Introduction
A starter culture can be defined as a microbial preparation of large numbers of cells of at least one microorganism to be added to a raw material to produce a fermented food by accelerating and steering its fermentation process. The group of lactic acid bacteria (LAB) occupies a central role in these processes, and has a long and safe history of application and consumption in the production of fermented foods and beverages. They cause rapid acidification of the raw material through the production of organic acids, mainly lactic acid. Also, their production of acetic acid, ethanol, aroma compounds, bacteriocins, exopolysaccharides, and several enzymes is of importance. In this way they enhance shelf life and microbial safety, improve texture, and contribute to the pleasant sensory profile of the end product. Lactic acid bacteria, propionibacteria, surface-ripening bacteria, yeasts, and molds are used as starter cultures for manufacturing of various fermented milk products. Starter cultures have a multifunctional role in dairy fermentations. The production of lactic acid, by fermenting lactose is the major role of dairy starters. The acid is responsible for development of characteristic body and texture of the fermented milk products, contributes to the overall flavour of the products, and enhances preservation. Diacetyl, acetaldehyde, acetic acid, also produced by the lactic starter cultures, contribute to flavor and aroma of the final product. Carbon-di-oxide produced by some hetero-fermentative lactic acid bacteria involves in very characteristics texturization in some fermented dairy products, viz. “eye” formation in cheeses. Development of flavor and changes in texture during ripening of cheeses is associated with enzymes originating from bacterial and fungal cultures, depending on the cheese variety. Dairy starters are also having some direct or indirect functional health promoting attributes, such as live probiotics, prebiotic exopolysaccharides and oligosaccharides, bioactive peptides and lipids, etc. Most of the cultured dairy products are produced using commercial starter cultures that have been selected for a variety of desirable properties in addition to rapid acid production. These may include flavor production, lack of associated off flavors, bacteriophage tolerance, ability to produce flavor during cheese ripening, salt tolerance, exo-polysaccharide production, bacteriocin production, temperature sensitivity, etc.
 |
Table 1: Starter cultures for fermented foods and beverages Click here to View table |
Novel Dairy Starters and Their Application
Milk contains many health promoting constituents including immunoglobulins, bioactive fatty acids and peptides amongst others. The healthy image of milk has resulted in dramatic growth in the diversification of dairy products in recent years and in huge increase in the varieties of products such as dairy desserts, flavoured milk drinks, cheeses, yoghurt etc. Apart from the milk components, the health attributes are associated with fermented and probiotic milks and dairy products. Milk has been preserved by fermentation through the action of lactic acid bacteria (LAB), which convert lactose to lactic acid and other organic acids, thereby lowering the pH and subsequently inhibiting the growth of pathogenic and spoilage bacteria. Moreover, these LAB produce a range of secondary metabolites, that can influence the products’ flavour, aroma and texture as well as antimicrobial peptides. These bacteria also possess a diverse complement of proteases and peptidases that aid in digestion of milk proteins. In addition, many bifidobacteria and lactobacilli are increasingly exploited in probiotic dairy products such as cheeses, yoghurt, milk drinks etc A novel trend in the food industry is to explore the use of functional starter cultures of LAB for the manufacture of fermented foods. Functional starter cultures are defined as starters that possess at least one inherent, functional property, aimed at improving the quality of the end product. Functional starter cultures of lactic acid bacteria are defined as strains that are used as starter cultures for food fermentation, and that are able to express certain functional properties which give an added value to the end-product (e.g. bacteriocin producers, exopolysaccharide producers, probiotic strains etc) (Bhat ZF and Bhat H, 2011). This functionality can contribute to microbial safety, or offer one or more organoleptic, technological, or nutritional and health advantages to the food. Promising examples are LAB that produces antimicrobial substances such as bacteriocins to assure food safety, sugar polymers to improve texture, desirable aromatic compounds to enhance taste properties, or strains that display probiotic effects. To develop such cultures, the biodiversity of traditional fermented foods and spontaneous fermentation processes is analyzed.
