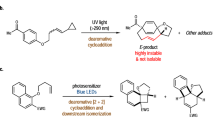
The photolysis of the cyclopentene-annelated DBH-type azoalkanes 1c,d with the n-propyl and acetoxymethyl substituents at the bridgehead positions affords under singlet conditions (high-temperature direct photolysis) predominantly the retained housanes anti-2c,d, while under triplet conditions (low-temperature direct or benzophenone-sensitized photolysis) the inverted diastereomer syn-2c,d is favored. The latter novel stereochemical feature of the triplet pathway is rationalized in terms of the unsymmetric nature of the n-propyl and acetoxymethyl substituents in regard to rotation about the bridgehead position of the planar cyclopentane-1,3-diyl triplet diradicals3DR(c,d). In their lowest-energy conformation (AM1 calculations), the rotationally unsymmetric substituents prefer to be located opposite to the annelated cyclopentene ring. The ratio of the diastereomeric syn and anti housanes is determined by the equilibrium distribution of the bridgehead-substituent conformers for the triplet diradicals on ring closure after ISC.
Similar content being viewed by others
References
W. R. Roth, M. Martin, Stereochemistry of the thermal and photochemical decomposition of 2,3-diazabicyclo[2.2.1]hept-2-ene, Liebigs Ann. Chem., 1967, 702, 1–5.
W. R. Roth, M. Martin, Stereochemistry of the 1,2-cycloaddition to the bicyclo[2.1.0] system, Tetrahedron Lett., 1967, 47, 4695–4698.
E. L. Allred, R. L. Smith, Thermal and photodecomposition studies with the exo- and endo-5-methoxy-2,3-diazabicyclo[2.2.1]hept-2-ene system, J. Am. Chem. Soc., 1969, 91, 6766–6775.
C. D. Sherill, E. T. Seidl, H. F. Schaefer III, Closs’s diradical: some surprises on the potential energy hypersurface, J. Phys. Chem., 1992, 96, 3712–3716.
D. C. Sorescu, D. L. Thompson, L. M. Raff, Molecular dynamics studies of the thermal decomposition of 2,3-diazabicyclo(2.2.1)hept-2-ene, J. Chem. Phys., 1995, 102, 7910–7924.
N. Yamamoto, M. Olivucci, P. Celani, F. Bernardi, M. A. Robb, An MC-SCF/MP2 study of the photochemistry of 2,3-diazabicyclo[2.2.1]hept-2-ene: Production and fate of diazenyl and hydrazonyl biradicals, J. Am. Chem. Soc., 1998, 120, 2391–2407.
W. Adam, T. Oppenländer, G. Zang, Photochemistry of the azoalkanes 2,3-diazabicyclo[2.2.1]hept-2-ene and spiro[cyclopropane-7,1’-[2,3]-diazabicyclo[2.2.1]hept-2-ene]: on the questions of one-bond vs. two-bond cleavage during the denitrogenation, cyclization vs. rearrangement of the 1,3-diradicals and double inversion, J. Org. Chem., 1985, 50, 3303–3312.
C. J. S. M. Simpson, G. J. Wilson, W. Adam, Dynamics of the thermal decomposition of 2,3-diazabicyclo[2.2.1]hept-2-ene, J. Am. Chem. Soc., 1991, 113, 4728–4732.
W. Adam, U. Denninger, R. Finzel, F. Kita, H. Platsch, H. Walter, G. Zang, Comparative study of the pyrolysis, photoinduced electron transfer (PET), and laser-jet and 185-nm photochemistry of alkyl-substituted bicyclic azoalkanes, J. Am. Chem. Soc., 1992, 114, 5027–5035.
M. B. Reyes, B. K. Carpenter, Mechanism of thermal deazetization of 2,3-diazabicyclo[2.2.1]hept-2-ene and its reaction dynamics in supercritical fluids, J. Am. Chem. Soc., 2000, 122, 10163–10176.
W. Adam, H. García, V. Martí, J. N. Moorthy, Homolytic substitution (SH2) versus triplet diradical (ISC) in the photochemical denitrogenation of a DBH azoalkane: temperature-dependent syn/anti diastereoselectivity as a mechanistic probe for the doubly inverted housane, J. Am. Chem. Soc., 1999, 121, 9475–9476.
