Abstract
Objectives
To compare preemptive single-dose etoricoxib and dexamethasone on postoperative patient satisfaction (pPS) and clinical parameters following the impacted mandibular third molar (IMTM) extraction.
Materials and Methods
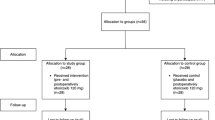
A parallel-group, triple-blinded, controlled clinical study included a total of 90 patients (n = 30), randomized to receive: etoricoxib 90 mg, dexamethasone 4 mg, or no premedication (control group) 1 h before surgery. Paracetamol 500 mg was prescribed as rescue medication (RM). Check-ups were scheduled at 24 h, 48 h, and day 7 post-surgery. At each time point, pPS was assessed using the 5-point Likert scale. RM parameters, swelling, trismus, and the occurrence of adverse events were also recorded, and patients were instructed to rate the perceived pain on Visual Analogue Scale.
Results
In all the follow-up periods, data indicated significantly higher pPS scores in the preemptive medication groups when compared to the control group (p < 0.05). Both regimens delayed the first RM intake when compared to controls. In the etoricoxib group, a significantly lower total RM consumption was observed (p < 0.05). Dexamethasone significantly decreased swelling at each check-up and increased mouth opening at day 7 after the surgery (p < 0.05).
Conclusions
Preemptive etoricoxib and dexamethasone elevate pPS after IMTM surgery. Etoricoxib improves RM parameters, while dexamethasone ameliorates the patient’s postoperative functional ability.
Clinical Relevance
Preemptive etoricoxib and dexamethasone use may decrease patients’ discomfort following the impacted mandibular third molar extraction.
Trial registration
ClinicalTrials.gov Identifier: NCT05791721. Date of Registration: 28/03/2023 (retrospectively registered).



Similar content being viewed by others
References
Martins-de-Barros AV, Barros AM, Siqueira AK, Lucena EE, Sette de Souza PH, Araújo FA (2021) Is Dexamethasone superior to Ketorolac in reducing pain, swelling and trismus following mandibular third molar removal? A split mouth triple-blind randomized clinical trial. Med Oral Patol Oral Cir Bucal 26:e141–e150. https://doi.org/10.4317/medoral.24088
Rodrigues ÉD, Pereira GS, Vasconcelos BC, Ribeiro RC (2019) Effect of preemptive dexamethasone and etoricoxib on postoperative period following impacted third molar surgery - a randomized clinical trial. Med Oral Patol Oral Cir Bucal 24:e746–e751. https://doi.org/10.4317/medoral.23095
Cetira Filho EL, Carvalho FSR, de Barros Silva PG, Barbosa DAF, Alves Pereira KM, Ribeiro TR et al (2020) Preemptive use of oral nonsteroidal anti-inflammatory drugs for the relief of inflammatory events after surgical removal of lower third molars: a systematic review with meta-analysis of placebo-controlled randomized clinical trials. J Craniomaxillofac Surg 48:293–307. https://doi.org/10.1016/j.jcms.2020.01.016
Pergolizzi JV, Magnusson P, LeQuang JA, Gharibo C, Varrassi G (2020) The pharmacological management of dental pain. Expert Opin Pharmacother 21:591–601. https://doi.org/10.1080/14656566.2020.1718651
Kim K, Brar P, Jakubowski J, Kaltman S, Lopez E (2009) The use of corticosteroids and nonsteroidal antiinflammatory medication for the management of pain and inflammation after third molar surgery: a review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 107:630–640. https://doi.org/10.1016/j.tripleo.2008.11.005
Ferrer MD, Busquets-Cortés C, Capó X, Tejada S, Tur JA, Pons A et al (2019) Cyclooxygenase-2 inhibitors as a therapeutic target in inflammatory diseases. Curr Med Chem 26:3225–3241. https://doi.org/10.2174/0929867325666180514112124
González-Barnadas A, Camps-Font O, Martín-Fatás P, Figueiredo R, Gay-Escoda C, Valmaseda-Castellón E (2020) Efficacy and safety of selective COX-2 inhibitors for pain management after third molar removal: a meta-analysis of randomized clinical trials. Clin Oral Investig 24:79–96. https://doi.org/10.1007/s00784-019-02910-3
Schjerning AM, McGettigan P, Gislason G (2020) Cardiovascular effects and safety of (non-aspirin) NSAIDs. Nat Rev Cardiol 17:574–584. https://doi.org/10.1038/s41569-020-0366-z
la Torre LF, Franco-González DL, Brennan-Bourdon LM, Molina-Frechero N, Alonso-Castro ÁJ, Isiordia-Espinoza MA (2021) Analgesic efficacy of etoricoxib following third molar surgery: a meta-analysis. Behav Neurol 2021:9536054. https://doi.org/10.1155/2021/9536054
