Abstract
Background
Diffuse malignant peritoneal mesothelioma (DMPM) is a subset of peritoneal mesothelioma with a poor clinical outcome. We performed a prognostic analysis in a cohort of DMPM patients treated homogeneously by cytoreductive surgery and intraperitoneal hyperthermic perfusion (IPHP).
Methods
Forty-nine DMPM patients who underwent 52 consecutive procedures were enrolled onto the study. Cytoreductive surgery was performed according to the peritonectomy technique, and the IPHP was performed with cisplatin plus doxorubicin or cisplatin plus mitomycin C. We assessed the correlation of the clinicopathologic variables (previous surgical score, age, sex, performance status, previous systemic chemotherapy, carcinomatosis extension, completeness of cytoreduction, IPHP drug schedule, mitotic count [MC], nuclear grade, and biological markers [epidermal growth factor receptor, p16, matrix metalloproteinase 2 and matrix metalloproteinase 9]) with overall and progression-free survival.
Results
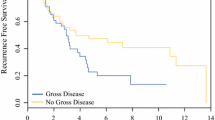
The mean age was 52 years (range, 22–74 years). The mean follow-up was 20.3 months (range, 1–89 months). Regarding the biological markers, the rates of immunoreactivity of epidermal growth factor receptor, p16, matrix metalloproteinase 2, and matrix metalloproteinase 9 were 94%, 60%, 100%, and 85%, respectively. The strongest factors influencing overall survival were completeness of cytoreduction and MC, whereas those for progression-free survival were performance status and MC. No biological markers were shown to be of prognostic value.
Conclusions
Completeness of cytoreduction, performance status, and MC seem to be the best determinants of outcome. These data warrant confirmation by a further prospective formal trial. No biological markers presented a significant correlation with the outcome. The overexpression of epidermal growth factor receptor, matrix metalloproteinase 2, and matrix metalloproteinase 9 and absent or reduced expression of p16 might be related to the underlining tumor kinetics of DMPM and warrant further investigation with other methods.





Similar content being viewed by others
References
Connelly RR, Spistas R, Myers MH, et al. Demographic patterns for mesothelioma in the United States. J Natl Cancer Inst 1987;78:1053–60
Antman K, Osteen R, Klegar K, et al. Early peritoneal mesothelioma: a treatable malignancy. Lancet 1985;11:977–81
Eltabbakh GH, Piver MS, Hempling RE, Recio FO, Intengen ME. Clinical picture, response to therapy, and survival of women with diffuse malignant peritoneal mesothelioma. J Surg Oncol 1999; 70:6–12
Sugarbaker PH, Welch LS, Mohamed F, Glehen O. A review of peritoneal mesothelioma at the Washington Cancer Institute. Surg Oncol Clin N Am 2003; 12:605–21
Kass ME. Pathology of peritoneal mesothelioma. In: Sugarbaker P, ed. Peritoneal Carcinomatosis: Drugs and Diseases. Boston: Kluwer Academic Publishers, 1996: 213–225
Weiss SW. Histological Typing of Soft Tissue Tumours. 2nd ed. Berlin: Springer-Verlag, 1994
Kannerstein M, Churg J. Peritoneal mesothelioma. Hum Pathol 1977; 8:83–94
Deraco M, Santoro N, Carraro O, et al. Peritoneal carcinomatosis: feature of dissemination. A review. Tumori 1999;85:1–5
Markman M, Kelsen D. Efficacy of cisplatin-based intraperitoneal chemotherapy as treatment of malignant peritoneal mesothelioma. J Cancer Res Clin Oncol 1992;118:547–50
Park BJ, Alexancer HR, Libutti SK, et al. Treatment of primary peritoneal mesothelioma by continuous hyperthermic peritoneal perfusion (CHPP). Ann Surg Oncol 1999;6:582–90
Sebbag G, Yan H, Shmookler BM, Chang D, Sugarbaker PH. Results of treatment of 33 patients with peritoneal mesothelioma. Br J Surg 2000;87:1587–93
Loggie BW, Fleming RA, McQuellon RP, Russell GB, Geisinger KR, Levine EA. Prospective trial for the treatment of malignant peritoneal mesothelioma. Am Surg 2001;67:999–1003
Sugarbaker PH. Peritonectomy procedures. Ann Surg 1995;221:29–42
Jacquet P, Sugarbaker PH. Current methodologies for clinical assessment of patients with peritoneal carcinomatosis. J Exp Clin Cancer Res 1996;15:49–58
Glehen O, Osinsky D, Cotte E, et al. Intraperitoneal chemohyperthermia using a closed abdominal procedure and cytoreductive surgery for the treatment of peritoneal carcinomatosis: morbidity and mortality analysis of 216 consecutive procedures. Ann Surg Oncol 2003;10:863–9
de Bree E, Rosing H, Beijnen JH, et al. Pharmacokinetic study of docetaxel in intraoperative hyperthermic i.p. chemotherapy for ovarian cancer. Anticancer Drugs 2003;14:103–10
Bozzetti F, Braga M, Gianotti L, Gavazzi C, Mariani L. Postoperative enteral versus parenteral nutrition in malnourished patients with gastrointestinal cancer: a randomised multicentre trial. Lancet 2001;358:1487–92
Ordonez NG. The immunohistochemical diagnosis of mesothelioma: a comparative study of epithelioid mesothelioma and lung adenocarcinoma. Am J Surg Pathol 2003;27:1031–51
Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. In: Sugarbaker PH, ed. Peritoneal Carcinomatosis: Principles of Management. Boston: Kluwer Academic, 1996:366–7
