Abstract
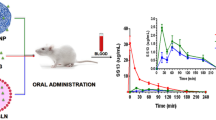
Recent studies in nanotechnology resulted in the development of novel formulations with improved bioavailability. This is especially important for oral administered drugs as the most convenient formulations for administration to patients. The review considers processes occurring in the gastro-intestinal (GI) tract during oral administration of drugs. The increase of bioavailability of the drug may be achieved through designing novel formulations according to the specific drug properties. These include capsules that release pharmaceutical agents at various parts of the GI tract, floating systems that prolong the presence of the drug in stomach, maximally dispersed forms containing surface-active soluble polymers or micelles that carry poor-soluble drugs inside their non-polar core, agents that facilitate tight junction opening, such as caprate and chitosan, and lipid-based formulations. The own data show the stimulating influence of phospholipid nanoparticles on peroral absorption of the drug, indomethacin, in rats and on passage of transport marker and drugs through Caco-2 cell monolayer in vitro. The review summarizes current understanding of factors that influence the bioavailability of the oral drug formulations, currently used models for pharmacokinetic studies, and various approaches to developing novel pharmaceutical formulations that increase the bioavailability of the drugs.
Similar content being viewed by others
References
Martinez, M.N., and Amidon, G.L., J. Clin. Pharmacol., 2002, vol. 42, pp. 620–643.
Storozhakov, G.I., Chukaeva, I.I., and Aleksandrov, A.A., Poliklinicheskaya terapiya (Polyclinic Therapy), Moscow: GEOTAR-Media.
Golovenko, N.Ya, and Borisyuk, I.Yu., Biomed. Khim., 2008, vol. 54, pp. 392–407.
Arora, S., Ali, J., Ahuja, A., Khar, R.K., and Baboota, S., AAPS Pharm. Sci. Tech., 2005, vol. 6, pp. E372–E390.
Basarab, G.S., Hill, P.J., Rastagar, A., and Webborn, P.J., Bioorg. Med. Chem. Lett., 2008, vol. 8, pp. 4716–4722.
Cano-Cebrián, M.J., Zornoza, T., Granero, L., and Polache, A., (2005) Curr. Drug Deliv., 2005, vol. 2, pp. 9–22.
des Rieux, A., Fievez, V., Garinot, M., Schneider, Y.J., and Préat, V., J. Control Release, 2006, vol. 116, pp. 1–27.
Takano, M.., Yumoto, R.., and Murakami, T., (2006) Pharmacol. Ther., 2006, vol. 109, pp. 137–161.
Zaïr, Z.M., Eloranta, J.J., Stieger, B., and Kullak-Ublick, G.A., Pharmacogenomics, 2008, vol. 9, pp. 597–624.
Kato, M., Drug Metab. Pharmacokinet., 2008, vol. 23, pp. 87–94.
van de Kerkhof, E.G., de Graaf, I.A., and Groothuis, G.M., Curr. Drug Metab., 2007, vol. 8, pp. 658–675.
Porter, C.J., and Charman, W.N., Adv. Drug Deliv. Rev., 2001, vol. 50, pp. 1–2.
Patel, G.N., Patel, G.C., Patel, R.B., Patel, S.S., Patel, J.K., Bharadia, P.D., and Patel, M.M., Drug Deliv. Technol., 2006, vol. 6, pp. 62–71.
Krishnaiah, Y.S.R., Veer Raju, P., Dinesh, K.B., Bhaskar, P., and Satyanarayana, V., J. Control Rel., 2001, vol. 77, pp. 87–95.
Chen, K., Yu, W.P., Song, L., and Zhu, Y.M., World J. Gastroenterol., 2005, vol. 11, pp. 641–644.
Ruh, J., Schmidt, E., and Vogel, F., Dig. Dis. Sci., 2003, vol. 48, pp. 882–889.
Chen, M., Yang, Y., Braunstein, E., Georgeson, K.E., and Harmon, C.M., Am. J. Physiol. Endocrinol. Metab., 2001, vol. 281, pp. E916–E923.
Katneni, K., Charman, S.A., and Porter, C.J., J. Pharm. Pharmacol., 2008, vol. 60, pp. 1311–1319.
Marcus, S.N., Schteingart, C.D., Marquez, M.L., Hofmann, A.F., Xia, Y., Steinbach, J.H., Ton-Nu, H.T., Lillienau, J., Angellotti, M.A., and Schmassmann, A., Gastroenterology, 1991, vol. 100, pp. 212–221.
Li, J.P., Chang, T.M., Wagner, D., Chey, W.Y., Am. J. Physiol. Gastrointest. Liver Physiol., 2001, vol. 281, pp. G526–G532.
