Abstract
Purpose
To assess goblet cell size and numbers in relation to the extent of multilayering of conjunctival impression cytology (CIC) samples as a basis for reducing variability in image selection for goblet cell density (GCD) estimates.
Methods
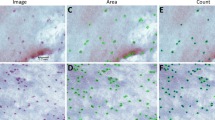
CIC was undertaken immediately postmortem off the superior bulbar conjunctiva of healthy young adult rabbits onto Millicell-CM Biopore filter units. After fixation with buffered glutaraldehyde and Giemsa staining, two × 200 images were selected from each sample representative of either slight multilayering or substantial multilayering, projected at × 1000, an overlay of the outlines of the goblet cells was made, and their dimensions and areas were measured.
Results
From measures of 4918 goblet cells, the average value (+/− SD) for the longest dimension was 17.7 ± 6.4 μm and 14.6 ± 5.3 μm for the shortest dimension. The GCD values ranged from 210 to 2069/mm2, with a mean of 1074 ± 601/mm2, but was lower for slightly multilayered images (at 537 ± 239 cells/mm ) compared with multilayered regions (at 1612 ± 601 cells/mm2; p < 0.001). The measured areas ranged from 72 to 491 μm2, with average values from any particular image ranging from 110 to 370 μm2, which were inversely correlated with the estimated GCD (Spearman’s rho = − 0.722, p < 0.05).
Conclusions
Larger goblet cells but in fewer numbers were predictably found across the filter surface where there were fewer layers of cells and vice versa. This difference could be considered in selection of images for counts of goblet cells from CIC specimens.






Similar content being viewed by others
References
Wolffsohn JS, Arita R, Chalmers R, Djalilian A, Dogru M, Dumbleton K, Gupta PK, Karpecki P, Lazreg S, Pult H, Sullivan BD, Tomlinson A, Tong L, Villani E, Yoon KC, Jones L, Craig JP (2017) TFOS DEWS II diagnostic methodology report. Ocul Surf. 15:539–574. https://doi.org/10.1016/j.jtos.2017.05.001
Baudouin C, Rolando M, Benitez Del Castillo JM, Messmer EM, Figueiredo FC, Irkec M, Van Setten G, Labetoulle M (2019) Reconsidering the central role of mucins in dry eye and ocular surface diseases. Prog Retin Eye Res 71:68–87. https://doi.org/10.1016/j.preteyeres.2018.11.007
Chao C, Richdale K, Jalbert I, Doung K, Gokhale M (2017) Non-invasive objective and contemporary methods for measuring ocular surface inflammation in soft contact lens wearers-a review. Cont Lens Anterior Eye 40:273–282. https://doi.org/10.1016/j.clae.2017.05.008
Sapkota K, Franco S, Sampaio P, Lira M (2015) Goblet cell density association with tear function and ocular surface physiology. Cont Lens Anterior Eye. 38:240–244. https://doi.org/10.1016/j.clae.2015.01.016
Doughty MJ (2012) Goblet cells of the normal human bulbar conjunctiva and their assessment by impression cytology sampling. Ocul Surf 10:149–169. https://doi.org/10.1016/j.jtos.2012.05.001
Doughty MJ (2015) Assessment of consistency in assignment of severe (grade 3) squamous metaplasia to human bulbar conjunctiva impression cytology cell samples. Ocul Surf 13:284–297. https://doi.org/10.1016/j.jtos.2015.05.003
Egbert PR, Lauber S, Maurice DM (1977) A simple conjunctival biopsy. Am J Ophthalmol 84:798–801. https://doi.org/10.1016/0002-9394(77)90499-8
Driot JY, Bonne C (1992) Beneficial effects of a retinoic acid analog, CBS-211 A, on an experimental model of keratoconjunctivitis sicca. Invest Ophthalmol Vis Sci 33:190–195
Doughty MJ (2015) A systematic assessment of goblet cell sampling of the bulbar conjunctiva by impression cytology. Exp Eye Res 136:16–28. https://doi.org/10.1016/j.exer.2015.04.007
Doughty MJ (2016) Giemsa-based cytological assessment of area, shape and nucleus:cytoplasm ratio of goblet cells of rabbit bulbar conjunctiva. Biotech Histochem 91:501–509. https://doi.org/10.1080/10520295.2016.1247988
Eördögh R, Schwendenwein I, Tichy A, Nell B (2015) Impression cytology: a novel sampling technique for conjunctival cytology of the feline eye. Vet Ophthalmol 18:276–284. https://doi.org/10.1111/vop.12244
Doughty MJ (2017) A grid-based nucleus counting method for estimates of the density of superficial conjunctival cells from impression cytology samples taken from normal healthy human eyes. Curr Eye Res 42:1228–1234. https://doi.org/10.1080/02713683.2017.1307414
Doughty MJ (2011) On the use of NIH image J for objective assessment of conjunctival cell and nucleus dimensions of impression cytology samples. Eye Contact Lens 37:50–56. https://doi.org/10.1097/ICL.0b013e31820c6df0
Doughty MJ (2011) Objective assessment of conjunctival squamous metaplasia by measures of cell and nucleus dimensions. Diagn Cytopathol 39:409–423. https://doi.org/10.1002/dc.21404
Chao C, Golebiowski B, Stapleton F, Zhou X, Chen S, Madigan MC (2018) Conjunctival MUC5AC+ goblet cell index: relationship with corneal nerves and dry eye. Graefes Arch Clin Exp Ophthalmol 256:2249–2257. https://doi.org/10.1007/s00417-018-4065-y
Labbé A, Pauly A, Liang H, Brignole-Baudouin F, Martin C, Warnet JM, Baudouin C (2006) Comparison of toxicological profiles of benzalkonium chloride and polyquaternium-1: an experimental study. J Ocul Pharmacol Ther 22:267–278. https://doi.org/10.1089/jop.2006.22.267
Doughty MJ (2020) On the in vivo assessment of goblet cells of the human bulbar conjunctiva by confocal microscopy-a review. Cont Lens Anterior Eye in press. https://doi.org/10.1016/j.clae.2020.01.004
Tseng SC (1985) Staging of conjunctival squamous metaplasia by impression cytology. Ophthalmology. 92:728–733. https://doi.org/10.1016/s0161-6420(85)33967-2
Nelson JD (1988) Impression cytology. Cornea. 7:71–81
Funding
This research was funded in part by a grant from the National Sciences and Engineering Research Council (NSERC, grant number A2579).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest.
Ethical approval
For these studies, all applicable international, national and institutional guidelines for the care and use of animals were followed. All procedures performed were in accord with the ethical standards of the University of Waterloo, Canada, specifically as formally assessed by the Animal Care Committee at that institution.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Doughty, M.J. Assessment of goblet cell size and density in relation to epithelial cell (multi)layering on conjunctival impression cytology samples. Graefes Arch Clin Exp Ophthalmol 258, 1727–1734 (2020). https://doi.org/10.1007/s00417-020-04725-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04725-5