Abstract
This study was aimed to design a simple and novel prototype device for the production of polymeric microparticles. To prove the effectiveness of this device, benznidazole microparticles using chitosan as carrier and NaOH, KOH, or SLS as counter ions were used. For comparison, benznidazole microparticles were prepared by the conventional dripping technique (syringe and gauge) using the same excipients. Microparticles were characterized in terms of encapsulation efficiency, particle shape, size and surface topography, crystallinity characteristics, thermal behavior, and dissolution rate. Then, the pharmacokinetic parameters were evaluated after the oral administration of the microparticles to healthy Wistar rats. The prepared formulations, by means of this device, showed good drug encapsulation efficiency (> 70%). Release studies revealed an increased dissolution of benznidazole from chitosan microparticles prepared using the novel device. It achieved more than 90% in 60 min, while those of the conventional microparticles and raw drug achieved 65% and 68%, respectively, during the same period. Almost spherical benznidazole microparticles with a smooth surface and size around 10–30 μm were observed using scanning electron microscopy. Thermal analysis and X-ray diffraction studies suggested a partial reduction of drug crystallinity. Moreover, the relative oral bioavailability of the novel benznidazole microparticles showed that the area under the curve for the microencapsulated drug was 10.3 times higher than the raw drug. Thus, these findings indicate that the designed glass prototype device is a useful alternative to formulate benznidazole polymeric microparticles with improved biopharmaceutical properties and could be useful for other therapeutic microparticulate systems.





Similar content being viewed by others
References
Tomaro-Duchesneau C, Saha S, Malhotra M, Kahouli I, Prakash S. Microencapsulation for the therapeutic delivery of drugs, live mammalian and bacterial cells, and other biopharmaceutics: current status and future directions. J Pharm. 2013;103527. https://doi.org/10.1155/2013/103527.
Kim KK, Pack DW. Microspheres for drug delivery. In: Ferrari M, Lee AP, Lee LJ, editors. BioMEMS and biomedical nanotechnology. Boston: Springer; 2006. https://doi.org/10.1007/978-0-387-25842-3_2.
Yang Y, Chen Q, Lin J, Cai Z, Liao G, Wang K, et al. Recent advance in polymer based microspheric systems for controlled protein and peptide delivery. Curr Med Chem. 2019;26(13):2285–96. https://doi.org/10.2174/0929867326666190409130207.
Xiao JX, Wang LH, Xu TC, Huang GQ. Complex coacervation of carboxymethyl konjac glucomannan and chitosan and coacervate characterization. Int J Biol Macromol. 2019;123:436–45. https://doi.org/10.1016/j.ijbiomac.2018.11.086.
Zhang Y, Fei S, Yu M, Guo Y, He H, Zhang Y, et al. Injectable sustained release PLA microparticles prepared by solvent evaporation-media milling technology. Drug Dev Ind Pharm. 2018;44(10):1591–7. https://doi.org/10.1080/03639045.2018.1483382.
Liu W, Wang S, Lu W, Cheng Z, Jiang N. Sustained release ziprasidone microparticles prepared by spray drying with Soluplus® and ethyl cellulose to eliminate food effect and enhance bioavailability. AAPS PharmSciTech. 2020;21(1):27–8. https://doi.org/10.1208/s12249-019-1592-8.
Davis M, Walker G. Recent strategies in spray drying for the enhanced bioavailability of poorly water-soluble drugs. J Control Release. 2018;269:110–27. https://doi.org/10.1016/j.jconrel.2017.11.005.
Perge L, Robitzer M, Guillemot C, Devoisselle JM, Quignard F, Legrand P. New solid lipid microparticles for controlled ibuprofen release: formulation and characterization study. Int J Pharm. 2012;422(1–2):59–67. https://doi.org/10.1016/j.ijpharm.2011.10.027.
Wu F, Jin T. Polymer-based sustained-release dosage forms for protein drugs, challenges, and recent advances. AAPS PharmSciTech. 2008;9(4):1218–29. https://doi.org/10.1208/s12249-008-9148-3.
Sosnik A, Seremeta KP. Advantages and challenges of the spray-drying technology for the production of pure drug particles and drug-loaded polymeric carriers. Adv Colloid Interf Sci. 2015;223:40–54. https://doi.org/10.1016/j.cis.2015.05.003.
Bianco S, Caron V, Tajber L, Corrigan OI, Nolan L, Hu Y, et al. Modification of the solid-state nature of sulfathiazole and sulfathiazole sodium by spray drying. AAPS PharmSciTech. 2012;13(2):647–60. https://doi.org/10.1208/s12249-012-9792-5.