Poirier, H., Mathieu, Y., Besnard, P., and Bernard, A., Comp. Biochem. Physiol. A. Physiol., 1997, vol. 116, pp. 253–260.
Porter, C.J., Trevaskis, N.L., and Charman, W.N., Nat. Rev. Drug Discov., 2007, vol. 6, pp. 231–248.
Dressman, J.B., Thelen, K., and Jantratid, E., Clin. Pharmacokinet., 2008, vol. 47, pp. 655–667.
Neuhoff, S., Ungell, A.L., Zamora, I., and Artursson, P., Eur. J. Pharm. Sci., 2005, vol. 25, pp. 211–220.
Custodio, J.M., Wu, C.-Y., and Benet, L.Z., Adv. Drug Deliv. Rev., 2008, vol. 60, pp. 717–733.
Pang, K.S., Drug Metab. Dispos., 2003, vol. 31, pp.1507–1519.
Cao, X., Gibbs, S.T., Fang, L., Miller, H.A., Landowski, C.P., Shin, H.C., Lennernas, H., Zhong, Y., Amidon, G.L, Yu, L.X, and Sun, D., Pharm. Res., 2006, vol. 23, pp. 1675–1686.
Kim, I.W., Booth-Genthe, C., and Ambudkar, S.V., Mini Rev. Med. Chem., 2008, vol. 8, pp. 193–200.
Agellon, L.B., Toth, M.J., and Thomson, A.B., Mol. Cell Biochem., 2002, vol. 239, pp. 79–82.
Besnard, P, Niot, I, Poirier, H, Clément, L., and Bernard, A., Mol. Cell Biochem., 2002, vol. 239, pp. 139–147.
Meunier-Durmort, C., Poirier, H., Niot, I., Forest, C., and Besnard, P., Biochem. J., 1996, vol. 319, pp. 483–487.
Trevaskis, N.L., Lo, C.M., Ma, L.Y., Tso, P., Irving, H.R., Porter, C.J., and Charman, W.N., Pharm. Res., 2006, vol. 23, pp. 1786–1796.
Wolfrum, C., Cell Mol. Life Sci., 2007, vol. 64, pp. 2465–2476.
Tso, P., Nauli, A., and Lo, C.M., Biochem. Soc. Trans., 2004, vol. 32, pp. 75–78.
Kim, R.B., Molecular Pharmaceutics, 2006, vol. 3, pp. 26–32.
Velkov, T., Horne, J., Laguerre, A., Jones, E., Scanlon, M.J., and Porter, C.J., Chem. Biol., 2007, vol. 14, pp. 453–465.
Takara, K., Ohnishi, N., Horibe, S., and Yokoyama, T., Drug Metab. Dispos., 2003, vol. 31, pp. 1235–1239.
Shayeganpour, A., El-Kadi, A.O., and Brocks, D.R., Drug Metab. Dispos., 2006, vol. 34, pp. 43–50.
Lin, J.H., Chiba, M., Balani, S.K., Chen, I.W., Kwei, G.Y., Vastag, K.J., and Nishime, J.A., Drug Metab. Dispos., 1996, vol. 24, pp. 1111–1120.
Behrens, D., Fricker, R., Bodoky, A., Drewe, J., Harder, F., and Heberer, M., J. Pharm. Sci., 1996, vol. 85, pp. 666–668.
Wasan, K.M., Ramaswamy, M., McIntosh, M.P., Porter, C.J., and Charman, W.N., J. Pharm. Sci., 1999, vol. 88, pp. 185–190.
Kraehenbuhl, J.-P., and Neutra, M.P., Annu. Res.Cell Dev., 2000, vol. 16, pp. 301–332.
Florence, A.T., Pharm. Res., 1997, vol. 14, pp. 259–266.
Jani, P., Halbert, G.W., Langridge, J., and Florence, A.T., J. Pharm. Pharmacol., 1990, vol. 42, pp. 821–826.
Salama, N.N., Eddington, N.D., and Fasano, A., Adv. Drug Deliv. Rev., 2006, vol. 58, pp. 15–28.
Lee, D.B., Jamgotchian, N., Allen, S.G., Abeles, M.B., and Ward, H.J., Am. J. Physiol. Renal Physiol., 2008, vol. 295, pp. F1601–F612.
Kondoh, M. and Yagi, K., Curr. Med. Chem., 2007, vol. 14, pp. 2482–2488.
Hayashi, M. and Tomita, M., Drug Metab. Pharmacokinet., 2007, vol. 22, pp. 67–77.
