Abstract
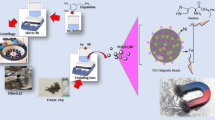
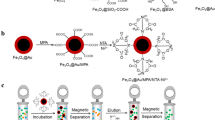
Nickel nanoparticles (NNPs) synthesized by a modified polyol method using ethylene glycol as a reducing agent, palladium chloride as a nucleating agent, and polyvinylpyrrolidone (PVP) as a protective agent were investigated as a potential magnetic adsorbent for the purification of hexahistidine-tagged (His6-tagged) recombinant proteins. The synthesis resulted in nanoparticles having an average diameter of 68 ± 28 nm. The x-ray diffraction pattern confirmed the presence of nickel metal, as well as the presence of unreacted nickel (II) hydroxide Ni(OH)2. Magnetic characterization showed that a magnetization saturation of 39.3 electromagnetic unit (emu)/g at 20,000 Oersted (Oe) was reached rapidly and that the material exhibited ferromagnetic behavior. Protein purification results showed that the synthesized NNPs were highly selective for binding to a His6-tagged recombinant protein single-domain antibody ToxA5.1. In addition, NNPs were used for four adsorption cycles without significant binding capacity losses. These particles have shown great potential such as being easily synthesized, cost-effective, and highly selective magnetic adsorbents for the purification of His6-tagged recombinant proteins.





Similar content being viewed by others
REFERENCES
E. Langer and J. Ranck: Capacity bottleneck squeezed by downstream processes. Bioprocess Int. 4, 14 (2006).
D. Low, R. O’Leary, and N.S. Pujar: Future of antibody purification. J. Chromatogr. B 848, 48 (2007).
M. Franzreb, M. Siemann-Herzberg, T.J. Hobley, and O.R.T. Thomas: Protein purification using magnetic adsorbent particles. Appl. Microbiol. Biotechnol. 70, 505 (2006).
D. Horák, M. Babič, H. Macková, and M.J. Beneš: Preparation and properties of magnetic nano- and microsized particles for biological and environmental separations. J. Sep. Sci. 30, 1751 (2007).
F. Ai, A. Yao, W. Huang, D. Wang, and X. Zhang: Synthesis of PVP-protected NiPd nanoalloys by modified polyol process and their magnetic properties. Physica E 42, 1281 (2010).
G.G. Couto, J.J. Klein, W.H. Schreiner, D.H. Mosca, A.J.A. de Oliveira, and A.J.G. Zarbin: Nickel nanoparticles obtained by a modified polyol process: Synthesis, characterization, and magnetic properties. J. Colloid Interface Sci. 311, 461 (2007).
H. Zhang, J. Ding, G. Chow, M. Ran, and J. Yi: Engineering magnetic properties of Ni nanoparticles by nonmagnetic cores. Chem. Mater. 21, 5222 (2009).
J. Porath and B. Olin: Immobilized metal ion affinity adsorption and immobilized metal ion affinity chromatography of biomaterials. Serum protein affinities for gel-immobilized iron and nickel ions. Biochemistry 22, 1621 (1983).
J. Porath: Immobilized metal ion affinity chromatography. Protein Expression Purif. 3, 263 (1992).
K.J. Carroll, J.U. Reveles, M.D. Shultz, S.N. Khanna, and E.E. Carpenter: Preparation of elemental Cu and Ni nanoparticles by the polyol method: An experimental and theoretical approach. J. Phys. Chem. C 115, 2656 (2011).
E.A. Baranova, A. Cally, A. Allagui, S. Ntais, and R. Wüthrich: Nickel particles with increased catalytic activity towards hydrogen evolution reaction. C.R. Chim. (2012). doi: 10.1016/j.crci.2012.02.003.
I.S. Lee, N. Lee, J. Park, B.H. Kim, Y.W. Yi, T. Kim, T.K. Kim, I.H. Lee, S.R. Paik, and T. Hyeon: Ni/NiO core/shell nanoparticles for selective binding and magnetic separation of histidine-tagged proteins. J. Am. Chem. Soc. 128, 10658 (2006).
J. Kim, Y. Piao, N. Lee, Y.I. Park, I.H. Lee, J.H. Lee, S.R. Paik, and T. Hyeon: Magnetic nanocomposite spheres decorated with NiO nanoparticles for a magnetically recyclable protein separation system. Adv. Mater. 22, 57 (2010).
L. Bai, J. Fan, Y. Cao, F. Yuan, A. Zuo, and Q. Tang: Shape-controlled synthesis of Ni particles via polyol reduction. J. Cryst. Growth 311, 2474 (2009).
G. Hussack, M. Arbabi-Ghahroudi, H. Van Faassen, J.G. Songer, K.K.S. Ng, R. MacKenzie, and J. Tanha: Neutralization of Clostridium difficile toxin A with single-domain antibodies targeting the cell receptor binding domain. J. Biol. Chem. 286, 8961 (2011).
M.D. Abràmoff, P.J. Magalhães, and S.J. Ram: Image processing with imageJ. Biophotonics Int. 11, 36 (2004).
U.K. Laemmli: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680 (1970).
B.D. Cullity: Elements of X-Ray Diffraction, 3rd ed. (Addison-Wesley Publishing Co., Boston, MA, 1956).
K.S. Chou and K.C. Huang: Studies on the chemical synthesis of nanosized nickel powder and its stability. J. Nanopart. Res. 3, 127 (2001).
A.R. West: Solid State Chemistry and its Applications (Wiley, Hoboken, NJ, 1984).
V. Gaberc-Porekar and V. Menart: Perspectives of immobilized-metal affinity chromatography. J. Biochem. Bioph. Methods 49, 335 (2001).
H. Block, B. Maertens, A. Spriestersbach, N. Brinker, J. Kubicek, R. Fabis, J. Labahn, and F. Schäfer: Immobilized-metal affinity chromatography (IMAC). A review, in Methods in Enzymology (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2009), p. 439.
C.W. Chen, H.L. Liu, J.C. Lin, and Y. Ho: Molecular dynamics simulations of metal ion binding to the His-tag motif. J. Chin. Chem. Soc. 52, 1281 (2005).
J.M. Nam, S.W. Han, K.B. Lee, X. Liu, M.A. Ratner, and C.A. Mirkin: Bioactive protein nanoarrays on nickel oxide surfaces formed by dip-pen nanolithography. Angew. Chem. Int. Ed. 43, 1246 (2004).
ACKNOWLEDGMENTS
The authors wish to thank Dr. Yun Liu for the SEM pictures, Dr. Tara Kell for the XRD scan, and Dr. Muralee Murugesu’s laboratory for the SQUID measurements. Financial support from the Natural Science and Engineering Council of Canada (NSERC) and Canadian Foundation of Innovation (CFI) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parisien, A., Al-Zarka, F., Hussack, G. et al. Nickel nanoparticles synthesized by a modified polyol method for the purification of histidine-tagged single-domain antibody ToxA5.1. Journal of Materials Research 27, 2884–2890 (2012). https://doi.org/10.1557/jmr.2012.323
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2012.323