Abstract
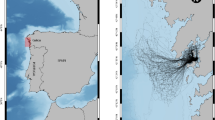
Five eastern Pacific yellowfin tuna samples (four northern: 10–25°N, 95–130°W, and one southern: 16–18°S, 95–97°W), collected from fishing vessels between 1994 and 2002 were analyzed for variation at seven microsatellite loci to evaluate their spatial genetic homogeneity. Single-locus homogeneity exact tests revealed significant genetic differentiation caused by three of seven loci. Pairwise sample comparisons of multilocus allele-frequency homogeneity tests and subpopulation division (F ST), revealed significant differentiation in comparisons between north equatorial samples and the south equatorial sample. AMOVA analysis among collections grouped as northern and southern populations separated by the equator in the eastern Pacific confirmed the differentiation observed. These results may be considered as preliminary evidence of the presence of discrete populations in the eastern Pacific yellowfin tuna. The possibility that the spatial differentiation observed corresponds to temporal variation or non-random sampling cannot be discarded. The genetic differences encountered need to be corroborated by increasing sample sizes, including temporal replicates, and by the use of an alternative marker.
Similar content being viewed by others
References
Food and Agricultural Organization of United Nations (FAO). FISHSTAT Plus Ver. 2.3. Universal software for fishery statistical time series. Fisheries Department, Fishery Information. Data and Statistics Unit, Rome. 2000.
Godsil HC, Greenhood EC. A comparison of the populations of yellowfin tuna (Neothunnus macropterus) from the eastern and central Pacific. Calif. Dept Fish Game Fish. Bull. 1951; 82: 33p.
Schaefer MB. Morphometric comparison of yellowfin tuna from southeast Polynesia, Central America, and Hawaii. Inter-Am. Trop. Tuna Comm. Bull. 1955; 1: 89–136.
Kurogane K, Hiyama Y. Morphometric comparison of the yellowfin tuna taken from the Equatorial Pacific. Jpn. Soc. Sci. Fish. Bull. 1957; 23: 388–293.
Schaefer KM. Morphometric analysis of yellowfin tuna, Thunnus albacares, from the eastern Pacific ocean. Bull. Inter-Am. Trop. Tuna Comm. 1989; 19: 387–427.
Schaefer KM. Geographical variation in morphometric characters and gill-raker counts of yellowfin tuna Thunnus albacares, from the Pacific Ocean. Fish. Bull. 1991; 91: 690–698.
Suzuki Z, Tomlinson PK, Honma M. Population structure of Pacific Yellowfin tuna. Bull. Inter-Am. Trop. Tuna Comm. 1978; 17: 273–441.
Fink BD, Bayliff WH. Migrations of yellowfin and skipjack tuna in the eastern Pacific Ocean as determined by tagging experiments, 1952–64. Inter-Am. Trop. Tuna Comm. Bull. 1978; 15: 1–227.
Barret I, Tsuyuki H. Serum transferrin polymorphism in some scombroid fishes. Copeia 1967; 3: 551–557.
Fujino K. Immunological and biochemical genetics of tunas. Trans. Am. Fish. Soc. 1970; 99: 152–178.
IATTC (Inter-American Tropical Tuna Commission). Annual report of the Inter-American Tropical Tuna Commission. IATTC. LaJolla, CA. 1975.
Scoles DR, Graves JE. Genetic analysis of the population structure of yellowfin tuna, Thunnus albacares, from the Pacific Ocean. Fish. Bull. 1993; 91: 690–698.
Ward RD, Elliott NG, Grewe PM, Smolenski A. Allozyme and mitochondrial DNA variation in yellowfin tuna (Thunnus albacares) from the Pacific Ocean. Mar. Biol. 1994; 118: 531–539.
Sharp GD. Behavioral and physiological properties of tunas and their effects on vulnerability to fishing gear. In: Sharp GD, Dizon AE (eds). The Physiological Ecology of Tunas. Academic Press, New York. 1978; 397–449.
Ward RD, Elliott NG, Innes BH, Smolenski AJ, Grewe PM. Global population structure of yellowfin tuna, Thunnus albacares, inferred from allozyme and mitochondrial DNA variation. Fish. Bull. 1997; 95: 566–575.
Appleyard SA, Grewe PM, Innes BH, Ward RD. Population structure of yellowfin tuna (Thunnus albacares) in the western Pacific Ocean, inferred from microsatellite loci. Mar. Biol. 2001; 139: 383–393.
Bentzen P, Taggart TC, Ruzzante DE, Cook D. Microsatellite polymorphism and the population structure of Atlantic cod (Gadus morhua) in the northwest Atlantic. Can. J. Fish. Aquat. Sci. 1996; 53: 2706–2721.
Gold JR, Kristmundsdóttir ÁÝ, Richardson LR. Mitochondrial DNA variation in king mackerel (Scomberomorus cavalla) from the western Atlantic Ocean and Gulf of Mexico. Mar. Biol. 1997; 129: 221–232.