Metabolites Produced by LAB
LAB has relatively simple homo- or hetero- fermentative metabolism. These bacteria rely on lactose as their main carbohydrate source. Dairy LAB includes members of the genera Lacobacillus, Lactococcus, Leuconostoc and Sterptococcus.LAB fermentation yields primarily lactic acid, which plays a vital function in safeguarding food products. LAB metabolism beneficially affects the texture and flavour of fermented foods. The viscosity and texture of fermented dairy products can be greatly enhanced by the production of polysaccharides by LAB, while compounds such as diacetyl, ethanol, acetaldehyde, etc play vital roles in flavour development. In addition, many LAB produce compounds of human nutritional value as regular end products in their metabolisms, including some B-vitamins. Many strains of LAB and bidifobacteria produce other metabolites that promote human health. Bioactive peptides generated from milk proteins as a results of their proteinase and peptidases activity and production of CLA from linoleic acid by strains of lactobacilli and bifidobacteria. For instance, the meat isolates Lb. sakei CTC 494 and Lb. curvatus LTH 1174 turned out to be promising novel bacteriocin-producing starters for sausage fermentation, whereas Lb. amylovorus DCE 471 seemed more suitable as a starter for industrial sourdough fermentation. E. faecium RZS C5 has potential to be used as a bacteriocin-producing coculture for food fermentations. Moreover, modelling indicated how exopolysaccharide production by S. thermophilus LY03 in yoghurt may be optimised by adjusting the process conditions.
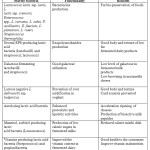 |
Table 2: Novel starter cultures and their applications Click here to View table |
Production of Aroma and Flavour
LAB contributes to the aroma and flavour of fermented products. They acidify the food, resulting in a tangy lactic acid taste, frequently exert proteolytic and lipolytic activities, and produce aromatic compounds. Wild strain starter cultures and NSLAB play an important role in flavour formation because they have a high biosynthetic capacity and produce aromatic compounds. The addition of NSLAB as adjunct cultures for cheese manufacturing increases the level of free amino acids, peptides, and free fatty acids, leading to flavour intensity and accelerated cheese ripening. Homofermentative LAB convert the available energy source (sugar) almost completely into lactic acid via pyruvate to produce energy and to equilibrate the redox balance. However, pyruvate can lead to the generation of many other metabolites such as acetate, ethanol, diacetyl, and acetaldehyde. In this way, LAB produce volatile substances that contribute to the typical flavour of certain fermented products, such as sourdough (determined by the lactate/acetate ratio), kefir and koumiss (ethanol), butter and buttermilk (diacetyl), and yoghurt (acetaldehyde).
EPS Production and Textural Improvement
Long-chain, high-molecular-mass polymers that dissolve or disperse in water to give a thickening or gelling properties are indispensable tools in food products formulation. Such polymers are also used for secondary effects that include emulsification, stabilization, and suspension of particulates, control of crystallization, and inhibition of syneresis, encapsulation, and film formation in foods. Most of the biothickeners in current use by the food industry are polysaccharides from plants (e.g. starch, pectin, locust bean gum, guar gum) or seaweeds (i.e. carrageenan, alginate). The animal proteinaceous hydrocolloids gelatin and casein are also used. The functional properties of these polymers in foods are determined by quite subtle structural characteristics. However, these polysaccharides may not always be readily available in the quality needed or their rheological properties may not exactly match those required. Alternative biothickeners are microbial exopolysaccharides, either associated with cell surface in form of capsules or secreted into the extracellularly in form of slime, referred to as capsular or slime exopolysaccharides, respectively. Microbial exopolysaccharides are biothickeners that can be added to a wide variety of food products, where they serve as viscosifying, stabilizing, emulsifying or gelling agents. Many food grade microorganisms also produce exopolysaccharides, in lactic acid bacteria, propionibacteria and bifidobacteria. The quest to find food ingredients with valuable bioactive properties has encouraged interest in exopolysaccharides from LAB. Functional food products that offer health and sensory benefits beyond their nutritional composition are becoming progressively more important to the food industry. The sensory benefits of exopolysaccharides are well established, and there is evidence for the health properties that are attributable to exopolysaccharides from LAB. However there is a wide variation in molecular structures of exopolysaccharides and the