Albuquerque AFM, Fonteles CSR, do Val DR, Chaves HV, Bezerra MM, Pereira KMA, et al (2017) Effect of pre-emptive analgesia on clinical parameters and tissue levels of TNF-α and IL-1β in third molar surgery: a triple-blind, randomized, placebo-controlled study. Int J Oral Maxillofac Surg 46:1615–1625. https://doi.org/10.1016/j.ijom.2017.05.007
Costa FW, Soares EC, Esses DF, Silva PG, Bezerra TP, Scarparo HC et al (2015) A split-mouth, randomized, triple-blind, placebo-controlled study to analyze the pre-emptive effect of etoricoxib 120 mg on inflammatory events following removal of unerupted mandibular third molars. Int J Oral Maxillofac Surg 44:1166–1174. https://doi.org/10.1016/j.ijom.2015.06.012
Isola G, Matarese G, Alibrandi A, Dalessandri D, Migliorati M, Pedullà E et al (2019) Comparison of effectiveness of etoricoxib and diclofenac on pain and perioperative sequelae after surgical avulsion of mandibular third molars: a randomized, controlled, clinical trial. Clin J Pain 35:908–915. https://doi.org/10.1097/AJP.0000000000000748
Xie L, Sang L, Li Z (2021) Does low dose of etoricoxib play pre-emptive analgesic effect in third molar surgery? A randomized clinical trial. BMC Oral Health 21:462. https://doi.org/10.1186/s12903-021-01837-0
Brown JD, Daniels SE, Bandy DP, Ko AT, Gammaitoni A, Mehta A et al (2013) Evaluation of multiday analgesia with etoricoxib in a double-blind, randomized controlled trial using the postoperative third-molar extraction dental pain model. Clin J Pain 29:492–498. https://doi.org/10.1097/AJP.0b013e318260c144
Xie L, Li Z, Shang ZJ (2021) Preemptive oral etoricoxib on health-related quality of life after mandibular third molar surgery: a randomized, double-blind, placebo-controlled clinical trial. Biomed Res Int 2021:8888151. https://doi.org/10.1155/2021/8888151
Peskar BM, Ehrlich K, Egger T, Sattler W (2006) Dexamethasone impairs the gastric mucosal integrity in rats treated with a cyclooxygenase-1 but not with a cyclooxygenase-2 inhibitor. Pharmacology 76:180–184. https://doi.org/10.1159/000091575
Tanabe T, Tohnai N (2002) Cyclooxygenase isozymes and their gene structures and expression. Prostaglandins Other Lipid Mediat 68–69:95–114. https://doi.org/10.1016/s0090-6980(02)00024-2
Falci SGM, Lima TC, Martins CC, Santos C, Pinheiro MLP (2017) Preemptive effect of dexamethasone in third-molar surgery: a meta-analysis. Anesth Prog 64:136–143. https://doi.org/10.2344/anpr-64-05-08
Santos BFE, Costa FO, Pinto Júnior AAC, Araújo AVA, Cyrino RM, Cota LOM (2021) Postoperative pain and edema control following different protocols of preemptive analgesia in the surgical removal of impacted third molars: A triple-blind parallel randomized placebo-controlled clinical trial. J Craniomaxillofac Surg 49:694–704. https://doi.org/10.1016/j.jcms.2021.01.002
Lima CAA, Favarini VT, Torres AM, da Silva RA, Sato FRL (2017) Oral dexamethasone decreases postoperative pain, swelling, and trismus more than diclofenac following third molar removal: a randomized controlled clinical trial. Oral Maxillofac Surg 21:321–326. https://doi.org/10.1007/s10006-017-0635-0
Al-Dajani M (2017) Can preoperative intramuscular single-dose dexamethasone improve patient-centered outcomes following third molar surgery? J Oral Maxillofac Surg 75:1616–1626. https://doi.org/10.1016/j.joms.2017.03.037
Kissin I (2000) Preemptive analgesia. Anesthesiology 93:1138–1143. https://doi.org/10.1097/00000542-200010000-00040
Sotto-Maior BS, Senna PM, de Souza Picorelli Assis NM (2011) Corticosteroids or cyclooxygenase 2-selective inhibitor medication for the management of pain and swelling after third-molar surgery. J Craniofac Surg 22:758–762. https://doi.org/10.1097/SCS.0b013e318207f3fe
Moher D, Hopewell S, Schulz KF, Montori V, Gøtzsche PC, Devereaux PJ et al (2010) CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ 340:c869. https://doi.org/10.1136/bmj.c869
World Medical Association (2013) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310:2191–2194. https://doi.org/10.1001/jama.2013.281053
Lima CJ, Silva LC, Melo MR, Santos JA, Santos TS (2012) Evaluation of the agreement by examiners according to classifications of third molars. Med Oral Patol Oral Cir Bucal 17:e281-286. https://doi.org/10.4317/medoral.17483
Markovic A, Todorovic L (2007) Effectiveness of dexamethasone and low-power laser in minimizing oedema after third molar surgery: a clinical trial. Int J Oral Maxillofac Surg 36:226–229. https://doi.org/10.1016/j.ijom.2006.10.006
Momesso GAC, Grossi-Oliveira GA, Silva WPP, Akira R, Chiba F, Polo TOB et al (2021) A triple-blind randomized clinical trial of different associations between dexamethasone and non-steroids anti-inflammatories for preemptive action in third molar extractions. Sci Rep 11:24445. https://doi.org/10.1038/s41598-021-04068-z
