Abstract—
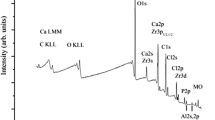
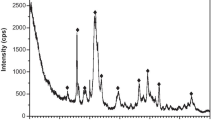
We have studied Mg2+ adsorption on hydroxyapatite (HAp) nanocrystals and constructed its isotherm at equilibrium cation concentrations in the range 0–0.35 mol/L. For a number of samples, corresponding to characteristic points in the adsorption isotherm, the composition and the oxidation state of the elements present on the surface of the sorbent have been determined by X-ray photoelectron spectroscopy (XPS). The surface magnesium concentration determined by XPS, which detects only ions in a surface layer a few nanometers in thickness, has been found to be considerably lower than the total amount of cations in the solid phase, evaluated from the experimentally determined adsorption isotherm. We assume that, during the sorption process, some of the magnesium ions substitute for calcium ions in the bulk of the HAp and some adsorb in the form of MgCl2. The results obtained in this study constitute a fundamental basis for practical solutions in designing integrated medications based on HAp with magnesium.






Similar content being viewed by others
REFERENCES
Melikhov, I.V., Teterin, Yu.A., Rudin, V.N., Teterin, A.Yu., Maslakov, K.I., and Severin, A.V., X-ray study of nano-dispersed hydroxyapatite, Russ. J. Phys. Chem. A, 2009, vol. 83, no. 1, pp. 91–97.
Webster, T.J., Massa-Schlueter, E.A., Smith, J.L., and Slamovich, E.B., Osteoblast response to hydroxyapatite doped with divalent and trivalent cations, Biomaterials, 2004, vol. 25, pp. 2111–2121.
Narasaraju, T.S.B. and Phebe, D.E., Review: some physico-chemical aspects of hydroxylapatite, J. Mater. Sci., 1996, vol. 3, pp. 11–21.
Zuev, V.P., Pankratov, A.S., and Alekseeva, A.N., Use of ultrafine hydroxyapatite particles in integrated treatment of mandibular fractures, Stomatologiya, 1994, no. 4, pp. 31–33.
Janning, C., Willbold, E., Vogt, C., Nellesen, J., Meyer-Lindenberg, A., Windhagen, H., Thorey, F., and Witte, F., Magnesium hydroxide temporarily enhancing osteoblast activity and decreasing the osteoclast number in peri-implant bone remodeling, Acta Biomater., 2010, vol. 6, pp. 1861–1868.
Melikhov, I.V., Komarov, V.F., Severin, A.V., Bozhevol’nov, V.E., and Rudin, V.N., Two-dimensional crystalline hydroxyapatite, Dokl. Phys. Chem., 2000, vol. 373, no. 3, pp. 125–128.
Suvorova, E.I. and Buffat, P.A., Electron diffraction from micro- and nanoparticles of hydroxyapatite, J. Microsc., 1999, vol. 196, no. 1, pp. 46–58.
Shirley, D.A., High-resolution X-ray photoemission spectrum of the valence bands of gold, Phys. Rev. B: Condens. Matter Mater. Phys., 1972, vol. 5, pp. 4709–4714.
Nefedov, V.N., Rentgenoelektronnaya spektroskopiya khimicheskikh soedinenii (X-ray Photoelectron Spectroscopy of Chemical Compounds), Moscow: Khimiya, 1984.
Sosulnikov, M.I. and Teterin, Yu.A., X-ray photoelectron studies of Ca, Sr, Ba and their oxides and carbonates, J. Electron Spectrosc. Relat. Phenom., 1992, vol. 59, pp. 111–126.
NIST X-ray Photoelectron Spectroscopy Database, Version 4.1, Gaithersburg: National Inst. of Standards and Technology, 2012). https://doi.org/10.18434/T4T88K
ACKNOWLEDGMENTS
In this study, we used equipment purchased through the Development of the Moscow State University Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Tsarev
Rights and permissions
About this article
Cite this article
Teterin, Y.A., Rudin, V.N., Severin, A.V. et al. X-ray Photoelectron Spectroscopy Study of Mg Adsorption on Nanocrystalline Hydroxyapatite. Inorg Mater 56, 1011–1019 (2020). https://doi.org/10.1134/S0020168520100155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168520100155