Summary
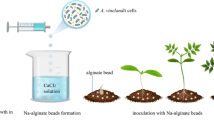
Shoot cultures of three Hibiscus moscheutos (L.) cultivars were infested with micro-arthropods (mites). Nodal segments (1 cm long) were excised from these cultures and encapsulated in a sodium alginate gelled Driver and Kuniyuki Walnut DKW medium containing 10, 50, or 100 mg l−1 acephate (insecticide) or 10 mg l−1 acephate plus 0, 50, or 100 mg l−1 benomyl (fungicide), then placed in refrigerated (5°C) darkness for 4 wk. Acephate was tested alone if visible fungus was not touching the shoot masses and benomyl was tested if fungus was in contact with the proliferating shoots. Cold-stored encapsulated nodes were then placed on DKW medium with 0.1 μM thidiazuron for 6 wk for subsequent shoot development. The presence of acephate in the encapsulation medium completely eradicated or killed the mites, with 38–69% of cultures fungus-free; 12% of the fungal-contaminated nodes encapsulated with 100 mg l−1 benomyl were fungus-free.
Similar content being viewed by others
References
Ali, A. D. Integrated arthropod pest management in plant tissue culture production. Comb. Proc. Intl Plant Prop. Soc. 37:104–107; 1987.
Bhagwat, B.; Lane, W. D. Eliminating thrips from in vitro shoot cultures of apple with insecticides. HortScience 38(1):97–100; 2003.
Blake, J. Mites and thrips as bacterial and fungal vectors between plant tissue cultures. Acta Hort. 225:163–166; 1988.
Driver, J. A.; Kuniyuki, A. H. In vitro propagation of paradox walnut rootstock. HortScience 19:507–509; 1984.
Gaspar, T.; Levers, C.; Penel, C.; Greppin, H.; Reid, D. M.; Thorpe, T. A. Plant hormones and plant growth regulators in plant tissue culture. In Vitro Cell. Dev. Biol. Plant 32:272–289; 1996.
Hartmann, H.T.; Kester, D. E.; Davies, F. T., Jr.; Geneve, R. L. Plant propagation: principles and practices, 7th edn. Upper Saddle River, NJ: Prentice Hall 2002:295–296.
Klocke, J. A.; Myers, P. Chemical control of thrips on cultured Simmondsia chinensis (Jojoba) shoots. HortScience 19(3):400; 1984.
Leifert, C.; Morris, C. E.; Waites, W. M. Ecology of microbial saprophytes and pathogens in tissue culture and field-grown plants: reasons for contamination problems in vitro. Crit. Rev. in Plant Sci. 13(2):139–183; 1994.
Piccioni, E.; Standardi, A. Encapsulation of micropropagated buds of six woody species. Plant Cell Tiss. Organ Cult. 42:221–226; 1995.
Pressman, E.; Palevitch, D. Cytokinin-like activities of benomyl as a senescence inhibitor of broccoli heads. HortScience 8(6):496–497; 1973.
Pype, J.; Everaert, K.; Debergh, P. Contamination by micro-arthropods in plant tissue cultures. In: Cassells, A. C., ed. Pathogen and microbial contamination mangaement in micropropagation. Dordrecht, The Netherlands: Kluwer 1997:259–266.
Riesen, T. K.; Close, R. C. Endophytic fungi in propiconazole-treated and untreated barley leaves. Mycologia 79:546–552; 1987.
SAS Institute, Inc. SAS Proprietary Software Version 8. Cary, NC: SAS Institute; 2002.
Smith, R. Plant tissue culture techniques and experiments, 2nd edn. San Diego, California: Academic Press; 2000: 77.
West, T. P.; Preece, J. E. Effects of thidiazuron and nutrient salt formulations on micropropagation of hardy hibiscus (Hibiscus moscheutos L.). Acta Hort. (ISHS) 630:293–297; 2004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
West, T.P., Preece, J.E. Use of acephate, benomyl and alginate encapsulation for eliminating culture mites and fungal contamination from in vitro cultures of hardy hibiscus (Hibiscus moscheutos L.). In Vitro Cell.Dev.Biol.-Plant 42, 301–304 (2006). https://doi.org/10.1079/IVP2006774
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2006774