complexity of the mechanisms by which physical changes in foods and bioactive effects are elicited. To give a desired texture and mouth feel to yoghurt, skim-milk powder or whey is frequently added to the milk. Although the consumer does not consider this as unnatural, it represents an extra cost for the producer. In some countries, however, gelatine or plant (e.g., starch, pectin, guar gum, and alginate) and microbial polysaccharides (e.g., xanthan and gellan) are added. Polysaccharides increase the viscosity and firmness, improve the texture, reduce susceptibility to syneresis, and contribute to the mouthfeel of low-fat products. Some polysaccharides, e.g., plant carbohydrates, xanthan and gellan, have the additional advantage of being suitable to chemical modification to improve their rheological properties. However, the modified molecules are perceived by the consumer as unnatural and the food regulation of several European countries forbids the use of additives in, for instance, yoghurt. The in-situ production of natural texture-improving sugar polymers (exopolysaccharides) by LAB for the manufacturing of yoghurts, of sour cream and whipped toppings of ice cream and of low-fat Mozzarella are being explored. Functional, exopolysaccharide-producing starters of Lb. delbrueckii subsp. bulgaricus or S. thermophilus are promising. Current research is investigating the biodiversity of exopolysaccharides produced by LAB from artisan yoghurts, fermented milks, vegetables, and cereals, the conditions for optimal production, and their technological implementation in the industrial production of fermented foods .
Bacteriocins Production
Bacteriocins, ribosomally-synthesized peptides or proteins with antimicrobial activity, are produced by different groups of bacteria. Many lactic acid bacteria produce bacteriocins with broad spectra of inhibitions that offer potential applications in food preservation (Bhattacharya and Das, 2010). The use of these bacteriocins in food industry can reduce the dependency on chemical preservation as well as the intensity of heat treatments, resulting in naturally preserved foods rich in organoleptic and nutritional value. The live bacteriocin-producing LAB incorporation into a food gives it its own built in bio-preservation, thereby returning to a more natural method of shelf-life extension and improving the safety of food. However, there may be a lack of compatibility between the bacteriocin-producing strain and other cultures used in the fermentation. This was found to be the case for nisin-producing cultures, which inhibit the starter cultures required for cheese making. In an attempt to overcome this problem, the viability of using nisin-producing starters was assessed, but unfortunately these starters suffered from poor acid development, possessed inadequate proteolytic activity and often displayed an enhanced susceptibility to bacteriophage attack. The use of cultures to produce bacteriocins in situ as a means of bio-preservation has received a great deal of interest in recent times. The inhibition of L. monocytogenes in dry-fermented sausage using lyophilized bacteriocin-producing cultures as a protective adjunct has been investigated. To reflect realistic pathogen contamination levels, sausage meat was artificially spiked with a four strain cocktail. Lb. curvatus was more successful than Lc. lactis, as following 4 h fermentation. The utilisation of Carnobacterium strains to combat listerial contamination in cold-smoked salmon has been studied and has shown potential, but due to the emergence of resistance to Class II bacteriocins there has been an increased interest in the use of bacteriocin-negative Carnobacterium strains as protective cultures. Recently, a food-grade strain has been developed to produce both lacticin 3147 and lacticin 481. This strain addresses both the food safety and food improvement aspects of bacteriocin production. Significantly, the killing effect of this double producer was more pronounced, when tested against Lb. fermentum and L. monocytogenes, than either bacteriocin producer alone. The use of bacteriocin-producing cultures in foods can be very beneficial. While the choice of bacteriocin-producing culture may have to vary to meet with the particular microenvironment associated with certain foods, the net result of this technology can be the production of foods that have a built-in preservation system.
Lactose-negative Starters
In yoghurt production, lactose is converted by a yoghurt culture into lactic acid until a final pH of 4.2–4.5 is achieved. Upon storage, the pH can decrease below 4.0. This undesirable post-acidification, ascribed to Lb. delbrueckii subsp. bulgaricus, leads to an acid and bitter taste. Lactose-negative mutants of Lb. delbrueckii subsp. bulgaricus enable production of mild yoghurts since such cells can, given their proto-co-operation; only grow in the presence of actively lactose fermenting S. thermophilus.
