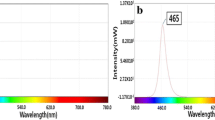
Abstract
Aquaculture activities are increasing worldwide, stimulated by the progressive reduction of natural fish stocks in the oceans. However, these activities also suffer heavy production and financial losses resulting from fish infections caused by microbial pathogens, including multidrug resistant bacteria. Therefore, strategies to control fish infections are urgently needed, in order to make aquaculture industry more sustainable. Antimicrobial photodynamic therapy (aPDT) has emerged as an alternative to treat diseases and prevent the development of antibiotic resistance by pathogenic bacteria. The aim of this work was to evaluate the applicability of aPDT to inactivate pathogenic fish bacteria. To reach this objective a cationic porphyrin Tri-Py+-Me-PF was tested against nine pathogenic bacteria isolated from a semi-intensive aquaculture system and against the cultivable bacteria of the aquaculture system. The ecological impact of aPDT in the aquatic environment was also tested on the natural bacterial community, using the overall bacterial community structure and the cultivable bacteria as indicators. Photodynamic inactivation of bacterial isolates and of cultivable bacteria was assessed counting the number of colonies. The impact of aPDT in the overall bacterial community structure of the aquaculture water was evaluated by denaturing gel gradient electrophoresis (DGGE). The results showed that, in the presence of Tri-Py+-Me-PF, the growth of bacterial isolates was inhibited, resulting in a decrease of ≈7-8 log after 60-270 min of irradiation. Cultivable bacteria were also considerably affected, showing decreases up to the detection limit (≈2 log decrease on cell survival), but the inactivation rate varied significantly with the sampling period. The DGGE fingerprint analyses revealed changes in the bacterial community structure caused by the combination of aPDT and light. The results indicate that aPDT can be regarded as a new approach to control fish infections in aquaculture systems, but it is clearly more difficult to inactivate the complex natural bacterial communities of aquaculture waters than pure cultures of bacteria isolated from aquaculture systems. Considering the use of aPDT to inactivate pathogenic microbial community of aquaculture systems the monitoring of microorganisms is needed in order to select the most effective conditions.
Similar content being viewed by others
Notes and references
M. Sorimachi and T. Hara, Characteristics and pathogenicity of a virus isolated from yellowtail fingerlings showing ascites, Fish Pathol., 1985, 19, 231–238.
H. H. Huss, Assurance of Seafood Quality, FAO Fishery Technical Paper, 1994, No. 334. FAO, Rome, Italy.
Z. Shao, Aquaculture pharmaceuticals and biological: current perspectives and future, Adv. Drug Delivery Rev., 2001, 50, 229–243.
A. E. Toranzo, B. Magariños and J. L. Romalde, A review of the main bacterial fish diseases in mariculture systems, Aquaculture, 2005, 246, 37–61.
F. P. Meyer, Aquaculture disease and health management, J. Anim. Sci., 1991, 69, 4201–4208.
A. Almeida, A. Cunha, N. C. M. Gomes, E. Alves, L. Costa and M. A. F. Faustino, Phage Therapy and Photodynamic Therapy: Low Environmental Impact Approaches to Inactivate Microorganisms in Fish Farming Plants, Mar. Drugs, 2009, 7, 268–313.
T. Defoirdt, N. Boon, P. Sorgeloos, W. Verstraete and P. Bossier, Alternatives to antibiotics to control bacterial infections: luminescent vibriosis in aquaculture as an example, Trends Biotechnol., 2007, 25, 472–479.
M. Wainwright, Photodynamic antimicrobial chemotherapy (PACT), J. Antimicrob. Chemother., 1998, 42, 13–28.
G. Jori and S. B. Brown, Photosensitized inactivation of microorganisms, Photochem. Photobiol. Sci., 2004, 3, 403–405.
G. Jori, C. Fabris, M. Soncin, S. Ferro, O. Coppellotti, D. Dei, L. Fantetti, G. Chiti and G. Roncucci, Photodynamic therapy in the treatment of microbial infections: basic principles and perspective applications, Lasers Surg. Med., 2006, 38, 468–481.
T. Maisch, A new strategy to destroy antibiotic resistant microorganisms: antimicrobial photodynamic treatment., Mini Rev. Med. Chem., 2009, 9, 974–983.
F. M. Lauro, P. Pretto, L. Covolo, G. Jori and G. Bertoloni, Photoinac-tivation of bacterial strains involved in periodontal diseases sensitized by porphycene-polylysine conjugates, Photochem. Photobiol. Sci., 2002, 1, 468–470.
L. A. Pedigo, A. J. Gibbs, R. J. Scott and C. N. Street, Absence of bacterial resistance following repeat exposure to photodynamic therapy, Proc. SPIE, 2009, 73803H.
