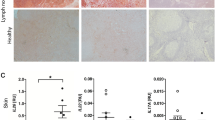
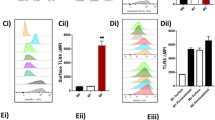
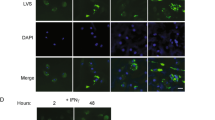
Abstract
Leptospira interrogans are zoonotic pathogens that have been linked to a recent increased incidence of morbidity and mortality in highly populated tropical urban centers. They are unique among invasive spirochetes in that they contain outer membrane lipopolysaccharide (LPS) as well as lipoproteins. Here we show that both these leptospiral outer membrane constituents activate macrophages through CD14 and the Toll-like receptor 2 (TLR2). Conversely, it seems that TLR4, a central component for recognition of Gram-negative LPS, is not involved in cellular responses to L. interrogans. We also show that for intact L. interrogans, it is LPS, not lipoprotein, that constitutes the predominant signaling component for macrophages through a TLR2 pathway. These data provide a basis for understanding the innate immune response caused by leptospirosis and demonstrate a new ligand specificity for TLR2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Pereira, M. M. et al. A clonal subpopulation of Leptospira interrogans sensu stricto is the major cause of leptospirosis outbreaks in Brazil. J. Clin. Microbiol. 38, 450–452 (2000).
Barnett, J. K. et al. Expression and distribution of leptospiral outer membrane components during renal infection of hamsters. Infect. Immun. 67, 853–861 (1999).
Faine, S. Adler, B., Bolin, C. & Perolat, P. Leptospira and Leptospirosis 2nd edn (Monash University Press, Melbourne, Australia, 1999).
Chu, K. M., Rathinam, R., Namperumalsamy, P. & Dean, D. Identification of Leptospira species in the pathogenesis of uveitis and determination of clinical ocular characteristics in South India. J. Infect. Dis. 177, 1314–1321 (1998).
Adler, B. & Faine, S. The antibodies involved in the human immune response to leptospiral infection. J. Med. Microbiol. 11, 387–400 (1978).
de Souza, L. & Koury, M. C. Isolation and biological activities of endotoxin from Leptospira interrogans. Can. J. Microbiol. 38, 284–289 (1992).
Mitchison, M. et al. Identification and characterization of the dTDP-rhamnose biosynthesis and transfer genes of the lipopolysaccharide-related rfb locus in Leptospira interrogans serovar copenhageni. J. Bacteriol. 179, 1262–1267 (1997).
Kalambaheti, T., Bulach, D. M., Rajakumar, K. & Adler, B. Genetic organization of the lipopolysaccharide O-antigen biosynthetic locus of Leptospira borgpetersenii serovar hardjobovis. Micro. Pathogen. 27, 105–117 (1999).
Bulach, D. M., Kalambaheti, T., da la Pena-Moctezuma, A. & Adler, B. Functional analysis of genes in the rfb locus of Leptospira borgpetersenii serovar hardjo subtype hardjobovis. Infect. Immun. 68, 3793–3798 (2000).
Bulach, D. M., Kalambaheti, T., de la Pena-Moctezuma, A. & Adler, B. Lipopolysaccharide biosynthesis in Leptospira. J. Mol. Microbiol. Biotechnol. 2, 375–380 (2000).
Haake, D. A. et al. Characterization of leptospiral outer membrane lipoprotein LipL36: downregulation associated with late log-phase growth and mammalian infection. Infect. Immun. 66, 1579–1587 (1998).
Shang, E. S., Summers, T. A. & Haake, D. A. Molecular cloning and sequence analysis of the gene encoding LipL41, a surface-exposed lipoprotein of pathogenic Leptospira species. Infect. Immun. 64, 2322–2330 (1996).
Haake, D. A. et al. The leptospiral major outer membrane protein LipL32 is a lipoprotein expressed during mammalian infection. Infect. Immun. 68, 2276–2285 (2000).
Ulevitch, R. J. & Tobias, P. S. Receptor-dependent mechanisms of cell stimulation by bacterial endotoxin. Annu. Rev. Immunol. 13, 437–457 (1995).
Ulevitch, R. J. Toll gates for pathogen selection. Nature 401, 755–756 (1999).
Ulevitch, R. J. Endotoxin opens the Toll gates of innate immunity. Nature Med. 5, 144–145 (1999).
Poltorak, A. et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282, 2085–2088 (1998).