Accelerated Ripening of Cheeses
The ripening of cheese is a slow and consequently an expensive process. The economic advantage of rapid development of more intense cheese flavour in shorter periods of time would be substantial. Lactic acid bacteria play a key role during ripening and can therefore be used as accelerating agents. This review describes the different strategies where lactic acid bacteria or their enzymes were used to reduce the ripening time of cheese. The advantages, limitations and technical feasibility as well as the commercial potential of the different approaches are also considered. During cheese maturation, several aromatic compounds are generated due to the action of endogenous milk enzymes as well as the proteolytic and lipolytic activities of LAB present in the cheese. The maturation stage is time and space consuming so that accelerated maturation techniques are being sought. Besides rational selection of the LAB starter and co-cultures, and the application of process conditions for optimal activity of the endogenous enzymes, the addition of exogenous enzymes (enzyme modified cheese) as well as the increased in situ autolysis of the LAB, represent alternative solutions. Autolysis of the starter cells is followed by the release of intracellular peptidases in the curd. Bacteriocins (see above) may cause bacteriolysis, for example by inducing autolysins or by deregulating enzyme action due to energetic deficiency, which will result in degradation of the cell wall and consequent cell lysis. Furthermore, genetic strategies exist for the induction of phage holins and lysins to promote lysis of the starter cells during maturation.
Reduction of Toxic or Antinutritive Factors
The fermentative action of specific LAB stains may lead to the removal of toxic or antinutritive factors, such as lactose and galactose from fermented milks to prevent lactose intolerance and accumulation of galactose). Other examples are the removal of raffinose, stachyose, and verbascose from soy to prevent flatulence and intestinal cramps, proteinase inhibitors from legumes and cereals to prevent maldigestion, phytic acid and tannins from cereals and legumes to increase mineral bioavailability, and natural toxins such as cyanogenic glucosides from cassava as well as biogenic amines from traditional fermented foods.
Production of Nutraceuticals
Nutraceuticals are food components that, through specific physiological action, contribute to the health of the consumer. Several nutraceuticals from bacterial origin have been added to food. Through strain selection and process optimisation, the activity of LAB can be modified to increase the content of nutraceuticals in fermented foods such as fermented dairy products. As an example, fermented milks can be produced with LAB starter strains that produce high amounts of low-calorie polyols so as to reduce the sugar content. Also, the use of oligosaccharide-producing LAB that produce sugar polymers with a controlled structure and chain length (and hence molecular mass) may yield fermented products with health applications. Health effects of such oligosaccharides are ascribed to their low-calorie character, their fibre-like nature, and their bifidogenic effect. Certain LAB, such as the yoghurt bacteria Lb. delbrueckii subsp. bulgaricus and S. thermophilus, are able to produce vitamins such as folate (Warrier, 2009). A controlled use of these bacteria may lead to dairy products with increased folate content. The proteolytic system of LAB can contribute to the liberation of health-enhancing bioactive peptides from milk. The latter may improve absorption in the intestinal tract, stimulate the immune system, exert antihypertensive or antithrombotic effects, display antimicrobial activity, or function as carriers for minerals, especially calcium.
