Abstract
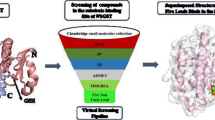
Filariasis causing nematode Brugia malayi is shown to harbor wolbachia bacteria as symbionts. The sequenced genome of the wolbachia endosymbiont from B.malayi (wBm) offers an unprecedented opportunity to identify new wolbachia drug targets. Genome analysis of the glycolytic/gluconeogenic pathway has revealed that wBm lacks pyruvate kinase (PK) and may instead utilize the enzyme pyruvate phosphate dikinase (PPDK; ATP: pyruvate, orthophosphate phosphotransferase, EC 2.7.9.1). PPDK catalyses the reversible conversion of AMP, PPi and phosphoenolpyruvate into ATP, Pi and pyruvate. Most organisms including mammals exclusively possess PK. Therefore the absence of PPDK in mammals makes this enzyme as attractive wolbachia drug target. In the present study we have modeled the three dimensional structure of wBm PPDK. The template with 50% identity and 67% similarity in amino acid sequence was employed for homology-modeling approach. The putative active site consists of His476, Arg360, Glu358, Asp344, Arg112, Lys43 and Glu346 was selected as site of interest for designing suitable inhibitor molecules. Docking studies were carried out using induced fit algorithms with OPLS force field of Schrödinger’s Glide. The lead molecules which inhibit the PPDK activity are taken from the small molecule library (Pubchem database) and the interaction analysis showed that these compounds may inhibit the function of PPDK in wBm.
Similar content being viewed by others
Abbreviations
- PPDK:
-
pyruvate phosphate dikinase
- PK:
-
pyruvate kinase
- PEP:
-
phosphoenol pyruvate
- OPLS-AA:
-
optimized liquid solution all atoms
- ATP:
-
adenosine triphosphate
- wBm :
-
Wolbachia Brugia malayi
References
Acosta, H., Dubourdieu, M., Quiñones, W., Cáceres, A., Bringaud, F., Concepción, J.L. 2004. Pyruvate phosphate dikinase and pyrophosphate metabolism in the glycosome of trypanosoma cruzei epimastogotes. Comp Biochem Mol Biol 138, 347–356.
Bateman, A., Coin, L., Durbin, R., Finn, R.D., Hollich, V., Griffiths, J.S., Khanna, A., Marshall, M., Moxon, S., Sonnhammer, E.L., Studholme, D.J., Yeats, C., Eddy, S.R. 2004. The Pfam protein families’ database. Nucleic Acids Res 32, 138–141.
Castrignanò, T., De Meo, P.D., Cozzetto, D., Talamo, I.G., Tramontano, A. 2006. The Pfam protein families database. Nucleic Acids Res 34, 306–309.
Cherie, A.M., David, G.B., James, N.B., Jason, R.D., Dianne, S.H., Catherine, H.L., Lyndon, E.L., Shilo, L., Andrew, M., Dianne, M.T. 2007. Screening marine fungi for inhibitors of the C4 plant enzyme pyruvate phosphate dikinase: Unguinol as a potential novel herbicide candidate. Appl Environ Microbiol 73, 1921–1927.
Cosenza, L.W., Bringuad, F., Baltz, T., Vellieux, F.M. 2002. The 3.0 Å resolution crystal structure of glycosomal pyruvate phosphate dikinase from trypanosoma brucei. J Mol Biol 318, 1417–1432.
Feng, X.M., Cao, L.J., Adam, R.D., Zhand, X.C., Lu, S.Q. 2008. The catalyzing role of PPDK in Giardia lamblia. Biochem Biophys Res Commun 367, 394–398.
Halgren, T.A., Murphy, R.B., Friesner, R.A., Beard, H.S., Frye, L.L., Pollard, W.T., Banks, J.L. 2004. Glide: A new approach for rapid, accurate docking and scoring. 2. Enrichment factors in database screening. J Med Chem 47, 1750–1759.
Harland, G.W., O’Brien, W.E., George, M. 1977. Properties of carboxytranphosphorylase; pyruvate phosphate dikinase, pyrophosphate-phosphofructokinase and phosphate acetate kinase and their roles in metabolism of inorganic phosphate. Adv Enzymol Relat Areas Mol Biol 45, 85–155.
Herzberg, O., Chen, C.C., Kapadia, G., McGuire, M., Carroll, L.J., Noh, S.J., Dunaway, M.D. 1996. Swiveling-domain mechanism for enzymatic phosphotransfer between reaction sites. Proc Natl Acad Sci USA 93, 2652–2657.
Herzberg, O., Chen, C.C., Liu, S., Tempczyk, A., Howard, A., Wei, M., Ye, D., Dunaway, M.D. 2002. Pyruvate site of pyruvate phosphate dikinase: Crystal structure of the enzyme phosphonopyruvate complex and mutant analysis. Biochemistry 41, 780–787.