Deraco M, Casali P, Inglese MG, et al. Peritoneal mesothelioma treated by induction chemotherapy, cytoreductive surgery, and intraperitoneal hyperthermic perfusion. J Surg Oncol 2003;83:147–53
Feldman AL, Libutti SK, Pingpank JF, et al. Analysis of factors associated with outcome in patients with malignant peritoneal mesothelioma undergoing surgical debulking and intraperitoneal chemotherapy. J Clin Oncol 2003;21:4560–7
Jacquet P, Stephens AD, Averbach AM, et al. Analysis of morbidity and mortality in 60 patients with peritoneal carcinomatosis treated by cytoreductive surgery and heated intraoperative intraperitoneal chemotherapy. Cancer 1996;77:2622–9
Stephens AD, Alderman R, Chang D, et al. Morbidity and mortality analysis of 200 treatments with cytoreductive surgery and hyperthermic intraoperative intraperitoneal chemotherapy using the coliseum technique. Ann Surg Oncol 1999;6:790–6
Verwaal VJ, van Tinteren H, Ruth SV, Zoetmulder FA. Toxicity of cytoreductive surgery and hyperthermic intra-peritoneal chemotherapy. J Surg Oncol 2004;85:61–7
Shen P, Hawksworth J, Lovato J, et al. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy with mitomycin C for peritoneal carcinomatosis from nonappendiceal colorectal carcinoma. Ann Surg Oncol 2004;11:178–86
Sugarbaker PH, Welch LS, Mohamed F, Glehen O. A review of peritoneal mesothelioma at the Washington Cancer Institute. Surg Oncol Clin North Am 2003;12:605–21
Ramael M, Jacobs W, Weyler J, et al. Proliferation in malignant mesothelioma as determined by mitosis counts and immunoreactivity for proliferating cell nuclear antigen (PCNA). J Pathol 1994;172:247–53
Beer TW, Carr NJ, Whittaker MA, et al. Mitotic and in situ end-labeling apoptotic indices as prognostic markers in malignant mesothelioma. Ann Diagn Pathol 2000;4:143–8
Kerrigan SA, Turnnir RT, Clement PB, et al. Diffuse malignant epithelial mesotheliomas of the peritoneum in women: a clinicopathologic study of 25 patients. Cancer 2002;94:378–85
Curran D, Sahmoud T, Therasse P, van Meerbeeck J, Postmus PE, Giaccone G. Prognostic factors in patients with pleural mesothelioma: the European Organization for Research and Treatment of Cancer experience. J Clin Oncol 1998;16:145–52
Edwards JG, Abrams KR, Leverment JN, Spyt TJ, Waller DA, O’Byrne KJ. Prognostic factors for malignant mesothelioma in 142 patients: validation of CALGB and EORTC prognostic scoring systems. Thorax 2000;55:731–5
Hirao T, Bueno R, Chen CJ, et al. Alterations of the p16(INK4) locus in human malignant mesothelial tumors. Carcinogenesis 2002;23:1127–30
Kratzke RA, Otterson GA, Lincoln CE, et al. Immunohistochemical analysis of the p16INK4 cyclin-dependent kinase inhibitor in malignant mesothelioma. J Natl Cancer Inst 1995;87:1870–5
Dazzi H, Hasleton PS, Thatcher N, et al. Malignant pleural mesothelioma and epidermal growth factor receptor (EGF-R). Relationship of EGF-R with histology and survival using fixed paraffin embedded tissue and the F4, monoclonal antibody. Br J Cancer 1990;61:924–6
Trupiano JK, Geisinger KR, Willingham MC, et al. Diffuse malignant mesothelioma of the peritoneum and pleura, analysis of markers. Mod Pathol 2004;17:476–81
Schwartz GK. Invasion and metastases in gastric cancer: in vitro and in vivo models with clinical correlations. Semin Oncol 1996;23:316–24
Karakiulakis G, Papanikolaou C, Jankovic SM, et al. Increased type IV collagen-degrading activity in metastases originating from primary tumors of the human colon. Invasion Metastasis 1997;17:158–68
Cox G, Jones JL, O’Byrne KJ. Matrix metalloproteinase 9 and the epidermal growth factor signal pathway in operable non-small cell lung cancer. Clin Cancer Res 2000;6:2349–55
Edwards JG, McLaren J, Jones JL, et al. Matrix metalloproteinases 2 and 9 (gelatinases A and B) expression in malignant mesothelioma and benign pleura. Br J Cancer 2003;88:1553–9
Liu Z, Klominek J. Regulation of matrix metalloprotease activity in malignant mesothelioma cell lines by growth factors. Thorax 2003;58:198–203
Lumb PD, Suvarna SK. Metastasis in pleural mesothelioma. Immunohistochemical markers for disseminated disease. Histopathology 2004;44:345–52
Hirano H, Tsuji M, Kizaki T, et al. Expression of matrix metalloproteinases, tissue inhibitors of metalloproteinase, collagens, and Ki67 antigen in pleural malignant mesothelioma: an immunohistochemical and electron microscopic study. Med Electron Microsc 2002;35:16–23
Liu Z, Ivanoff A, Klominek J. Expression and activity of matrix metalloproteases in human malignant mesothelioma cell lines. Int J Cancer 2001;91:638–43
Acknowledgments
Supported by the Associazione Italiana Ricerca sul Cancro.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deraco, M., Nonaka, D., Baratti, D. et al. Prognostic Analysis of Clinicopathologic Factors in 49 Patients With Diffuse Malignant Peritoneal Mesothelioma Treated With Cytoreductive Surgery and Intraperitoneal Hyperthermic Perfusion. Ann Surg Oncol 13, 229–237 (2006). https://doi.org/10.1245/ASO.2006.03.045
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2006.03.045