A. Tavares, C. M. B. Carvalho, M. A. F. Faustino, M. G. P. M. S. Neves, J. P. C. Tomé, A. C. Tomé, J. A. S. Cavaleiro, Â. Cunha, N. C. M. Gomes, E. Alves and A. Almeida, Antimicrobial Photodynamic Therapy: Study of Bacterial Recovery Viability and Potential Development of Resistance after Treatment, Mar. Drugs, 2010, 8, 91–105.
K. D. Winckler, Special section: focus on anti-microbial photody-namic therapy (PDT), J. Photochem. Photobiol., B, 2007, 86, 43–44.
G. Jori and O. Coppellotti, Inactivation of pathogenic microorganisms by photodynamic techniques: mechanistic aspects and perspective applications, Anti-Infect. Agents Med. Chem., 2007, 6, 119–131.
T. J. Dougherty, C. J. Gomer, B. W. Henderson, G. Jori, D. Kessel, M. Korbelik, J. Moan and Q. Peng, Photodynamic therapy, J. Natl. Cancer Inst., 1998, 90, 889–905.
M. R. Hamblin and T. Hasan, Photodynamic therapy: a new antimicrobial approach to infectious disease?, Photochem. Photobiol. Sci., 2004, 3, 436–450.
R. Bonnett, Chemical aspects of photodynamic therapy, Gordon and Breach Science, Amsterdam, 2000.
M. Wainwright, Methylene blue derivatives - suitable photoantimicro-bials for blood product disinfection?, Int. J. Antimicrob. Agents, 2000, 16, 381–394.
T. Maisch, Anti-microbial photodynamic therapy: useful in the future?, Lasers Med. Sci., 2006, 22, 83–91.
R. Bonnett, M. A. Krysteva, I. G. Lalov and S. V. Artarsky, Water disinfection using photosensitizers immobilized on chitosan, Water Res., 2006, 40, 1269–1275.
M. Jemli, Z. Alouini, S. Sabbahi and M. Gueddari, Destruction of fecal bacteria in wastewater by three photosensitizers, J. Environ. Monit., 2002, 4, 511–516.
C. M. B. Carvalho, A. Gomes, S. C. D. Fernandes, A. C. B. Prata, M. A. Almeida, M. A. Cunha, J. P. C. Tome, M. A. F. Faustino, M. Neves and A. C. Tome, Photoinactivation of bacteria in wastewater by porphyrins: bacterial beta-galactosidase activity and leucine-uptake as methods to monitor the process, J. Photochem. Photobiol., B, 2007, 88, 112–118.
L. Costa, E. Alves, C. Carvalho, J. Tomé, M. Faustino, M. Neves, A. Tomé, J. Cavaleiro, Â. Cunha and A. Almeida, Sewage bacteriophage photoinactivation by cationic porphyrins: a study of charge effect, Photochem. Photobiol. Sci., 2008, 7, 415–422.
E. Alves, L. Costa, C. Carvalho, J. Tome, M. Faustino, M. Neves, A. Tome, J. Cavaleiro, A. Cunha and A. Almeida, Charge effect on the photoinactivation of Gram-negative and Gram-positive bacteria by cationic meso-substituted porphyrins, BMC Microbiol., 2009, 9, 70.
M. Magaraggia, F. Faccenda, A. Gandolfi and G. Jori, Treatment of mi-crobiologically polluted aquaculture waters by a novel photochemical technique of potentially low environmental impact, J. Environ. Monit., 2006, 8, 923–931.
P. N. Wong, S. K. Mak, M. W. Lo, K. Y. Lo, G. M. W. Tong, Y. Wong and A. K. M. Wong, Vibrio vulnificus peritonitis after handling of seafood in a patient receiving CAPD, Am. J. Kidney Dis., 2005, 46, e87–e90.
D. Faust, K. H. Funken, G. Horneck, B. Milow, J. Ortner, M. Sattlegger, M. Schafer and C. Schmitz, Immobilized photosensitizers for solar photochemical applications, Sol. Energy, 1999, 65, 71–74.
M. E. Jiménez-Hernández, F. Manjón, D. Garcia-Fresnadillo and G. Orellana, Solar water disinfection by singlet oxygen photogenerated with polymer-supported Ru(II) sensitizers, Sol. Energy, 2006, 80, 1382–1387.
M. Krouit, J. Bras and M. N. Belgacem, Cellulose surface grafting with polycaprolactone by heterogeneous click-chemistry, Eur. Polym. J., 2008, 44, 4074–4081.
