Abstract
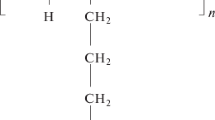
The formation and properties of adsorption layers of poly(dimethyldiallylammonium chloride) with different molecular masses on the surface of fused quartz are studied by the capillary electrokinetics method. It is shown that the value of ζ potential depends on the flow rate of liquid determined by the pressure drop. Such behavior can be explained by the deformation of the adsorption layer. At low rates of liquid flow, constant values of adsorption and time needed to achieve these values decrease for the samples of lower molecular masses, which is probably related to the more closely packed structure and, hence, to the lower deformability of the adsorption layers, as well as to the shortest times during which conformational rearrangements proceed in the layer. The time of conformational changes in the adsorption layer significantly exceeds the time of adsorption. The adsorption of cationic polyelectrolyte is irreversible. It is found that the compaction of adsorption layers increases with time; the rate of compaction of layers of a low-molecular-mass polyelectrolyte is higher and the layers of a high-molecular-mass polyelectrolyte retain the residual deformability even for six days. The measurements of the filtration of polyelectrolyte solutions through thin quartz capillaries allow the thickness of adsorption layers and their deformation under pressure to be estimated.
Similar content being viewed by others
References
Bertran, P., Jonas, A., Laschewsky, A., and Legras, R., Macromol. Rapid Commun., 2000, vol. 21, p. 319.
Caruso, F., Furlong, D.N., Ariga, K., et al., Langmuir, 1998, vol. 14, p. 4559.
Onoda, M., Ariga, K., and Kunitake, T., J. Biosci. Bioeng., 1999, vol. 87, p. 69.
Caruso, F. and Schuler, C., Langmuir, 2000, vol. 16, p. 9595.
Wu, A., Yoo, D., Lee, J.K., and Rubner, M.F., J. Am. Chem. Soc., 1999, vol. 121, p. 4883.
Sukhorukov, G.B., Donath, E., Lichtenfeld, H., et al., Colloids Surf., A, 1998, vol. 137, p. 253.
Caruso, F., Tran, D., Mohwald, H., and Renneberg, R., Langmuir, 2000, vol. 16, p. 1485.
Antipov, A.A., Sukhorukov, G.B., Lepotatti, S., et al., Colloid Surf., A, 2002, vol. 198, p. 535.
Levaesalmi, J. and McCarthy, T.J., Macromolecules, 1997, vol. 30, p. 1752.
McAloney, R.A., Sinyor, M., Dudnik, V., and Goh, M.C., Langmuir, 2001, vol. 17, p. 6655.
Claesson, P.M., Dedinate, A., and Rojas, O., Adv. Colloid Interface Sci., 2003, vol. 104, p. 53.
Steiz, R., Leiner, V., Siebrecht, R., and Klitzing, R., J. Colloid Interface Sci., 2000, vol. 163, p. 63.
Buscher, K., Graf, K., Ahrens, H., and Helm, C., Langmuir, 2002, vol. 18, p. 3585.
Baba, A., Kaneko, F., and Advincula, R., J. Physicochem. Eng. Aspects, 2000, vol. 173, p. 39.
Kong, C.Y. and Muthukumar, M., J. Chem. Phys., 1998, vol. 109, p. 1522.
Messina, R., Holm, C., and Kremer, K., Langmuir, 2003, vol. 19, p. 4473.
Messina, R., J. Chem. Phys., 2003, vol. 119, p. 8133.
Shafir, A. and Andellman, D., Phys. Rev. E: Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top., 2004, vol. 70, p. 061804.
Joanny, J.F., Eur. Phys. J., B, 1999, vol. 86, p. 117.
Muthukumar, M., J. Chem. Phys., 1987, vol. 86, p. 7230.
Sergeeva, I.P., Ermakova, T.B., Churaev, N.V., et al., Kolloidn. Zh., 2002, vol. 64, p. 531.
Sergeeva, I.P., Ermakova, T.B., Sobolev, V.D., and Churaev, N.V., Kolloidn. Zh., 2003, vol. 65, p. 662.
Sergeeva, I.P., Ermakova, T.B., Anuchkina, A.D., et al., Kolloidn. Zh., 2005, vol. 67, p. 573.
Ermakova, T.B., Sergeeva, I.P., Anuchkina, A.D., et al., Prog. Colloid Polym. Sci., 2006, vol. 132, p. 95.
Schlenoff, J.B. and Dubas, S.T., Macromolecules, 2001, vol. 34, p. 592.
Churaev, N.V., Sobolev, V.D., and Somov, A.N., J. Colloid Interface Sci., 1984, vol. 97, p. 574.
Dubas, S.T. and Schlenoff, J.B., Langmuir, 2001, vol. 17, p. 7725.
Dubas, S.T. and Schlenoff, J.B., Macromolecules, 1999, vol. 32, p. 8153.
Kramer, G., Estel, K., Schmitt, F.-J., and Jacobasch, H.-J., J. Colloid Interface Sci., 1998, vol. 208, p. 302.
Ladam, G., Schaad, P., Voegel, J.C., et al., Langmuir, 2000, vol. 16, p. 1249.
Cohen, J., Mashe, B., and Priel, Z., Acta Polym., 1998, vol. 49, p. 557.
Lowack, K. and Helm, C.A., Macromolecules, 1998, vol. 31, p. 823.
Khesselink, F., in Adsorption from Solution on Solid/Liquid Interface, Parfitt, G.D. and Rochester, C.H., Eds., London: Academic, 1983.
Author information
Authors and Affiliations
Additional information
Original Russian Text © I.P. Sergeeva, T.B. Ermakova, D.A. Semenov, V.D. Sobolev, O.A. Kiseleva, N.V. Churaev, 2008, published in Kolloidnyi Zhurnal, 2008, Vol. 70, No. 5, pp. 656–662.
Rights and permissions
About this article
Cite this article
Sergeeva, I.P., Ermakova, T.B., Semenov, D.A. et al. Layer-to-layer adsorption of oppositely charged polyelectrolytes: The effect of molecular mass: 1. Formation of the first layer. Colloid J 70, 609–615 (2008). https://doi.org/10.1134/S1061933X08050104
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X08050104