Summary
A balanced 2-way crossover study involving 12 elderly volunteers was used to determine the pharmacokinetic parameters of the antidepressant tianeptine following a single dose administered by oral and intravenous routes. Pharmacokinetic parameters of metabolite MC5, the C5 side-chain β-oxidation product of tianeptine, were simultaneously determined.
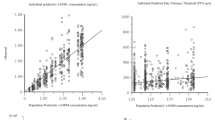
Tianeptine was absorbed rapidly [peak concentration (Cmax) = 261 ± 70 µg/L, and time to Cmax (tmax) = 1.59 ± 0.63 hours] and efficiently, with a bioavailability of 84.6 ± 14.0%.
After intravenous administration, following limited distribution in the tissues [volume of distribution at steady-state (Vdss) = 0.44 ± 0.08 L/kg] tianeptine was rapidly eliminated from the plasma (t½z = 3.1 ± 1.1 hours) mainly through biotransformation [renal clearance (CLR) < 1 ml/min; total clearance (CLT) = 139 ± 28 ml/min].
The MC5 metabolite appeared 0.18 ± 0.04 hours following the beginning of intravenous administration (tmax 2.96 ± 0.95 hours; Cmax = 83 ± 29 µg/L). MC5 was excreted with a terminal half-life of 11.9 ± 8.0 hours; its renal clearance was low (3.1 ± 2.3 ml/min). After oral administration, the pharmacokinetics of the MC5 metabolite remained unchanged, except for tmax which was delayed (tmax = 3.70 ± 0.61 hours).
This study has shown that the bioavailability of tianeptine is high in elderly subjects. Apart from drowsiness observed only following intravenous administration, tianeptine was well tolerated, particularly with regard to cardiovascular effects.
Similar content being viewed by others
References
Baccino E, Bressollette L, Mottier D, Mocaër E, Salvadori C, et al. Effects of the antidepressant tianeptine on platelet 5-HT uptake in elderly patients. Abstract. 5th International Meeting on Clinical Pharmacology in Psychiatry, University of Tromso, Norway, June 26–29, p. 28, 1988
Benet LZ, Galeazzi RL. Non-compartmental determination of the steady-state volume of distribution. Journal of Pharmaceutical Sciences 68: 1071–1073, 1979
Defrance R, Marey C, Kamoun A. Antidepressant and anxiolytic activities of tianeptine. An overview of clinical trials. Clinical Neuropharmacology 11 (Suppl. 2): S74–S82, 1988
Delalleau B, Dulcire C, Le Moine P, Kamoun A. Analysis of the side-effects of tianeptine. Clinical Neuropharmacology 11 (Suppl. 2): S83–S89, 1988
Gomeni R. Pharm: an interactive graphic program for individual and population pharmacokinetic parameter estimation. Computers in Biology and Medicine 14: 25–34, 1984
Grislain L, Gele P, Bertrand M, Luijten W, Bromet N, et al. The metabolic pathways of tianeptine, a new antidepressant, in healthy volunteers. Drug Metabolism and Disposition, in press, 1991
Kato G, Weitsch AF. Neurochemical profile of tianeptine, a new antidepressant drug. Clinical Neuropharmacology 11 (Suppl. 2): S43–S50, 1988
Lôo H, Malka R, Defrance R, Barrucand D, Benard JY, et al. Tianeptine and amitriptyline: controlled double-blind trial in depressed alcoholic patients. Neuropsychobiology 19: 79–85, 1988
Lôo H, Deniker P. Position of tianeptine among antidepressive chemotherapies. Clinical Neuropharmacology 11 (Suppl. 2): S97–S102, 1988
Mennini T, Mocaer E, Garattini S. Tianeptine, a selective enhancer of serotonin uptake in rat brain. Naunyn Schmiedeberg’s Archives of Pharmacology 336: 478–482, 1987
Nicot G, Lachatre G, Gonnet C, Mallon J, Mocaër E. Paired ion extraction and high performance liquid Chromatographic determination of tianeptine and its metabolites in human plasma, urine and tissues. Journal of Chromatography Biomedical Applications 381: 115–126, 1986
Renaud B, Mocaër E, Weitsch AF, Kato G, Mennini T. Stimulation of serotonin uptake induced by a new antidepressant: Proceedings of the ECNP Constituent meeting, Brussels, May 1987. Pharmacopsychiatry 21: 66, 1988
Royer RJ, Royer-Morrot MJ, Paille F, Barrucand D, Schmitt J, et al. Tianeptine and its main metabolite: pharmacokinetics in chronic alcoholism and cirrhosis. Clinical Pharmacokinetics 16: 186–191, 1989
Salvadori C, Ward C, Defrance R, Hopkins R. The pharmacokinetics of the antidepressant tianeptine and its main metabolite: absolute bioavailability and influence of alcohol co-administration. Fundamental and Clinical Pharmacology 4: 115–126, 1990
Zini R, Morin D, Salvadori C, Tillement JP. Tianeptine binding to human plasma proteins and plasma from risk populations. British Journal of Clinical Pharmacology 29: 9–18, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carlhant, D., Le Garrec, J., Guedes, Y. et al. Pharmacokinetics and Bioavailability of Tianeptine in the Elderly. Drug Invest. 2, 167–172 (1990). https://doi.org/10.1007/BF03259191
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03259191