Abstract
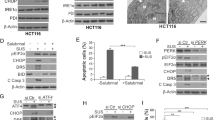
Understanding the mechanisms that underlie the antitumour activity of non-steroidal anti-inflammatory drugs (NSAIDs) against colorectal cancer will allow the development of more effective and specific chemopreventative agents. Modulation of the NF-κB pathway has been implicated as a key effector of the antitumour effect of aspirin, but the effects of non-aspirin NSAIDs on this pathway have yet to be fully defined. Here, we demonstrate that sulindac, sulindac sulfone and indomethacin activate the NF-κB pathway in colorectal cancer cells, as determined by western blot analysis of cytoplasmic levels of IκBα and immunocytochemical analysis of nuclear NF-κB/RelA. Furthermore, we show that all of these NSAIDs induce nucleolar translocation of the RelA subunit of NF-κB. Using RelA deleted for the previously described nucleolar localization signal, we demonstrate that this response is causally involved in the apoptotic effects of these agents. Finally, we demonstrate that NSAID-mediated nucleolar translocation of RelA is associated with downregulation of NF-κB-driven transcription and of the NF-κB target gene, ICAM-1. These data identify nucleolar translocation of RelA and the associated repression of the NF-κB-driven transcription as a central molecular mechanism of NSAID-mediated growth inhibition and apoptosis. As well as providing new understanding of the molecular determinants of RelA function, these findings also have relevance to the development of novel chemotherapeutic and chemopreventative agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brandli D, Sarkissian WE, Ng SC, Paulus HE . (1991). Individual variability in concentrations of urinary sulindac sulfide. Clin Pharmacol Ther 50: 650–655.
Bresalier RS, Sandler RS, Quan H, Bolognese JA, Oxenius B, Horgan K et al. (2005). Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 352: 1092–1102.
Campbell KJ, Rocha S, Perkins ND . (2004). Active repression of antiapoptotic gene expression by RelA(p65) NF-kappa B. Mol Cell 13: 853–865.
Din FV, Dunlop MG, Stark LA . (2004). Evidence for colorectal cancer cell specificity of aspirin effects on NF kappa B signalling and apoptosis. Br J Cancer 91: 381–388.
Dippold W, Wittig B, Schwaeble W, Mayet W, Meyer zum Buschenfelde KH . (1993). Expression of intercellular adhesion molecule 1 (ICAM-1, CD54) in colonic epithelial cells. Gut 34: 1593–1597.
Dutta J, Fan Y, Gupta N, Fan G, Gelinas C . (2006). Current insights into the regulation of programmed cell death by NF-kappa B. Oncogene 25: 6800–6816.
Gardner SH, Hawcroft G, Hull MA . (2004). Effect of nonsteroidal anti-inflammatory drugs on beta-catenin protein levels and catenin-related transcription in human colorectal cancer cells. Br J Cancer 91: 153–163.
Grau MV, Rees JR, Baron JA . (2006). Chemoprevention in gastrointestinal cancers: current status. Basic Clin Pharmacol Toxicol 98: 281–287.
Hoffmann A, Natoli G, Ghosh G . (2006). Transcriptional regulation via the NF-kappa B signaling module. Oncogene 25: 6706–6716.
Ledebur HC, Parks TP . (1995). Transcriptional regulation of the intercellular adhesion molecule-1 gene by inflammatory cytokines in human endothelial cells. Essential roles of a variant NF-kappa B site and p65 homodimers. J Biol Chem 270: 933–943.
Perkins ND . (2004). NF-kappa B: tumor promoter or suppressor? Trends Cell Biol 14: 64–69.
Smith M, Hawcroft G, Hull MA . (2000). The effect of non-steroidal anti-inflammatory drugs on human colorectal cancer cells. Evidence of different mechanisms of action. Eur J Cancer 36: 664–674.
Stark LA, Din FVN, Zwacka RM, Dunlop MG . (2001). Aspirin-induced activation of the NF-κB signalling pathway: a novel mechanism for aspirin-mediated apoptosis in colon cancer cells. FASEB J 7: 1273–1275, and 10.1096/fj.00-0529fje, 2002.
Stark LA, Dunlop MG . (2005). Nucleolar sequestration of RelA (p65) regulates NF-kappa B-driven transcription and apoptosis. Mol Cell Biol 25: 5985–6004.
Stark LA, Reid K, Sansom OJ, Din FV, Guichard S, Mayer I et al. (2007). Aspirin activates the NF-kappa B signalling pathway and induces apoptosis in intestinal neoplasia in two in vivo models of human colorectal cancer. Carcinogenesis 28: 968–976.
Sun Y, Sinicrope FA . (2005). Selective inhibitors of MEK1/ERK44/42 and p38 mitogen-activated protein kinases potentiate apoptosis induction by sulindac sulfide in human colon carcinoma cells. Mol Cancer Ther 4: 51–59.
Thoms HC, Dunlop MG, Stark LA . (2007). p38-Mediated inactivation of cyclin D1/cyclin-dependent kinase 4 stimulates nucleolar translocation of RelA and apoptosis in colorectal cancer cells. Cancer Res 67: 1660–1669.
Vainer B, Horn T, Nielsen OH . (2006). Colonic epithelial cell expression of ICAM-1 relates to loss of surface continuity: a comparative study of inflammatory bowel disease and colonic neoplasms. Scand J Gastroenterol 41: 318–325.
Wolfe MM, Lichtenstein DR, Singh G . (1999). Gastrointestinal toxicity of nonsteroidal antiinflammatory drugs. N Engl J Med 340: 1888–1899.
Acknowledgements
We gratefully acknowledge the gifts from Ron T Hay (University of Dundee) of the IκBα antibody and the 3enhancer CON-A luciferase reporter constructs. We also thank E Quarnstrom (University of Sheffield) for kindly providing the RelA-GFP expression vector and T Parks (Netherlands Institute for Developmental Biology) for providing the ICAM-1 luciferase reporter construct. The work was supported by grants from the Scottish Executive Chief Scientist's Office (K/MRS/50/C2719 and CZB/4/41) the AICR (02-330) and Cancer Research UK (C20658/A6656). MGD is supported by Cancer Research UK Programme Grant funding (C348/A3758 and C48/A6361).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loveridge, C., MacDonald, A., Thoms, H. et al. The proapoptotic effects of sulindac, sulindac sulfone and indomethacin are mediated by nucleolar translocation of the RelA(p65) subunit of NF-κB. Oncogene 27, 2648–2655 (2008). https://doi.org/10.1038/sj.onc.1210891
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210891
Keywords
This article is cited by
-
Albumin-based multidrug delivery system enriched in Golgi apparatus against metastatic breast cancer
Nano Research (2023)
-
The effect of sulindac on redox homeostasis and apoptosis-related proteins in melanotic and amelanotic cells
Pharmacological Reports (2023)
-
PSMB4 inhibits cardiomyocyte apoptosis via activating NF-κB signaling pathway during myocardial ischemia/reperfusion injury
Journal of Molecular Histology (2021)
-
RETRACTED ARTICLE: Cadherin 6 is activated by Epstein–Barr virus LMP1 to mediate EMT and metastasis as an interplay node of multiple pathways in nasopharyngeal carcinoma
Oncogenesis (2017)
-
Sulindac, a non-steroidal anti-inflammatory drug, mediates breast cancer inhibition as an immune modulator
Scientific Reports (2016)