Abstract
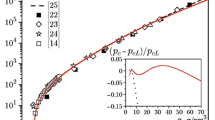
The correlation between the volumetric thermal expansion coefficient β(T) and the heat capacity C(T) of aluminum is considered in detail. It is shown that a clear correlation is observed in a significantly wider temperature range, up to the melting temperature of the metal, along with the low-temperature range where it is linear. The significant deviation of dependence β(C) from the low-temperature linear behavior is observed up to the point where the heat capacity achieves the classical Dulong–Petit limit of 3R (R is the universal gas constant).
Similar content being viewed by others
References
P. D. Pathak and N. P. Shah, “Debye temperature of silver and aluminum at high temperatures—some new correlations,” Phys. Stat. Sol. A 55 (2), K159–K162 (1979).
V. Yu. Bodryakov and V. M. Zamyatin, “Peculiarities of thermodynamic functions of solid-state aluminum,” Russian Metallurgy (Metally), No. 4, 123–128 (1999).
V. Yu. Bodryakov and A. A. Powzner, Self-Consistent Thermodynamic Model of Crystal Lattice of a Solid: Part 2. Nonferromagnetic Metals (UGTU–UPI, Yekaterinburg, 2003).
W. F. Glaque and P. F. Meads, “The heat capacities and entropies of aluminum and copper from 15 to 300 K,” J. Amer. Chem. Soc. 63 (7), 1897–1901 (1941).
T. E. Pochapsky, “Heat capacity and resistance measurements of aluminum and lead wires,” Acta Metallurgica 1 (11), 747–751 (1953).
K. K. Kelley, Contributions to the Data on Theoretical Metallurgy XIII: High Temperature Heat Content, Heat Capacity and Entropy Data for the Elements and Inorganic Compounds (US Government Printing Office, Washington, 1960).
R. J. Corruccini and J. J. Guiewek, “Specific heats of technical solids at low temperatures: a compilation from the literature,” in National Bureau of Standard Monograph NBS-21 (US Government Printing Office, Washington, 1960).
A. J. Leadbetter, “Anharmonic effects in the thermodynamic properties of solids. 1. An adiabatic calorimeter for the temperature range 25–500°C: the heat capacities of Al2O3, Al, and Pb,” J. Phys. C.: Solid State Phys. 1 (6), 1481–1488 (1968).
C. R. Brooks and R. E. Bingham, “The specific heat of aluminum from 330 to 890 K and contributions from the formation of vacancies and anharmonic effects,” J. Phys. Chem. Solids 29 (9), 1553–1560 (1968).
E. N. Buyco and F. E. Davis, “Specific heat of aluminum from zero to its melting temperature and beyond. Equation for representation of the specific heats of solids,” J. Chem. Eng. Data 15 (4), 518–523.
L. A. Novitskii and I. G. Kozhevnikov, Thermal-Physical Properties of Materials at Low Temperatures: A Handbook (Mashinostroenie, Moscow, 1975).
R. A. Robie, B. S. Hemingway, and J. R. Fisher, “Thermodynamic properties of minerals and related substances at 298.15 K (25°C) and one-atmosphere (1.013 Bars) pressure and at higher temperatures,” in Geological Survey Bulletin (US Government Printing Office, Washington, 1979), No. 1452.
D. B. Downie and J. F. Martin, “An adiabatic calorimeter for heat-capacity measurements between 6 and 300 K,” J. Chem. Thermodyn. 12 (8), 779–786 (1980).
L. V. Gurvich, I. V. Veits, V. A. Medvedev, et al., Thermodynamic Properties of Individual Materials: A Handbook (Nauka, Moscow, 1981), Vol. III.
M. E. Drits, P. B. Budberg, G. S. Burkhanov, A. M. Drits, and V. M. Panovko, Properties of Elements, Ed. by M. E. Drits (Metallurgiya, Moscow, 1985).
D. A. Ditmars, C. A. Plint, and R. C. Shukla, “Aluminum. 1. Measurement of the relative enthalpy from 273 to 929 K and derivation of thermodynamic functions for Al(s) from 0 K to its melting point,” Intern. J. Thermophys. 6 (5), 499–515 (1985).
P. D. Desai, “Thermodynamic properties of aluminum,” Intern. J. Thermophys. 8 (5), 621–628 (1987).
Y. Takahashi, T. Azumi, and Y. Sekine, “Heat capacity of aluminum from 80 to 880 K,” Thermochimica Acta 139 (1), 133–137 (1989).
Physical Quantities: A Handbook, Ed. by I. S. rigor’ev and E. Z. Meilikhov (Energoatomizdat, Moscow, 1991).