Xie J, Wang CH. Encapsulation of proteins in biodegradable polymeric microparticles using electrospray in the Taylor cone-jet mode. Biotechnol Bioeng. 2007;97(5):1278–90. https://doi.org/10.1002/bit.21334.
Paulo F, Santos L. Design of experiments for microencapsulation applications: a review. Mater Sci Eng C Mater Biol Appl. 2017;77:1327–40. https://doi.org/10.1016/j.msec.2017.03.219.
Bansal A, D'Sa S, D'Souza MJ. Biofabrication of microcapsules encapsulating beta-TC-6 cells via scalable device and in-vivo evaluation in type 1 diabetic mice. Int J Pharm. 2019;572:118830. https://doi.org/10.1016/j.ijpharm.2019.118830.
Akamatsua K, Ogawa M, Katayama R, Yonemura K, Nakao S. A facile microencapsulation of phase change materials within silicone-based shells by using glass capillary devices. Colloids Surf A Physicochem Eng Asp. 2019;567:297–303. https://doi.org/10.1016/j.colsurfa.2019.01.076.
Pérez-Molina JA, Molina I. Chagas disease. Lancet. 2018;391(10115):6–12. https://doi.org/10.1016/S0140-6736(17)31612-4.
Chagas Coalition, breaking the silence: an opportunity for patients with Chagas disease. 2019. http://www.coalicionchagas.org/documents/5415804/5524305/breakingthe silence report/65091404-85cf-4796-bebb-64c120a26216.
Arrúa EC, Seremeta KP, Bedogni GR, Okulik NB, Salomon CJ. Nanocarriers for effective delivery of benznidazole and nifurtimox in the treatment of Chagas disease: a review. Acta Trop. 2019;198:105080. https://doi.org/10.1016/j.actatropica.2019.105080.
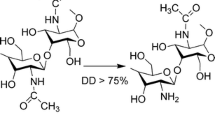
Salomon C, Goycoolea FM, Moerschbacher B. Recent trends in the development of chitosan-based drug delivery systems. AAPS PharmSciTech. 2017;18(4):933–5. https://doi.org/10.1208/s12249-017-0764-7.
Calixto GMF, de Annunzio SR, Victorelli FD, Frade ML, Ferreira PS, Chorilli M, et al. Chitosan-based drug delivery systems for optimization of photodynamic therapy: a review. AAPS PharmSciTech. 2019;20(7):253. https://doi.org/10.1208/s12249-019-1407-y.
Paul P, Kolesinska B, Sujka W. Chitosan and its derivatives - biomaterials with diverse biological activity for manifold applications. Mini-Rev Med Chem. 2019;19(9):737–50. https://doi.org/10.2174/1389557519666190112142735.
Simonazzi A, Davies C, Cid AG, Gonzo E, Parada L, Bermúdez JM. Preparation and characterization of poloxamer 407 solid dispersions as an alternative strategy to improve benznidazole bioperformance. J Pharm Sci. 2018;107(11):2829–36. https://doi.org/10.1016/j.xphs.2018.06.027.
Palmeiro-Roldán R, Fonseca-Berzal C, Gómez-Barrio A, Arán VJ, Escario JA, Torrado-Durán S, et al. Development of novel benznidazole formulations: physicochemical characterization and in vivo evaluation on parasitemia reduction in Chagas disease. Int J Pharm. 2014;472(1–2):110–7. https://doi.org/10.1016/j.ijpharm.2014.06.015.
Leonardi D, Salomon CJ. Unexpected performance of physical mixtures over solid dispersions on the dissolution behavior of benznidazole from tablets. J Pharm Sci. 2013;102(3):1016–23. https://doi.org/10.1002/jps.23448.
Sá-Barreto LC, Gustmann PC, Garcia FS, Maximiano FP, Novack KM, Cunha-Filho MS. Modulated dissolution rate from the inclusion complex of antichagasic benznidazole and cyclodextrin using hydrophilic polymer. Pharm Dev Technol. 2013;18(5):1035–41. https://doi.org/10.3109/10837450.2011.644299.
Soares-Sobrinho JL, Santos FL, Lyra MA, Alves LD, Rolim LA, Lima AA, et al. Benznidazole drug delivery by binary and multicomponent inclusion complexes using cyclodextrins and polymers. Carbohydr Polym. 2012;89(2):323–30. https://doi.org/10.1016/j.carbpol.2012.02.042.
