Abstract
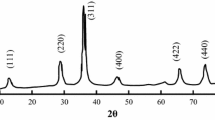
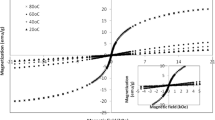
Nanopowders of cobalt iron oxide (CoFe2O4) were successfully fabricated by the co-precipitation method followed by a technique to prevent particle agglomeration. Particle sizes were in the range of 24 to 44 nm. The size of cobalt iron oxide particles decreases with increasing the concentration of the precipitation agent. The crystal structure was confirmed by X-ray diffraction (XRD), the chemical composition by energy dispersive spectroscopy (EDS), and phase changes by thermogravimetric differential thermal analysis (TGA-TDA). The particle morphology was analyzed by scanning electron microscopy (SEM). Magnetic properties were investigated by SQUID magnetometry and Mössbauer spectroscopy. Being nearly monodisperse and non-agglomerated the prepared cobalt iron oxide powders are the base for synthesizing magnetoelectric composites embedded in a ferroelectric BaTiO3 matrix.
Similar content being viewed by others
References
H. Yue, L. Yong, F. Chunlong, Y. Zhi, Z. Lu, X. Rui, Y. Di and S. Jing, J. Appl. Phys. 108, 084312 (2010).
A. Franco and F. C. e Silva, Appl. Phys Lett. 96, 172505 (2010).
R. M. Mohamed, M. M. Rashad, F. A. Haraz, and W. Sigmund, J. Magn. Mag. Mater. 322(14) 2058 (2010).
W. D. Callister, Material Science and Engineering, an Introduction, 5 th Ed. John Wiley & Sons, p. 684 (2000).
M. M. el-Okr, M. A. Salem, M. S. Salim, R. M. El-Okr, M. Ashoush, and H. M. Talaat, J. Magn. Magn. Mater. 323, 920 (2011).
R. M. Bozorth, E. F. Tilden, and A. J. Williams, Phys. Rev. 99, 1788 (1955).
C. A. F. Vaz, J. Hoffman, C. H. Ahn, and R. Ramesh, Adv. Mater. 22, 2900 (2010).
G. V. Duong, R. Groessinger, and R. S. Turtelli, J. Magn. Magn. Mater. 310, 1157 (2007).
V. V. Shvartsman, F. Alawneh, P. Borisov, D. Kozodaev, and D. C. Lupascu, Smart. Mater. Struct. 20, 075006 (2011).
Y. Zhang, Z. Yang, D. Yin, Y. Liu, C. Fei, R. Xiong, J. Shi, and G. Yan, J. Magn. Magn. Mater. 322, 3470 (2010).
Z. Zi, Y. Sun, X. Zhu, Z. Yang, J. Dai, and W. Song, J. Magn. Magn. Mater. 321, 1251 (2009).
K. Maaz, A. Mumtaz, S. K. Hasanain, and A. Ceylan, J. Magn. Magn. Mater. 308, 289 (2007).
B. G. Toksha, S. E. Shirsath, S. M. Patange, and K. M. Jadhav, Solid. State Commun. 147, 479 (2008).
N. Hanh, O. K. Quy, N. P. Thuy, L. D. Tung, and L. Spinu, physica B 327, 382 (2003).
Giovanni Baldi, Daniele Bonacchi, Mauro Comes Franchini, Denis Gentili, Giada Lorenzi, Alfredo Ricci and Costanza Ravagli, Langmuir, 23, 4026–4028 (2007)
Natl. Bur. Stand. (U.S.) Monogr. 25, 9, 22, (1971).
E.P. Wohlfarth, Ferromagnetic Materials, Elsevier Science Publishers B.V. Vol. 3, p 196 (1982).
R. M. Persoons, E. De Grave, P. M. A. de Bakker, and R.E. Vandenberghe, Phys. Rev. B 47, 5894 (1993).
C. Vazquez-Vazquez, M. A. Lopez-Quintela, M. C. Bujan-Nunez, and J. Rivas, J. Nanopart. Res. 13, 1663 (2011).
A. Virden, S. Wells and K. O’Grady, J. Magnt. Magnt. Mater. 316, 768 (2007).
L. Neel, J. Phys. Soc. Japan suppl. 17, 676 (1962).
S. Bedanta and W. Kleemann, J. Phys. D: Appl. Phys. 42, 013001 (2009).
Acknowledgments
The authors appreciate fruitful discussions with Yanling Gao and Dr. Firas Alawneh. We are grateful to Anna Elsukova for TEM measurements. Morad Etier acknowledges financial support by Deutscher Akademischer Austauschdienst. This work was partly supported by DAAD-GRISEC program (Grant 50750877).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Etier, M.F., Shvartsman, V.V., Stromberg, F. et al. Synthesis and Magnetic Properties of Cobalt Ferrite Nanoparticles. MRS Online Proceedings Library 1398, 1–8 (2012). https://doi.org/10.1557/opl.2012.699
Published:
Issue Date:
DOI: https://doi.org/10.1557/opl.2012.699