M. W. Chase, Jr., “NIST–JANAF Thermochemical Tables,” in J. Phys. Chem. Ref. Data, (1998), Monograph 9, pp. 1–1951.
A. T. Dinsdale, SGTE Data for Pure Elements (NPL (National Physical Laboratory, Teddington, 2007).
Tables of Physical and Chemical Constants (National Physical Laboratory). http://www.kayelaby.npl.co.uk/general_physica/2_3/2_3_6.html
A. J. C. Wilson, “The thermal expansion of aluminum from 0 to 650°C,” Proc. Phys. Soc. 53 (3), 235–244 (1941).
F. C. Nix and D. MacNair, “The thermal expansion of pure metals: copper, gold, aluminum, nickel, and iron,” Phys. Rev. 60 (10), 597–605 (1941).
E. Huzan, C. P. Abbiss, and G. O. Jones, “Thermal expansion of aluminum at low temperatures,” Phil. Mag. 6 (62), 277–285 (1961).
K. Andres, “The measurement of thermal expansion of metals at low temperatures,” Cryogenics 2 (2), 93–97 (1961).
R. J. Corruccini and J. J. Gnievek, Thermal Expansion of Technical Solids at Low Temperatures: A Compilation from Literature (US Government Printing Office, Washington, 1961).
R. M. Nicklow and R. A. Young, “Thermal expansion of silver chloride,” Phys. Rev. 129 (5), 1936–1943 (1963).
R. H. Carr and C. A. Swenson, “Application of a variable transformer to the study of low temperature thermal expansion,” Cryogenics 4 (2), 76–82 (1964).
D. B. Fraser and A. C. Hollis Hallett, “The coefficient of thermal expansion of various cubic metals below 100 K,” Can. J. Phys. 43 (2), 193–219 (1965).
A. J. Leadbetter, “Anharmonic effects in the thermodynamic properties of solids II. Analysis of data for lead and aluminum,” J. Phys. C: Solid State Phys. 1 (6), 1489–1504 (1968).
P. D. Pathak and N. D. Vasavada, “Thermal expansion and the law of corresponding states,” J. Phys. C: Solid State Phys. 3 (2), L44–L47 (1970).
M. E. Straumanis and C. L. Woodward, “Lattice parameters and thermal expansion coefficients of Al, Ag, and Mo at low temperatures. Comparison with dilatometric data,” Acta Cryst. A 27 (6), 549–551 (1971).
F. G. Awad and D. Gugan, “The thermal expansion of copper, aluminum, potassium chloride, and potassium iodide between 10 and 80 K,” Cryogenics 11 (5), 414–415 (1971).
J. G. Collins, G. K. White, and S. A. Swenson, “The thermal expansion of aluminum below 35 K,” J. Low Temp. Phys. 10 (1–2), 69–77 (1973).
S. I. Novikov, Thermal Expansion of Solids (Nauka, Moscow, 1974).
Y. S. Touloukian, R. K. Kirby, R. E. Taylor, and P. D. Desai, Thermal Expansion of Metallic Elements and Alloys in Thermodynamic Properties of Matter (IFI/Plenum, New York, 1975), Vol.12.
F. R. Kroeger and C. A. Swenson, “Absolute linear thermal-expansion measurements on copper and aluminum from 5 to 320 K,” J. Appl. Phys. 48 (3), 853–864 (1977).
B. Hallstedt, “Molar volumes of Al, Li, Mg, and Si,” Calphad 31 (2), 292–302 (2007).
Tables of Physical and Chemical Constants (National Physical Laboratory). http://www.kayelaby.npl.co.uk/general_physica/2_3/2_3_5.html
G. Korn and T. Korn Mathematical Handbook for Sciences and Engineers. Definitions, Theorems, and Formulas for References and Review (McGraw-Hill, Inc. New York, 1968).
V. Yu. Bodryakov, “On correlation between temperature dependences of the thermal expansion and the heat capacity up to melting point of a refractory metal: molybdenum,” Teplofiz, Vys. Temp. 52 (6), 863–869 (2014).
V. Yu. Bodryakov, “On correlation between temperature dependences of the heat capacity and thermal expansivity of cubic Pt-metals (following to the John Arblaster’s evalutions),” Open Sci. J. Mod. Phys. 2 (1), 10–13 (2015).
V. Yu. Bodryakov and Yu. N. Babintsev, “Correlation analysis of the heat capacity and thermal expansion of solid mercury,” Phys. Sol. States 57 (6), 1264–1268 (2015).
G. N. Katt and G. A. Alers, “Low-temperature elastic moduli of aluminum,” J. Appl. Phys. 35 (2), 327–330 (1964).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.Yu. Bodryakov, A.A. Bykov, 2016, published in Metally, 2016, No. 3, pp. 61–66.
Rights and permissions
About this article
Cite this article
Bodryakov, V.Y., Bykov, A.A. Correlation dependence of the volumetric thermal expansion coefficient of metallic aluminum on its heat capacity. Russ. Metall. 2016, 450–454 (2016). https://doi.org/10.1134/S0036029516050074
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036029516050074