Leonardi D, Salomón CJ, Lamas MC, Olivieri AC. Development of novel formulations for Chagas’ disease: optimization of benznidazole chitosan microparticles based on artificial neural networks. Int J Pharm. 2009;367(1–2):140–7. https://doi.org/10.1016/j.ijpharm.2008.09.036.
Tessarolo LD, de Menezes RRPPB, Mello CP, Lima DB, Magalhães EP, Bezerra EM, et al. Nanoencapsulation of benznidazole in calcium carbonate increases its selectivity to Trypanosoma cruzi. Parasitology. 2018;145(9):1191–8. https://doi.org/10.1017/S0031182018000197.
Dos Santos-Silva AM, de Caland LB, De S L Oliveira ALC, de Araújo-Júnior RF, Fernandes-Pedrosa MF, Cornélio AM, et al. Designing structural features of novel benznidazole-loaded cationic nanoparticles for inducing slow drug release and improvement of biological efficacy. Mater Sci Eng C Mater Biol Appl. 2017;78:97887. https://doi.org/10.1016/j.msec.2017.04.053.
Davanço MG, Campos ML, Rosa TA, Padilha EC, Alzate AH, Rolim LA, et al. Benznidazole extended-release tablets for improved treatment of Chagas disease: preclinical pharmacokinetic study. Antimicrob Agents Chemother. 2016;60(4):2492–8. https://doi.org/10.1128/AAC.02506-15.
Leonardi D, Lamas MC, Olivieri A. Multiresponse optimization of the properties of albendazole microparticles. J Pharm Biomed Anal. 2008;48(3):802–7. https://doi.org/10.1016/j.jpba.2008.08.006.
Elsayed A, Al-Remawi M, Qinna N, Farouk A, Al-Sou’od KA, Badwan AA. Chitosan-sodium lauryl sulfate nanoparticles as a carrier system for the in vivo delivery of oral insulin. AAPS PharmSciTech. 2011;12(3):958–64. https://doi.org/10.1208/s12249-011-9647-5.
Pandit SS, Hase DP, Bankar MM, Patil AT, Gaikwad NJ. Ketoprofen-loaded Eudragit RSPO microspheres: an influence of sodium carbonate on in vitro drug release and surface topology. J Microencapsul. 2009;26(3):195–201. https://doi.org/10.1080/02652040802224793.
Seremeta KP, Arrúa EC, Okulik NB, Salomon CJ. Development and characterization of benznidazole nano- and microparticles: a new tool for pediatric treatment of Chagas disease? Colloids Surf B: Biointerfaces. 2019;177:169–77. https://doi.org/10.1016/j.colsurfb.2019.01.039.
Berthold A, Cremer K, Kreutera J. Preparation and characterization of chitosan microspheres as drug carrier for prednisolone sodium phosphate as model for anti-inflammatory drugs. J Control Release. 1996;39(1):17–25. https://doi.org/10.1016/0168-3659(95)00129-8.
Rawat M, Saraf S, Saraf S. Influence of selected formulation variables on the preparation of enzyme-entrapped Eudragit S100 microspheres. AAPS PharmSciTech. 2007;8(4):E116. https://doi.org/10.1208/pt0804116.
Maghsoodi M. Physicomechanical properties of naproxen-loaded microparticles prepared from Eudragit l100. AAPS PharmSciTech. 2009;10(1):120–8. https://doi.org/10.1208/s12249-009-9186-5.
Aungst BJ. Absorption enhancers: applications and advances. AAPS J. 2012;14(1):10–18. https://doi.org/10.1208/s12248-011-9307-4.
Yewale C, Patil S, Kolate A, Kore G, Misra A, et al. Crit Rev Ther Drug Carrier Syst. 2015;32(5):363–87. https://doi.org/10.1615/critrevtherdrugcarriersyst.2015011865.
Pandya P, Gattani S, Jain P, Khirwal L, Surana S. Co-solvent evaporation method for enhancement of solubility and dissolution rate of poorly aqueous soluble drug simvastatin: in vitro-in vivo evaluation. AAPS PharmSciTech. 2008;9(4):1247–452. https://doi.org/10.1208/s12249-008-9176-z.
Saibi Y, Sato H, Tachiki H. Developing in vitro-in vivo correlation of risperidone immediate release tablet. AAPS PharmSciTech. 2012;13(3):890–5. https://doi.org/10.1208/s12249-012-9814-3.
Hari PR, Chandy T, Sharma CP. Chitosan/calcium alginate beads for oral delivery of insulin. J Appl Polym Sci. 1996;59:1795–801. https://doi.org/10.1002/(SICI)1097-4628(19960314)59:11<1795::AID-APP16>3.0.CO;2-T.
