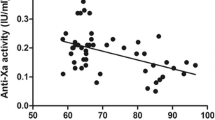
Background: Obese patients undergoing bariatric surgery are at a high risk of developing fatal pulmonary embolism or post-thrombotic syndrome.The prophylactic use of low molecular weight heparins (LMWHs) is correlated with a significant reduction in post-operative venous thrombosis in patients undergoing orthopedic or general surgery. In morbidity obese patients, the limited number of comparative trials are too sparse to allow a consensus on the effective dose and dosing schedule. Methods: In a prospective study to evaluate the effect of two doses of nadroparin as prophylaxis for venous thromboembolism following bariatric surgery, 60 consecutive patients undergoing Rouxen-Y gastric bypass were randomized to receive either 0.6 ml (5700 IU) or 1.0 ml (9500 IU) of nadroparin started pre-operatively and then given once daily post-operatively until discharge. Results: No statistically significant differences between the two groups were detected in any of the measured coagulation parameters either preoperatively or at days 1, 3 and 5 postoperatively.No thrombotic events were observed pre- or post- operatively, and no patient developed meta-thrombotic syndrome at the 3 and 6 months follow-up. No bleeding events occurred in the patients given the lower dose compared with two major hemorrhages in those given the higher dose. Conclusion: Our results indicate that 0.6 ml (5700 IU) of nadroparin once daily is safe and well-tolerated, and it is as effective in prophylaxis of venous thromboembolism as the higher dose of 1 ml (9500 IU), in such high risk patients.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalfarentzos, F., Yarmenitis, S., Kehagias, I. et al. Prophylaxis of Venous Thromboembolism Using Two Different Doses of Low-Molecular-Weight Heparin (Nadroparin) in Bariatric Surgery: A Prospective Randomized Trial. OBES SURG 11, 670–676 (2001). https://doi.org/10.1381/09608920160558588
Published:
Issue Date:
DOI: https://doi.org/10.1381/09608920160558588