Low Calorie Sugars Production
Mannitol is a polyol or sugar alcohol that is produced by several organisms. Mannitol is assumed to have several beneficial effects, as an antioxidant (protection against oxidative damage by oxygen radicals) and as a non-metabolizable sweetener. Mannitol can therefore be applied to foods leading to health-promoting effects (functional foods). Mannitol-producing lactic acid bacteria may directly be applied in the manufacture of foods and this may lead to fermented food products with an extra nutritional value. Mannitol is a non-toxic, nonhygroscopic sweetener estimated to be about half as sweet as sucrose. Human are incapable of metabolizing mannitol entirely and therefore do not suffer from hyperglycaemia. Many lactic acid bacteria can synthesize mannitol and the mode of synthesis is depended on the pathway used for hexose fermentation. These are Leuconostoc mesenteriodes, Leuconostoc pseudomesenteriodes, Lactobacillus plantarum etc. Mannitol production by lactic acid bacteria and other food-grade microorganisms offer several important advantages. Firstly, food-grade microorganisms and their products are directly applicable in food products, without any restriction. Secondly, there is no need for a careful separation of products and microorganisms, which would be the case if microorganisms are not of food grade. Thirdly, some lactic acid bacteria are claimed as beneficial in the gastrointestinal tract. Several lactic acid bacterial strains are known to produce mannitol. Some homofermentative lactic acid bacteria were found to produce small amounts of mannitol. In the presence of large amounts of glucose or sucrose, cell suspensions of Streptococcus mutans produce approximately 0.1–0.3 mg mannitol. Lactobacillus leichmanii also showed mannitol production. Since in homofermentative lactic acid bacteria the carbon flow from carbohydrates is directed mainly to lactate production, the formation of other fermentation products like mannitol is often only possible when strains are more or less hampered in the lactate production pathway. A lactate dehydrogenase-negative mutant of Lactobacillus plantarum produced 6 mM mannitol from 50 mM glucose whereas the wild type was not able to produce any mannitol from glucose. The best mannitol producing lactic acid bacteria show a heterofermentative metabolism. In the presence of fructose or sucrose Leuconostoc (pseudo) mesenteroides produces high levels of mannitol. Two other heterofermentative lactic acid bacteria, Lactobacillus sp. And Leuconostoc sp., also produced mannitol from fructose and sucrose. The fructose is bio converted to mannitol. Under optimized conditions Leuconostoc mesenteroides converted up to 95 % (mol/mol) of fructose consumed into mannitol.
Vitamins Production
Folate the salt of folic acid is vital component of the human diet and involve in the biosynthesis of nucleotides and co-factors in many metabolic reactions. As mammalian cells cannot synthesize folate, the folate synthesizing LAB can be used to ugment the folate content in fermented dairy products. Milk contains 20-50 microgram per liter folate and fermented milk contains higher amounts up to 150 µg per liter in yoghurt. Some starter cultures have been shown to synthesize folate and secreate into the medium during fermentation. These are Bifidobacterium longum, Lactobacillus acidophilus, L. delbrueckii ssp. bulgaricus, and Streptococcus thermophilus strains. Vitamin B12 is an important cofactor for the metabolism of fatty acids, amino acids, carbohydrates and nucleic acids. This vitamin is synthesized by propionibacteria. Vitamin K, an essential co-factor, is involved in the post-translational modification of glutamic acid residues in certain proteins, such as blood clotting proteins and proteins involve in tissue calcification. Genera Lactococcus, Lactobacillus, Leuconostoc, Bifidobacterium and Enterococcus synthesize the vitamin K. Such strains may be useful as dairy starters to increase the vitamin K content in fermented dairy products.
Bioactive Peptide Production
Bioactive peptides have been identified within the amino acid sequences of milk proteins and may be released through the action of digestive enzymes in the intestine, added proteases or lactic cultures. Such peptides potentially have a wealth of health-related activities such as antimicrobial, opiate, antithrombic, antihypertensive, mineral binding properties, etc. Milk proteins have received increasing attention as potential ingredients of health-promoting functional foods targeted at diet-related chronic diseases, such as cardiovascular disease, diabetes type two and obesity. To this end, growing interest has been focused on physiologically active peptides derived from milk proteins (Abdel-Salam, 2010). These peptides are inactive within the sequence of the parent protein molecule and can be liberated by gastrointestinal digestion of milk, fermentation of milk with proteolytic starter cultures or hydrolysis by proteolytic enzymes. Milk protein-derived peptides have been shown under in vitro and in vivo conditions to exert a number of activities affecting the digestive, endocrine, cardiovascular, immune