Takka S, Gürel A. Evaluation of chitosan/alginate beads using experimental design: formulation and in vitro characterization. AAPS PharmSciTech. 2010;11(1):460–6. https://doi.org/10.1208/s12249-010-9406-z.
Pani NR, Nath LK, Acharya S, Bhuniya B. Application of DSC, IST, and FTIR study in the compatibility testing of nateglinide with different pharmaceutical excipients. J Therm Anal Calorim. 2012;108:219–26. https://doi.org/10.1007/s10973-011-1299-x.
Verma RK, Garg S. Selection of excipients for extended release formulations of glipizide through drug-excipient compatibility testing. J Pharm Biomed Anal. 2005;38(4):633–44. https://doi.org/10.1016/j.jpba.2005.02.026.
García MC, Martinelli M, Ponce NE, Sanmarco LM, Aoki MP, Manzo RH, et al. Multi-kinetic release of benznidazole-loaded multiparticulate drug delivery systems based on polymethacrylate interpolyelectrolyte complexes. Eur J Pharm Sci. 2018;120:107–22. https://doi.org/10.1016/j.ejps.2018.04.034.
García MC, Manzo RH, Jimenez-Kairuz A. Polysaccharides-based multiparticulated interpolyelectrolyte complexes for controlled benznidazole release. Int J Pharm. 2018;545(1–2):366–77. https://doi.org/10.1016/j.ijpharm.2018.05.017.
Maximiano FP, de Paula LM, Figueiredo VP, de Andrade IM, Talvani A, Sá-Barreto LC, et al. Benznidazole microcrystal preparation by solvent change precipitation and in vivo evaluation in the treatment of Chagas disease. Eur J Pharm Biopharm. 2011;78(3):377–84. https://doi.org/10.1016/j.ejpb.2011.03.003.
Oliveira BG, Alencar Filho E, Vasconcellos MLLA. Comparisons between crystallography data and theoretical parameters and the formation of intramolecular hydrogen bonds: Benznidazole. Crystals. 2016;6(5):56. https://doi.org/10.3390/cryst6050056.
Honorato SB, Mendonça JS, Boechat N, Oliveira AC, Mendes Filho J, Ellena J, et al. Novel polymorphs of the anti-Trypanosoma cruzi drug benznidazole. Spectrochim Acta A Mol Biomol Spectrosc. 2014;118:389–94. https://doi.org/10.1016/j.saa.2013.08.096.
Maximiano FP, Novack KM, Bahia MT, Sá-Barreto LC, Cunha-Filho MS. Polymorphic screen and drug–excipient compatibility studies of the antichagasic benznidazole. J Therm Anal Calorim. 2011;106:819–24. https://doi.org/10.1007/s10973-011-1371-6.
Vieira de Castro AC, Olsson IA. Does the goal justify the methods? Harm and benefit in neuroscience research using animals. Curr Top Behav Neurosci. 2015;19:47–78. https://doi.org/10.1007/7854-2014-319.
Brown AP, Dinger N, Levine BS. Stress produced by gavage administration in the rat. Contemp Top Lab Anim Sci. 2000;39(1):17–21.
Turner PV, Brabb T, Pekow C, Vasbinder MA. Administration of substances to laboratory animals: routes of administration and factors to consider. J Am Assoc Lab Anim Sci. 2011;50(5):600–13.
Perin L, Moreira da Silva R, Fonseca KD, Cardoso JM, Mathias FA, Reis LE, et al. Pharmacokinetics and tissue distribution of benznidazole after oral administration in mice. Antimicrob Agents Chemother. 2017;61(4):e02410–6. https://doi.org/10.1128/AAC.02410-16.
Cano-Cebrián MJ, Zornoza T, Granero L, Polache A. Intestinal absorption enhancement via the paracellular route by fatty acids, chitosans and others: a target for drug delivery. Curr Drug Deliv. 2005;2(1):9–22. https://doi.org/10.2174/1567201052772834.
Funding
The National University of Rosario (UNR) and the National Council Research (CONICET) provided financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was authorized by the Ethical Committee on Animal Experimentation of the Faculty of Biochemical and Pharmaceutical Sciences, National University of Rosario (Argentina). Also, experiments with animals were performed according to the ethical standards formulated in the Declaration of Helsinki, and appropriate measures were taken to protect animals from pain or discomfort.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barrera, M.G., Tejada, G., Leonardi, D. et al. A Novel Prototype Device for Microencapsulation of Benznidazole: In Vitro/In Vivo Studies. AAPS PharmSciTech 21, 112 (2020). https://doi.org/10.1208/s12249-020-01659-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-020-01659-3