Hussain, N., Jaitley, V., and Florence A.T., Adv. Drug Deliv. Rev., 2001, vol. 50, pp. 107–142.
Florence, A.T. and Hussain, N., Adv. Drug Deliv. Rev, 2001, vol. 50, pp. S69–S89.
McClean, S., Prosser, E., Meehan, E., O’Malley, D., Clarke, N., Ramtoola, Z., and Brayden, D., Eur. J. Pharm. Sci., 1998, vol. 6, pp. 153–163.
Florence, A.T., J. Drug Target, 2004, vol. 12, pp. 65–70.
Simovic, S., Heard, P., Hui, H., Song, Y., Peddie, F., Davey, A.K., Lewis, A., Rades, T., and Prestidge, C.A., Mol. Pharm., 2009, vol. 6, pp. 861–872.
Miyazaki, S., Yamaguchi, H., Yokouchi, C., Takada, M., and Hou, W.M., Chem. Pharm. Bull. (Tokyo), 1988, vol. 36, pp. 4033–4038.
Upadhye, A.A., Ambike, A.A., Mahadik, K.R., and Paradkar, A., Drug Dev. Ind. Pharm., 2008, vol. 34, pp. 1117–1124.
Patel, A., Modasiya, M., Shah, D., and Patel, V., AAPS Pharm. Sci. Tech., 2009, vol. 10, pp. 310–315.
Sasaki, K., Yonebayashi, S., Yoshida, M., Shimizu, K., Aotsuka, T., Takayama, K., Int. J. Pharm., 2003, vol. 265, pp. 95–102.
Artursson, P., and Karlsson, J., Biochem. Biophys. Res. Commun., 1991, vol. 175, pp. 880–885.
Lo, Y.L. and Huang, J.D., Biochem. Pharmacol., 2000, vol. 59, pp. 665–672.
Schmidt, P.G., Campbell, K.M., Hinds, K.D., and Cook, G.P., Expert Opin. Biol. Ther., 2007, vol. 7, pp. 1427–1436.
Carrier, R.L., Miller, L.A., and Ahmed, I., J. Control Release, 2007, vol. 123, pp. 78–99.
Croy, S.R. and Kwon, G.S., Curr. Pharm. Des., 2006, vol. 12, pp. 4669–4684.
Porter, C.J., Kaukonen, A.M., Boyd, B.J., Edwards, G.A., and Charman, W.N., Pharm. Res., 2004, vol. 21, pp. 1405–1412.
McConville, J.T., Ross, A.C., Florence, A.J., and Stevens, H.N., Drug Dev. Ind. Pharm., 2005, vol. 31, pp. 79–89.
Wikling, I.R., and Prior, D.V., Crit. Rev. Ther. Drug Carrier Syst., 2003, vol. 20, pp. 405–431.
Muller, R.H., and Keck, C.M., J. Biotechnol., 2004, vol. 113, pp. 151–170.
Vyas, S.P., and Gupta, P.N., Expert Rev. Vaccines, 2007, vol. 6, pp. 401–418.
Hauss, D.J., Adv. Drug Deliv. Rev., 2007, vol. 59, pp. 667–676.
Pouton, C.W., and Porter, C.J., Adv. Drug Deliv. Rev., 2008, vol. 60, pp. 625–637.
Pouton, C.W., Eur. J. Pharm. Sci., 2000, vol. 11, Suppl 2, pp. S93–S98.
Gursoy, R.N., and Benita, S., Biomed. Pharmacother., 2004, vol. 58, pp. 173–182.
Kapitza, S.B., Michel, B.R, van Hoogevest, P., Leigh, M.L., and Imanidis, G., Eur. J. Pharm. Biopharm., 2007, vol. 66, pp. 146–158.
Porter, C.J., Charman, S.A., and Charman, W.N., J. Pharm. Sci., 1996, vol. 85, pp. 351–356.
Lacombe, O., Woodley, J., Solleux, C., Delbos, J.-M, Boursier-Neyret, C., and Houin, G., Eur. J. Pharmaceutical Sciences, 2004, vol. 23, pp. 385–391.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.M. Ipatova, T.I. Torkhovskaya, N.V. Medvedeva, V.N. Prozorovsky, N.D. Ivanova, A.V. Shironin, V.S. Baranova, A.I. Archakov, 2010, published in Biomeditsinskaya Khimiya.
Rights and permissions
About this article
Cite this article
Ipatova, O.M., Torkhovskaya, T.I., Medvedeva, N.V. et al. Bioavailability of oral drugs and the methods for its improvement. Biochem. Moscow Suppl. Ser. B 4, 82–94 (2010). https://doi.org/10.1134/S1990750810010117
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990750810010117