and nervous systems. A great variety of naturally formed bioactive peptides have been found in fermented dairy products, such as yoghurt, sour milk and cheese. Recently, industrial-scale technologies suitable for the industrial production of bioactive milk peptides have been developed. In addition, a few commercial food products supplemented with milk protein-derived bioactive peptides have been launched on limited markets. Some of these products carry clinically documented benefits, in particular for reduction of mild hypertension. The multifunctional properties of milk peptides appear to offer considerable potential for the development of many similar products in the near future. Fermentation of milk with single industrial dairy cultures generated antioxidant activity in the whey fraction. The activity correlated positively with the degree of proteolysis suggesting that peptides were responsible for the antioxidative property. Fermentation of milk with a commercial starter culture mixture of five lactic acid bacteria (LAB) strains followed by hydrolysis with a microbial protease increased ACE inhibitory activity of the hydrolysate. Several novel ACE inhibitory peptides in milk fermented with Enterococcus faecalis strains isolated from raw milk. The proteolytic activity of several dairy LAB cultures and probiotic strains (Lactobacillus acidophilus, Bifidobacterium lactis and Lactobacillus casei) as determinant of growth and in vitro ACE-inhibitory activity in milk fermented with these single cultures. All the cultures released ACE inhibitor peptides during growth with a Bifidobacterium longum strain and the probiotic Lb. acidophilus strain showing the strongest ACE-inhibitory activity. ACE-inhibitory peptide from milk fermented with Lactobacillus delbrueckii ssp. Bulgaricus was identified and sequenced as Ser-Lys-Val-Tyr-Pro-Phe-Pro-Gly-Pro-Ile. In combination with Streptococcus salivarius ssp. thermophilus and Lactococcus lactis biovar. diacetylactis, a hypotensive peptide structure with a sequence of Ser-Lys-Val- Tyr-Pro was obtained from beta-casein.
Production of Conjugated Linoleic Acid (CLA)
CLA refers to mixture of positional and geometrical isomers of linoleic aicd. A number of these isomers have attracted considerable attention due to their potentially beneficial effects, including anticarcinogenic and anti-inflamatory activities and inhibition of the onset of diabetes. A number of LAB can also produce CLA and CLA are higher in fermented dairy produced than in unprocessed milk (Yadav et al., 2008). Although, CLA production is not a general feature of LAB and bifidobacteria, strains are now available that can efficiently convert exogenous linoleic acid to CLA and such strains can be used to increase the CLA content in fermented dairy products. The productivity of conjugated linoleic acid synthesis by bacteria of the Lactobacillus and Bifidobacterium genera is a strain-specific trait. Bifidobacterium synthesize a higher number of different CLA isomers compared Lactobacillus.
Probiotics for Functional Foods
Probiotics are products aimed at delivering living, potentially beneficial, bacterial cells to the gut ecosystem of humans and other animals, whereas prebiotics are non-digestible carbohydrates delivered in food to the large bowel to provide fermentable substrates for selected bacteria. Strains of LAB are the most common microbes employed as probiotics. Two principal kinds of probiotic bacteria, members of the genera Lactobacillus and Bifidobacterium, have been studied in detail. Most probiotic strains belong to the genus Lactobacillus. Probiotics have been evaluated in research studies in animals and humans with respect to antibiotic-associated diarrhoea, travellers’ diarrhoea, pediatric diarrhoea, inflammatory bowel disease, and irritable bowel syndrome (Arvind et al., 2008). It is possible that, in the future, probiotics will be used for different gastrointestinal diseases, vaginosis, or as delivery systems for vaccines, immunoglobulins, and other therapies. The basic notion behind probiotics is pretty straightforward: restore the depleted ecology of the intestine with new, healthful bacteria. Many different microorganisms are added to dairy products for their probiotic potential. Probiotic microorganisms designed for delivery in food or dairy products, via supplementation or fermentation. These include Lactobacilli, such as Lactobacillus acidophilus, L. casei, L. delbrueckii ssp. bulgaricus, L. reuteri, L. brevis, L. cellobiosus, L. carvatus, L. fermentum and L. plantarum; Gram-positive cocci, such as Lactococcus lactis ssp. cremoris, Streptococcus thermophilus, Enterococcus faecium, S. diacetylactis and S. intermedius and Bifidobacteria, such as B. fifidum, B. adolescentis, B. animalis, B. infantis, B. longum and B. thermophilum. Nonpathogenic microorganisms that occupy important niches in the host gut or tissues, such as yeasts, enterococci and Enterobacteriaceae, are used as human and animal probiotics. Though, Lactobacillus and Bifidobacterium are the most commonly species of bacteria used as probiotics for the production of fermented milks and other dairy products (Panesar and Shinde, 2010).