B. C. Cho and F. Azam, Biogeochemical significance of bacterial biomass in the ocean’s euphotic zone, Mar. Ecol.: Prog. Ser., 1990, 63, 253–259.
L. R. Pomeroy, Status and future needs in protozoan ecology, in: Protozoan and their role in marine processes, ed. P. C. Reid, C. M. Turley and P. H. Burkhill, Springer-Verlag, Heidelberg, 1991, vol. 25, pp. 475–492.
J. J. Cole, S. Findlay and M. L. Pace, Bacterial production in fresh and saltwater ecosystems: a cross-system overview, Mar. Ecol.: Prog. Ser., 1988, 43, 1–10.
H. W. Ducklow and C. A. Carlson, Oceanic bacterial production, Adv. Microb. Ecol., 1992, 12, 113–181.
B. C. Cho and F. Azam, Major role of bacteria in biogeochemical fluxes in the ocean’s interior, Nature, 1988, 332, 441–443.
D. J. W. Moriarty, The role of microorganisms in aquaculture ponds, Aquaculture, 1997, 151, 333–349.
M. A. Almeida, M. A. Cunha and F. Alcântara, Loss of estuarine bacteria by viral infection and predation in microcosm conditions, Microb. Ecol., 2001, 42, 562–571.
R. K. Saiki, D. H. Gelfand, S. Stoffel, S. J. Scharf, R. Higuchi, G. T. Horn, K. B. Mullis and H. A. Erlich, Primer-directed enzymatic amplification of DNA with thermostable DNA polymerase, Science, 1988, 239, 487–491.
M. Sela, F. H. Jr. White and C. B. Anfinsen, Reductive cleavage of disulfide bridges in ribonuclease, Science, 1957, 125, 691–692.
G. E. Sheridan, C. I. Masters, J. A. Shallcross and B. M. MacKey, Detection of mRNA by reverse transcription-PCR as an indicator of viability in Escherichia coli cells, Appl. Environ. Microbiol., 1998, 64, 1313–1318.
T. Demidova and M. Hamblin, Effect of cell-photosensitizer binding and cell density on microbial photoinactivation, Antimicrob. Agents Chemother., 2005, 49, 2329–2335.
S. Banfi, E. Caruso, L. Buccafurni, V. Battini, S. Zazzaron, P. Barbieri and V. Orlandi, Antibacterial activity of tetraaryl-porphyrin photosensitizers: An in vitro study on Gram negative and Gram positive bacteria, J. Photochem. Photobiol., B, 2006, 85, 28–38.
D. Caminos and E. Durantini, Photodynamic inactivation of Es-cherichia coli immobilized on agar surfaces by a tricationic porphyrin, Bioorg. Med. Chem., 2006, 14, 4253–4259.
H. Li, O. S. Fedorova, A. N. Grachev, W. R. Trumble, G. A. Bohach and L. Czuchajowski, A series of meso-tris(N-methyl-pyridiniumyl)-(4-alkylamidophenyl) porphyrins: Synthesis, interaction with DNA and antibacterial activity, Biochim. Biophys. Acta, Gene Struct. Expression, 1997, 1354, 252–260.
S. Mettath, B. R. Munson and R. K. Pandey, DNA interaction and pho-tocleavage properties of porphyrins containing cationic substituents at the peripheral position, Bioconjugate Chem., 1999, 10, 94–102.
F. P. Imray and D. G. MacPhee, The role of DNA polymerase I and the rec system in survival of bacteria and bacteriophages damaged by the photodynamic action of acridine-orange, MGG, Mol. Gen. Genet., 1973, 123, 289–298.
T. A. Dahl, W. R. Midden and P. E. Hartman, Pure singlet oxygen cytotoxicity for bacteria, Photochem. Photobiol., 1987, 46, 345–352.
T. A. Dahl, W. R. Midden and P. E. Hartman, Comparison of killing of gram-negative and gram-positive bacteria by pure singlet oxygen, J. Bacteriol., 1989, 4, 2188–2194.
K. E. Sanderson, T. MacAlister, J. W. Costerton and K. J. Cheng, Permeability of lipopolysaccharide-deficient (rough) mutants of Salmonella typhimurium to antibiotics, lysozyme, and other agents, Can. J. Microbiol., 1974, 20, 1135–1145.
M. Szpakowska, K. Lasocki and J. Grzybowski, Photodynamic activity of the haematoporphyrin derivative with rutin and arginine substituents (HpD-Rut (2)-Arg(2)) against Staphylococcus aureus and Pseudomonas aeruginosa, Pharmacol. Res., 2001, 44, 243–246.