Beutler, B. Tlr4: central component of the sole mammalian LPS sensor. Curr. Opin. Immunol. 12, 20–26 (2000).
Aderem, A. & Ulevitch, R. J. Toll-like receptors in the induction of the innate immune response. Nature 406, 782–787 (2000).
Hirschfeld, M., Ma, Y., Weis, J. H., Vogel, S. N. & Weis, J. J. Cutting edge: repurification of lipopolysaccharide eliminates signaling through both human and murine toll-like receptor 2. J. Immunol. 165, 618–622 (2000).
Tapping, R. I., Akashi, S., Miyake, K., Godowski, P. & Tobias, P. S. Toll-like receptor 4, but not Toll-like receptor 2, is a signaling receptor for Escherichia and Salmonella LPSs. J. Immunol. 165, 5780–5787 (2000).
Leturcq, D. J. et al. Antibodies against CD14 protect primates from endotoxin-induced shock. J. Clin. Invest. 98, 1533–1538 (1996).
Heine, H. et al. Cutting edge: cells that carry a null allele for toll-like receptor 2 are capable of responding to endotoxin. J. Immunol. 162, 6971–6975 (1999).
Underhill, D. M. et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811–815 (1999).
Kirschning, C. J., Wesche, H., Merrill, A. T. & Rothe, M. Human toll-like receptor 2 confers responsiveness to bacterial lipopolysaccharide. J. Exp. Med. 188, 2091–2097 (1998).
Lien, E. et al. Toll-like receptor 2 functions as a pattern recognition receptor for diverse bacterial products. J. Biol. Chem. 274, 33419–33425 (1999).
Sellati, T. J. et al. Treponema pallidum and Borrelia burgdorferi lipoproteins and synthetic lipopeptides activate monocytic cells via a CD14-dependent pathway distinct from that used by lipopolysaccharide. J. Immunol. 160, 5455–5464 (1998).
Brightbill, H. D. et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 285, 732–736 (1999).
Akashi, S. et al. Cutting edge: cell surface expression and lipopolysaccharide signaling via the toll-like receptor 4-MD-2 complex on mouse peritoneal macrophages. J. Immunol. 164, 3471–3475 (2000).
Radolf, J. D. et al. Lipoproteins of Borrelia burgdorferi and Treponema pallidum activate cachectin/tumor necrosis factor synthesis. Analysis using a CAT reporter construct. J. Immunol. 147, 1968–1974 (1991).
Manthey, C. L. & Vogel, S. L. Elimination of trace endotoxin protein from rough chemotype LPS. J. Endotoxin Res. 1, 84–91 (1994).
Isogai, E., Kitagawa, H., Isogai, H., Kurebayashi, Y. & Ito, N. Phagocytosis as a defense mechanism against infection with Leptospira. Zentralbl Bakteriol Mikrobiol Hyg [A] 261, 65–74 (1986).
Vinetz, J. M., Glass, G. E., Flexner, C. E., Mueller, P. & Kaslow, D. C. Sporadic urban leptospirosis. Ann. Intern. Med. 125, 794–798 (1996).
Arean, V. M. The pathologic anatomy and pathogenesis of fatal human leptospirosis (Weils disease). Am. J. Pathol. 40, 393–423 (1962).
Vinh, T., Adler, B. & Faine, S. Ultrastructure and chemical composition of lipopolysaccharide extracted from Leptospira interrogans serovar copenhageni. J. Gen. Microbiol. 132, 103–109 (1986).
Vinh, T. U., Shi, M. H., Adler, B. & Faine, S. Characterization and toxonomic significance of lipopolysaccharides of Leptospira interrogans serovar hardjo. J. Gen. Microbiol. 135, 2663–2673 (1989).
Norgard, M. V. et al. Activation of human monocytic cells by Treponema pallidum and Borrelia burgdorferi lipoproteins and synthetic lipopeptides proceeds via a pathway distinct from that of lipopolysaccharide but involves the transcriptional activator NF-kappa B. Infect. Immun. 64, 3845–3852 (1996).
Beutler, B. Endotoxin, toll-like receptor 4, and the afferent limb of innate immunity. Curr. Opin. Microbiol. 3, 23–28 (2000).
Hoshino, K. et al. Cutting edge: Toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J. Immunol. 162, 3749–3752 (1999).
Takeuchi, O. et al. Differential roles of Toll-like receptor (TLR) 2 and TLR4 in recognition of Gram-negative and Gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Lien, E. et al. Toll-like receptor 4 imparts ligand-specific recognition of bacterial lipopolysaccharide. J. Clin. Invest. 105, 497–504 (2000).