Human Probiotic Species and Strains
♦ Bifidobacterium breve Yakult
♦ Bifidobacterium lactis (BB12)
♦ Bifidobacterium longum (SBT2928, BB536)
♦ Lactobacillus acidophilus (NCFM, SBT2062)
♦ Lactobacillus casei (Shirota, CRL431, DN014001, immunits)
♦ Lactobacillus delbrueckii ssp. bulgaricus (2038)
♦ Lactobacillus fermentum
♦ Lactobacillus johnsonii (La1, Lj 1)
♦ Lactobacillus paracasei (CRL431, F19)
♦ Lactobacillus plantarum (299V)
♦ Lactobacillus reuteri (SD 2112)
♦ Lactobacillus rhamnosus (GG, 271, GR1)
♦ Lactobacillus salivarius (UCC118)
♦ Streptococcus thermophilus (1131)
Selection and Maintenance of Suitable Strains
Selecting for strains with interesting properties to be used as new, functional starter cultures may lead to an improved fermentation process and an enhanced quality of the end product. However, as it has been shown for bacteriocin-producing LAB, the success of using functional starter cultures in a particular food is strongly strain dependent. Rational selection of appropriate strains is crucial. Because of legislation and marketing reasons, the industrial application of carefully selected natural food isolates with functional properties seems attractive. Of course, selection of starter cultures must not only aim at expressing functional properties, but also at eliminating undesirable side effects, such as the formation of D-lactic acid or a racemate of lactic acid (DL), or the formation of biogenic amines. One of the challenges of using wild-type strains will be to allow large-scale production of fermented foods without losing their unique flavour and other traits. In the new generation of fermented foods, LAB with diverse physiological and metabolic traits is combined. Their metabolic and technological properties are often different from those of traditional starter cultures, so that appropriate production processes must be developed. The positive results observed with functional starters under optimal laboratory conditions are not necessarily obtained in practice, i.e. in the actual food matrix.
Conclusion
Novel insights into the metabolism of LAB offer perspectives for the application of a new generation novel starter cultures. Functional LAB starters may offer several health, marketing, and technological advantages. However, fundamental and applied research is still needed to optimally implement functional starter cultures in the existing production technology and to obtain quantitative data. Mathematical analysis of the kinetics of functional starter cultures may yield precious information about the relationship between the food environment and bacterial functionality, and may contribute to optimal strain selection and process design. This may result in better process control, enhanced food safety and quality, and reduction of economic losses.
References
- Cogan T.M. and Hill C., Cheese starter cultures, Ch.6 in: P.F. Fox, ed., Cheese: Chemistry, Physics and Microbiology, Vol. 1, General Aspects, 2nd ed., pp. 193-206. Chapman and Hall, London (1993).
- Heap H. A. and Lawrence R.C., New Zealand J. Dairy Technol., 11-16 (1976).
- Tamime A. Y. and Robinson R.K., Preservation and production of starter cultures., in : Yoghurt, Science and Technology., pp. 486-514. CRC Press, New York and Woodhead Pub. Ltd., Cambridge, UK (1999).
- de Vuyst L., Food Technol. Biotechnol., Application of Functional Starter Cultures, 38, 2, 105–112 (2000).
- Yadav H., Jain S. and Sinha P.R., J Dairy Res., 75, 189-95 (2008).
CrossRef - Arvind, Sinha P.R., Singh N.K. and Kumar R., Int J Prob. Preb., 4, 195-200 (2009).
- Abdel-Salam A.M., American J. Food Technol., 5, 86-99 (2010).
- Bhat Z.F. and Bhat H., Int J Dairy Sci., 6, 1-12 (2011).
- Bhattacharay S. and Das A., American J. Food Technol., 5, 111-120 (2010).

This work is licensed under a Creative Commons Attribution 4.0 International License.





