Abstract
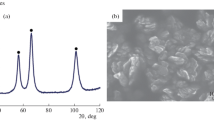
In this paper, the synthesis of nickel nanopowders by the chemicometallurgical method under dynamic conditions, when the composition of the gas medium is changed in the process of synthesis (argon is changed for hydrogen or otherwise) with the retention of the overall time of synthesis, is studied for the first time. It is demonstrated that the synthesis under dynamic conditions is an efficient method for controlling the morphology and sizes of synthesized nanopowders.



Similar content being viewed by others
REFERENCES
T. Shishido and K. Takehira, “Preparation of highly dispersed ni catalysts for H2 production for polymer electrolyte fuel cells,” J. Jpn. Pet. Inst. 58, 353–364 (2015).
S. Qiu, X. Zhang, Q. Liu, T. Wang, Q. Zhang, and L. Ma, “A simple method to prepare highly active and dispersed Ni/MCM-41 catalysts by co-impregnation,” Catal. Commun. 42 (5), 73–78 (2013).
X. Ning, Y. Lu, H. Fu, H. Wan, Z. Xu, and S. Zheng, “Template-mediated Ni(II) dispersion in mesoporous SiO2 for preparation of highly dispersed Ni catalysts: influence of template type,” ACS Appl. Mater. Interfaces 9, 19335–19344 (2017).
K. H. Park, S. H. Im, and O. O. Park, “The size control of silver nanocrystals with different polyols and its application to low-reflection coating materials,” Nanotechnology 22, 045602 (2011).
Y. Zhang, J. Wen, J. Wang, D. Pan, M. Shen, and Yu. Lu, “Synthesis of monodisperse CexZR1 – xO2 nanocrystals and the size-dependent enhancement of their properties,” Nano Res. 4, 494–504 (2011).
P. Kundu, C. Nethravathi, P. A. Deshpande, M. Rajamathi, G. Madras, and N. Ravishankar, “Ultrafast microwave-assisted route to surfactant-free ultrafine Pt nanoparticles on graphene: synergistic co-reduction mechanism and high catalytic activity,” Chem. Mater. 23, 2772–2780 (2011).
J. Rubio-Garcia, Y. Coppel, P. Lecante, C. Mingotaud, B. Chaudret, G. Fabienne, and M. L. Kahn, “One-step synthesis of metallic and metal oxide nanoparticles using amino-PEG oligomers as multi-purpose ligands: size and shape control, and quasi-universal solvent dispersibility,” Chem. Commun. 47, 988–990 (2011).
M. A. Malik, M. Yu. Wani, and M. A. Hashim, “Microemulsion method: a novel route to synthesize organic and inorganic nanomaterials: 1st nano update,” Arab. J. Chem. 5, 397–417 (2012).
A. K. Ganguli, A. Ganguly, and S. Vaidya, “Microemulsion-based synthesis of nanocrystalline materials,” Chem. Soc. Rev. 39, 474–485 (2010).
V. F. Puntes, K. M. Krishnan, and A. P. Alivisatos, “Colloidal nanocrystal shape and size control: the case of cobalt,” Science (Washington, D.C., U. S.) 291, 2115–2117 (2001).
E. V. Shevchenko, D. V. Talapin, H. Schnablegger, A. Kornowski, O. Festin, P. Svedlindh, M. Haasse, and H. Weller, “Study of nucleation and growth in the organometallic synthesis of magnetic alloy nanocrystals: the role of nucleation rate in size control of CoPt3 nanocrystals,” J. Am. Chem. Soc. 125, 9090–9101 (2003).
Y. Hou, H. Kondoh, T. Ohta, and S. Gao, “Size-controlled synthesis of nickel nanoparticles,” Appl. Surf. Sci. 241, 218–222 (2005).
J. G. Dean, “Thermal decomposition of nickel formate,” Ind. Eng. Chem. Soc. 44, 985–993 (1952).
M. I. Alymov, A. B. Ankudinov, I. V. Tregubova, and A. A. Zablotskii, “Tungsten based nanopowder synthesis,” Fiz. Khim. Obrab. Mater., No. 6, 81–82 (2005).
ACKNOWLEDGMENTS
This study was supported by the Presidium of the Russian Academy of Sciences, program of fundamental studies no. 32 “Nanostructures: Physics, Chemistry, Biology, and Principles of Technologies.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Glushachenkova
Rights and permissions
About this article
Cite this article
Alymov, M.I., Rubtsov, N.M., Seplyarskii, B.S. et al. Synthesis of Nickel Nanopowders under Dynamic Conditions. Nanotechnol Russia 13, 557–560 (2018). https://doi.org/10.1134/S1995078018060022
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995078018060022