Grant SW, Clark AM, Bowen BW. Why restriction fragment length polymorphism analysis of mitochondrial DNA failed to resolve sardine (Sardinops) biogeography: insights from mitochondrial DNA cytochrome b sequences. Can. J. Fish. Aquat. Sci. 1998; 55: 2539–2547.
O’Connell M, Wright JM. Microsatellite DNA in fishes. Rev. Fish Biol. Fish 1997; 7: 331–363.
Hoffman IJ, Amos W. Microsatellite genotyping errors: detection approaches, common sources and consequences for paternal exclusion. Mol. Ecol. 2005; 14: 599–612.
McDowell JR, Diaz-Jaimes P, Graves JE. Isolation and characterization of seven tetra-nucleotide microsatellite loci from Atlantic northern bluefin tuna Thunnus thynnus thynnus. Mol. Ecol. Notes 2002; 2: 214–216.
Carlsson J, McDowell JR, Díaz-Jaimes P, Carlsson JEL, Boles SB, Gold JR, Graves JE. Microsatellite and mitochondrial DNA analyses of Atlantic bluefin tuna (Thunnus thynnus thynnus) population structure in the Mediterranean Sea. Mol. Ecol. 2004; 13: 3345–3356.
Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning: A Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory. Cold Sprig Harbor, NY. 1989.
Laird PW, Zijdervel A, Linders K, Rudnicki MA, Jaenisch R, Berns A. Simplified mammalian DNA isolation procedure. Nucleic Acids Res. 1991; 19: 4293.
Takagi M, Okamura T, Chow S, Taniguchi N. PCR primers for microsatellite loci in tuna species of the genus Thunnus and its application for population genetic study. Fish. Sci. 1999; 65: 571–576.
van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P. Micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol. Ecol. Notes 2004; 4: 535–538.
Wattier E, Enger CL, Saumitou-Laprade P, Valero M. Short allele dominance as a source of heterozygote deficiency at microsatellite loci: experimental evidence at the dinucleotide locus Gv1CT in Gracilaria gracilis (Rodophyta). Mol. Ecol. 1998; 7: 1569–1573.
Schneider S, Kueffer JM, Roessli D, Excoffier L. Arlequin Ver. 1.1: a Software for Population Genetic Data Analysis. Genetics and Biometry Laboratory, University of Geneva, Switzerland. 1997.
Guo SW, Thompson EA. Performing the exact test for Hardy-Weinberg proportion for multiple alleles. Biometrics 1992; 48: 361–372.
Weir BS, Cockerham CC. Estimating F statistics for the analysis of population structure. Evolution 1984; 38: 1358–1370.
Goudet J. Fstat (vers. 1.2): a computer program to calculate F-statistics. J. Hered. 1995; 86: 485–486.
Raymond M, Rousset F. Genepop (version 1.2): population genetics software for exact tests and ecumenicism. J. Hered. 1995; 86: 248–249.
Rice WR. Analyzing tables of statistical tests. Evolution 1989; 43: 223–225.
Cabin JR, Mitchell RJ. To Bonferroni or not Bonferroni: when and how are the questions. ESA Bull. 2000; 81: 246–248.
Moran DM. Arguments for rejecting the sequential Boferroni in ecological studies. Oikos 2003; 100: 403–405.
Perneger TV. What’s wrong with the Bonferroni adjustments. BMJ 1998; 316: 1236–1238.
Klein AP, Kovac I, Sorant AJM, Baffoe-Bonnie A, Doan QB, Ibay G, Lockwood E, Mandal D, Santhosh L, Weissbecker K, Woo J, Zambelli-Weiner A, Zhang J, Naiman DQ, Malley J, Bailey JEW. Importance sampling method of correction for multiple testing in affected sib-pair linkage analysis. BMC Genet. 2003; 4: S73.
Ueyanagi S, Mori K, Nishikawa Y. Research on distribution of larvae. S. Ser. Far Seas Fish. Res. Lab. 1969; 1: 7–12.
Schaefer KM. Reproductive biology of the yellowfin tuna (Thunnusalbacares) in the eastern Pacific ocean. Bull. Inter-Am. Trop. Tuna Comm. 1998; 21: 205–272.
Wyrtki K. Circulation and water masses in the eastern equatorial Pacific Ocean. Int. J. Oceanol. Limnol. 1967; 1: 117–147.
Ely B, Viñas J, Alvarado-Bremer J, Black D, Lucas L, Covello K, Labrie AV, Thelen E. Consequences of the historical demography on the global population structure of two highly migratory cosmopolitan marine fishes: the yellowfin tuna (Thunnus albacares) and the skipjack tuna (Katsuwonis pelamis). BMC Evol. Biol. 2005; 5: 19.
Waples RS. Separating the wheat from the chaff: patterns of genetic differentiation in high flow species. J. Hered. 1998; 89: 438–450.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Díaz-Jaimes, P., Uribe-Alcocer, M. Spatial differentiation in the eastern Pacific yellowfin tuna revealed by microsatellite variation. Fish Sci 72, 590–596 (2006). https://doi.org/10.1111/j.1444-2906.2006.01188.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2006.01188.x