W. Adam, H. García, M. Diedering, V. Martí, M. Olivucci, E. Palomares, Stereochemical memory in the temperature-dependent photodenitrogenation of bridgehead-substituted DBH-type azoalkanes: inhibition of inverted-housane formation in the diazenyl diradical through the mass effect (inertia) and steric hindrance, J. Am. Chem. Soc., 2002, 124, 12192–12199.
K. Beck, S. Hünig, Azo bridges from azines. VI. Substituted isopyrazoles as electron-deficient dienes for the synthesis of 2,3-diazabicyclo[2.2.1]heptenes and their photochemistry, Chem. Ber., 1987, 120, 477–483.
W. Adam, M. Grüne, M. Diedering, A. V. Trofimov, Temperature and viscosity dependence in the stereoselective formation of the inverted housane for the photochemical nitrogen loss from the deuterium-stereolabeled parent diazabicyclo[2.2.1]hept-2-ene, J. Am. Chem. Soc., 2001, 123, 7109–7112.
W. Adam, V. Martí, C. Sahin, A. V. Trofimov, Stereoselective formation of inverted housane in the denitrogenation of the diazenyl diradical photolytically derived from a stereolabeled diazabicyclo[2,2,1]hept-2-ene with bridgehead substituents as a function of solvent- and temperature-varied viscosity, Chem. Phys. Lett., 2001, 340, 26–32.
W. Adam, M. Diedering, A. V. Trofimov, Pressure dependence of the stereoselectivity in the photodenitrogenation of diazabicyclo[2.2.1]hept-2-ene in supercritical fluids: evidence for the diazenyl diradical, Chem. Phys. Lett., 2001, 350, 453–458.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, V. G. Zakrzewski, J. A. Montgomery, Jr., R. E. Stratmann, J. C. Burant, S. Dapprich, J. M. Millam, A. D. Daniels, K. N. Kudin, M. C. Strain, O. Farkas, J. Tomasi, V. Barone, M. Cossi, R. Cammi, B. Mennucci, C. Pomelli, C. Adamo, S. Clifford, J. Ochterski, G. A. Petersson, P. Y. Ayala, Q. Cui, K. Morokuma, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. Cioslowski, J. V. Ortiz, A. G. Baboul, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. Gomperts, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, C. Gonzalez, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, J. L. Andres, C. Gonzalez, M. Head-Gordon, E. S. Replogle and J. A. Pople, Gaussian 98, Revision A.7, Gaussian, Inc.: Pittsburgh, PA, 1998.
W. Adam, V. Martí, C. Sahin, A. V. Trofimov, Viscosity-controlled stereoselective inversion in the photochemical denitrogenation of a stereolabeled diazabicyclo[2.2.1]heptene (DBH)-type azoalkane, J. Am. Chem. Soc., 2000, 122, 5002–5003.
S. L. Buchwalter, G. L. Closs, Electron-spin-resonance study of matrix-isolated 1,3-cyclopentadiyl, a localized 1,3-carbon biradical, J. Am. Chem. Soc., 1975, 97, 3857–3858.
S. L. Buchwalter, G. L. Closs, Electron-spin-resonance and CIDNP studies on 1,3-cyclopentadiyls. A localized 1,3 carbon biradical system with a triplet ground state. tunneling in carbon-carbon bond formation, J. Am. Chem. Soc., 1979, 101, 4688–4694.
W. Adam, S. Grabowski, R. M. Wilson, Localized cyclic triplet diradicals. Lifetime determination by trapping with oxygen, Acc. Chem. Res., 1990, 23, 165–72.
L. Salem, C. Rowland, Electronic properties of diradicals, Angew. Chem., Int. Ed. Engl., 1972, 11, 92–111.
D. C. Sorescu, D. L. Thompson, L. M. Raff, Statistical effects in the skeletal inversion of bicyclo[2.1.0] pentane, J. Chem. Phys., 1994, 101, 3729–3741.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adam, W., Diedering, M. Enhanced formation of inverted housane through steric effects by rotationally unsymmetric bridgehead substituents in the ring closure of triplet cyclopentane-1,3-diyl diradicals, generated photolytically from 2,3-diazabicyclo[2.2.1]heptene(DBH)-type azoalkanes. Photochem Photobiol Sci 2, 393–397 (2003). https://doi.org/10.1039/b211319h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/b211319h