M. Grinholc, B. Szramka, J. Kurlenda, A. Graczyk and K. P. Bielawski, Bactericidal effect of photodynamic inactivation against methicillin-resistant and methicillin-susceptible Staphylococcus aureus is strain-dependent, J. Photochem. Photobiol., B, 2008, 90, 57–63.
D. Evenberg, R. van Boxtel, B. Lugtenberg, F. Schurer, J. Blommaert and R. Bootsma, Cell surface of the fish pathogenic bacterium Aeromonas salmonicida. I. Relationship between autoagglutination and the presence of a major cell envelope protein, Biochim. Biophys. Acta, Biomembr., 1982, 684, 241–248.
W. W. Kay, J. T. Buckley, E. E. Ishiguro, B. M. Phipps, J. P. L. Monette and T. J. Trust, Purification and disposition of a surface protein associated with virulence of Aeromonas salmonicida, J. Bacteriol., 1981, 147, 1077–1084.
W. W. Kay, B. M. Phipps, E. E. Ishiguro and T. J. Trust, Porphyrin binding by the surface array virulence protein of Aerononas salmonicida, J. Bacteriol., 1985, 164, 1332–1336.
C. Pereira, Use of bacteriophages on the inactivation of pathogenic bacteria in aquaculture system, MSc Thesis, University of Aveiro, 2009.
I. S. Henriques, A. Almeida, A. Cunha and A. Correia, Molecular sequence analysis of prokaryotic diversity in the middle and outer sections of the Portuguese estuary Ria de Aveiro, FEMS Microbiol. Ecol., 2004, 49, 269–279.
P. Amato, M. Ménager, M. Sancelme, P. Laj, G. Mailhot and A. Delort, Microbial population in cloud water at the Puy de Dôme: Implications for the chemistry of clouds, Atmos. Environ., 2005, 39, 4143–4153.
A. P. J. Maestrin, A. O. Ribeiro, A. C. Tedesco, C. R. Neri, F. S. Vinhado, O. A. Serra, P. R. Martins, Y. Iamamoto, A. M. G. Silva, A. C. Tomé, M. Neves and J. A. S. Cavaleiro, A novel chlorin derivative of meso-tris(pentafluorophenyl)-4-pyridylporphyrin: Synthesis, photo-physics and photochemical properties, J. Braz. Chem. Soc., 2004, 15, 923–930.
J. P. C. Tome, M. G. P. M. S. Neves, A. C. Tome, J. A. S. Cavaleiro, M. Soncin, M. Magaraggia, S. Ferro and G. Jori, Synthesis and antibacterial activity of new poly-S-lysine-porphyrin conjugates, J. Med. Chem., 2004, 47, 6649–6652.
J. Romalde, Photobacterium damselae subsp. piscicida: an integrated view of a bacterial fish pathogen, Int. Microbiol., 2002, 5, 3–9.
M. M. Moeseneder, W. Christian and J. H. Gerhard, Horizontal and vertical complexity of attached and free-living bacteria of the eastern Mediterranean Sea, determined by 16S rDNA and 16S rRNA fingerprints, Limnol. Oceanogr., 2001, 46, 95–107.
W. G. Weisburg, S. M. Barns, D. A. Pelletier and D. J. Lane, 16S ribosomal DNA amplification for phylogenetic study, J. Bacteriol., 1991, 173, 697–703.
H. Heuer, M. Krsek, P. Baker, K. Smalla and E. M. H. Wellington, Analysis of actinomycete communities by specific amplification of genes encoding 16S rRNA and gel-electrophoretic separation in denaturing gradients, Appl. Environ. Microbiol., 1997, 63, 3233–3241.
J. Sambrook, E. F. Fritsch and T. Maniatis, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1989.
K. Smalla, G. Wieland, A. Buchner, A. Zock, J. Parzy, S. Kaiser, N. Roskot, H. Heuer and G. Berg, Bulk and Rhizosphere Soil Bacterial Communities Studied by Denaturing Gradient Gradient Gel Electrophoresis: Plant-Dependent Enrichment and Seasonal Shifts Revealed, Appl. Environ. Microbiol., 2001, 67, 4742–4751.
A. Ramette, Multivariate analyses in microbial ecology, FEMS Microbiol. Ecol., 2007, 62, 142–160.
K. R. Clarke and R. N. Gorley, Primer v5: User manual/tutorial. PRIMER-E, Plymouth, UK, 2001, p. 9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arrojado, C., Pereira, C., Tomé, J.P.C. et al. Applicability of photodynamic antimicrobial chemotherapy as an alternative to inactivate fish pathogenic bacteria in aquaculture systems. Photochem Photobiol Sci 10, 1691–1700 (2011). https://doi.org/10.1039/c1pp05129f
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c1pp05129f