Boonstra AM, Schiphorst Preuper HR, Balk GA, Stewart RE (2014) Cut-off points for mild, moderate, and severe pain on the visual analogue scale for pain in patients with chronic musculoskeletal pain. Pain 155:2545–2550. https://doi.org/10.1016/j.pain.2014.09.014
Dourado GB, Volpato GH, de Almeida-Pedrin RR, PedronOltramari PV, Freire Fernandes TM, de Castro Ferreira Conti AC (2021) Likert scale vs visual analog scale for assessing facial pleasantness. Am J Orthod Dentofacial Orthop 160:844–852. https://doi.org/10.1016/j.ajodo.2020.05.024
Carrillo JS, Calatayud J, Manso FJ, Barberia E, Martinez JM, Donado M (1990) A randomized double-blind clinical trial on the effectiveness of helium-neon laser in the prevention of pain, swelling and trismus after removal of impacted third molars. Int Dent J 40:31–36
Almeida RAC, Lemos CAA, de Moraes SLD, Pellizzer EP, Vasconcelos BC (2019) Efficacy of corticosteroids versus placebo in impacted third molar surgery: systematic review and meta-analysis of randomized controlled trials. Int J Oral Maxillofac Surg 48:118–131. https://doi.org/10.1016/j.ijom.2018.05.023
Erdil A, Akbulut N, Altan A, Demirsoy MS (2021) Comparison of the effect of therapeutic elastic bandage, submucosal dexamethasone, or dexketoprofen trometamol on inflammatory symptoms and quality of life following third molar surgery: a randomized clinical trial. Clin Oral Investig 25:1849–1857. https://doi.org/10.1007/s00784-020-03487-y
Nehme W, Fares Y, Abou-Abbas L (2021) Piezo-surgery technique and intramuscular dexamethasone injection to reduce postoperative pain after impacted mandibular third molar surgery: a randomized clinical trial. BMC Oral Health 21:393. https://doi.org/10.1186/s12903-021-01759-x
Fernandes IA, de Souza GM, Pinheiro MLP, Falci SGM (2019) Intramuscular injection of dexamethasone for the control of pain, swelling, and trismus after third molar surgery: a systematic review and meta-analysis. Int J Oral Maxillofac Surg 48(5):659–668. https://doi.org/10.1016/j.ijom.2018.09.014
Shoohanizad E, Parvin M (2020) Comparison of the effects of dexamethasone administration on postoperative sequelae before and after “third molar” extraction surgeries. Endocr Metab Immune Disord Drug Targets 20:356–364. https://doi.org/10.2174/1871530319666190722120405
Daniels SE, Bandy DP, Christensen SE, Boice J, Losada MC, Liu H et al (2011) Evaluation of the dose range of etoricoxib in an acute pain setting using the postoperative dental pain model. Clin J Pain 27:1–8. https://doi.org/10.1097/ajp.0b013e3181ed0639
Canellas J, Ritto FG, Tiwana P (2022) Comparative efficacy and safety of different corticosteroids to reduce inflammatory complications after mandibular third molar surgery: a systematic review and network meta-analysis. Br J Oral Maxillofac Surg 60:1035–1043. https://doi.org/10.1016/j.bjoms.2022.05.003
Gulen M, Satar S, Acehan S, Yildiz D, Aslanturkiyeli EF, Aka Satar D et al (2022) Perfusion index versus visual analogue scale: as an objective tool of renal colic pain in emergency department. Heliyon 8:e10606. https://doi.org/10.1016/j.heliyon.2022.e10606
Author information
Authors and Affiliations
Contributions
I.M., A.M., and B.J. conceptualized the research. A.T., B.I., and N.M. searched and prepared the literature data. I.M., A.M., A.T., and T.M. designed the research methodology. B.M. performed the formal analysis. I.M., A.M., and B.J. performed the investigation. I.M., B.I., T.M., and N.M. registered and stored the study data. I.M., A.M., A.T., and T.M. wrote the original manuscript draft. I.M. and B.M. prepared tables and figures. A.M., I.M., B.I., and N.M. critically reviewed and edited the manuscript. A.M., B.J., and I.M. supervised the research.
Corresponding author
Ethics declarations
Ethics approval
This study was performed according to the principles of the 1964 Helsinki Declaration, as well as its later amendments. Ethical approval was granted by the Ethics Committee of Belgrade School of Dental Medicine (Date 15/09/2021/No. 36/22).
Consent to participate
Informed consent was obtained from all the participants included in the clinical trial.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mijailovic, I., Janjic, B., Milicic, B. et al. Comparison of preemptive etoricoxib and dexamethasone in third molar surgery – a randomized controlled clinical trial of patient-reported and clinical outcomes. Clin Oral Invest 27, 5263–5273 (2023). https://doi.org/10.1007/s00784-023-05146-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05146-4