Holm, L., Park, J. 2000. DaliLite workbench for protein structure comparison. Bioinformatics 16, 566–567.
Jeremy, F., Mehul, G., Ibrahim, K., Jennifer, W., Kira, M., Natalia, I., Anamitra, B., Vinayak, K., Sanjay, K., Janos, P., Tamas, V., Jessica, I., Laurie, M., Alla, L., Marina, O., Nikos, K., Elodie, G., Shiliang, W., Eugene, G., Victor, J., Olga, O., Kiryl, T., Mikhail, M., Donald, C., Eugene, K., Barton, S. 2005. The Wolbachia genome of Brugia malayi: Endosymbiont evolution with a human pathogenic nematode. PLos Biol 3, 121.
Jeyaprakash, A., Hoy, M.A. 2000. Long PCR improves Wolbachia DNA amplification: WSP sequences found in 76% of 63 arthropod species. Insect Mol Biol 9, 393–405.
Kozek, W.J., Marroquin, H.F. 1977. Intracytoplasmic bacteria in Onchocerca volvulus. Am J Trop Med Hyg 26, 663–678.
Larkin, M.A., Blackshields, G., Brown, N.P., Chenna, R., McGettigan, P.A., McWilliam, H., Valentin, F., Wallace, I.M., Wilm, A., Lopez, R., Thompson, J.D., Gibson, T.J., Higgins, D.G. 2007. ClustalW and ClustalX version 2.0. Bioinformatics 23, 2947–2945.
Laskowski, R.A., MacArthur, M.W., Moss, D.S., Thornton, J.M. 1993. PROCHECK — a program to check the stereochemical quality of protein structures. J App Cryst 26, 283–291.
Laurie, A.T., Jackson, R.M. 2005. Q-Site Finder: An energy-based method for the prediction of proteinligand binding sites. Bioinformatics 21, 1908–1916.
Lin, Y., Lusin, J.D., Ye, D., Dunaway, M.D., Ames, J.B. 2006. Examination of the structure, stability and catalytic potential in the engineered phosphoryl carrier domain of pyruvate phosphate dikinase. Biochemistry 45, 1702–1711.
Lipinski, C.A., Lombardo, F., Dominy, B., Wand Feeney, F.J. 1997. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Delivery Rev 23, 3–25.
McLaren, D.J., Worms, M.J., Laurence, B.R., Simpson, M.G. 1975. Micro-organisms in filarial larvae (Nematoda). Trans R Soc Trop Med Hyg 69, 509–514.
Mertens, E. 1993. ATP versus pyrophosphate: Glycolysis revisited in parasitic protists. Parasitol Today 9, 122–126.
Pfarr, K.M., Hoerauf, A. 2007. A niche for Wolbachia. Trends Parasitol 23, 5–7.
Raverdy, S., Foster, J.M., Roopenian, E., Carlow, C.K. 2008. The wolbachia endosymbiont of Brugia malayi has an active pyruvate phosphate dikinase. Mol Biochem Parasitol 160, 163–166.
Saavedra-Lira, E., Perez-Montfort, R. 1996. Energy production in Entamoeba histolytica: New perspective in rational drug design. Arch Med Res 27, 257–264.
Sali, A., Blundell, T.L. 1993. Comparative protein modeling by satisfaction of spatial restraints. J Mol Biol 234, 779–815.
Werren, J.H., Windsor, D.M. 2000. Wolbachia infection frequencies in insects: Evidence of a global equilibrium? Proc Biol Sci 267, 1277–1285.
World Health Organization. 2010. Global programme to eliminate Lymphatic Filariasis. whqlibdoc.who.int/publications/2010/9789241500722_eng.pdf
Ye, D., Wei, M., McGuire, M. 2001. Investigation of catalytic site within the ATP-grasp domain of clostridium symbiosum. Pyruvate phosphate dikinase. J Mol Chem 276, 37630–37639.
Yuan, X., Linda, Y., Li, S., Young-Shik, J., Patrick, S.M., Dunaway, M.D., Brian, M.M. 1995. Location for the catalytic site for phosphoenolpyruvate formation within the primary structure of Clostridium symbiosum pyruvate phosphate dikinase. Biochemistry 34, 2181–2187.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palayam, M., Lakshminarayanan, K., Radhakrishnan, M. et al. Preliminary analysis to target pyruvate phosphate dikinase from wolbachia endosymbiont of Brugia malayi for designing anti-filarial agents. Interdiscip Sci Comput Life Sci 4, 74–82 (2012). https://doi.org/10.1007/s12539-011-0109-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-011-0109-2