Poltorak, A., Ricciardi-Castagnoli, P., Citterio, S. & Beutler, B. Physical contact between lipopolysaccharide and toll-like receptor 4 revealed by genetic complementation. Proc. Natl Acad. Sci. USA 97, 2163–2167 (2000).
Baril, C. & Saint Girons I. Sizing of the Leptospira genome by pulsed-field agarose gel electrophoresis. FEMS Microbiol. Lett. 71, 100 (1990).
Zuerner, R. L., Knudtson, W., Bolin, C. A. & Trueba, B. Characterization of outer membrane and secreted proteins of Leptospira interrogans serovar pomona. Microb. Pathog. 10, 311–322 (1991).
Fraser, C. M. et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 390, 580–586 (1997).
Ellinghausen, H. C. & McCulloch, W. G. Nutrition of Leptospira pomona and growth of 13 other serotypes: fractionation of oleic albumin complex and a medium of bovine albumin and polysorbate 80. Am. J. Vet. Res. 26, 45–51 (1965).
Johnson, R. C. & Harris, V. G. Differentiation of pathogenic and saprophytic leptospires. I. Growth at low temperatures. J. Bacteriol. 94, 27–31 (1967).
Westphal, O. & Jann, K. Bacterial lipopolysaccharides: extraction with phenol-water and further application of the procedure. Methods in Carbohydrate Chem. 5, 83–91 (1965).
Mathison, J. C. et al. Adaptation to bacterial lipopolysaccharide (LPS) controls LPS-induced tumor necrosis factor production in rabbit macrophages. J. Clin. Invest. 85, 1108–1118 (1990).
Lee, C.-H. & Tsai, C.-M. Quantification of bacterial lipopolysaccharides by the purpald assay: measuring formaldehyde generated from 2-keto-deoxyoctonate and heptose at the inner core by periodate oxidation. Anal. Biochem. 267, 161–168 (1999).
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680 (1970).
Tsai, C.-M. & Frasch, C. E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal. Biochem. 119, 115–119 (1982).
Kirkland, T. N. et al. Analysis of lipopolysaccharide binding by CD14. J. Biol. Chem. 268, 24818–24823 (1993).
Yang, R. B. et al. Toll-like receptor-2 mediates lipopolysaccharide-induced cellular signalling. Nature 395, 284–288 (1998).
Pugin, J. et al. Cell activation mediated by glycosylphosphatidylinositol-anchored or transmembrane forms of CD14. Infect. Immun. 66, 1174–1180 (1998).
Kravchenko, V. V. et al. Platelet-activating factor induces NF-κB activation through a G protein-coupled pathway. J. Biol. Chem. 270, 14928–14934 (1995).
Kravchenko, V. V., Steinemann, S., Kline, L., Feng, L. & Ulevitch, R. J. Endotoxin tolerance is induced in Chinese hamster ovary cell lines expressing human CD14. Shock 5, 194–201 (1996).
Acknowledgements
Supported by grants AI-15136, GM-28485 and GM-37696 from the National Institutes of Health (to R. J. U.), a Beginning Grant-in-Aid from the American Heart Association Western States Affiliate (to R. I. T.) and by a postdoctoral fellowship from North Atlantic Treaty Organization (to C. W.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Werts, C., Tapping, R., Mathison, J. et al. Leptospiral lipopolysaccharide activates cells through a TLR2-dependent mechanism. Nat Immunol 2, 346–352 (2001). https://doi.org/10.1038/86354
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/86354
This article is cited by
-
Peptidoglycan mediates Leptospira outer membrane protein Loa22 to toll-like receptor 2 for inflammatory interaction: a novel innate immune recognition
Scientific Reports (2021)
-
Toll-Like Receptor 2-Mediated Glial Cell Activation in a Mouse Model of Cuprizone-Induced Demyelination
Molecular Neurobiology (2018)
-
Active Components of Leptospira Outer Membrane Protein LipL32 to Toll-Like Receptor 2
Scientific Reports (2017)
-
A new model of self-resolving leptospirosis in mice infected with a strain of Leptospira interrogans serovar Autumnalis harboring LPS signaling only through TLR4
Emerging Microbes & Infections (2017)
-
Leptospiral outer membrane protein LipL32 induces inflammation and kidney injury in zebrafish larvae
Scientific Reports (2016)