Abstract
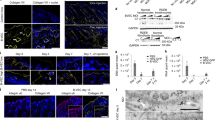
It is well documented that responses to growth factor treatment typically display bell-shaped dose responses that can significantly affect efficacy. Here we tested the hypothesis that nonviral liposomal gene delivery also displays this characteristic. We chose two different growth factors, keratinocyte growth factor (KGF) and insulin-like growth factor-I (IGF-I) CMV-driven transfecting constructs at three different concentrations and assessed efficacy on several physiological parameters that are descriptive of wound healing progress in a burn-wound healing model. Rats were given a 60% TBSA scald burn and randomly divided into one of seven groups to receive weekly subcutaneous injections of liposomes containing the cDNA for KGF (0.2 μg, 2.2 μg, or 22.2 μg), or liposomes containing the cDNA for IGF-I (0.2 μg, 2.2 μg, or 22.2 μg) at various concentrations, but constant liposome:DNA ratios and a LacZ gene (0.2 μg) CMV-driven construct for β-galactosidase as vehicle and marker gene. Transfection was confirmed by histology for β-galactosidase. Physiological efficacy was evaluated by measuring the wound healing parameters that define dermal and epidermal regeneration. Transfection products were found in the cytoplasm of rapidly dividing cells of the granulation tissue. Different doses of the nonviral cDNA gene transfer coding for KGF or IGF-I resulted in different outcomes for dermal and epidermal regeneration. There was a dose-dependent response to both growth factor gene transfers that was not dissimilar from that typically displayed by treatment with growth factor proteins. Both concentrations below and above the optimal concentration of DNA:liposomal preparations did not yield the results observed at the optimal concentration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yang KF et al. Optimizing liposome-mediated gene transfer in primary rat septo-hippocampal cell culture Neurosci Lett 1994 182: 287–290
Yang KF et al. Sustained expression of functional nerve growth in primary septo-hippocampal cell cultures by liposome-mediated gene transfer Neurosci Lett 1994 182: 291–294
Yang K et al. Regional and temporal profiles of c-fos and NGF mRNA expression in rat brain after lateral cortical impact injury in rats J Neurosci Res 1995 42: 571–578
Yang K et al. Increased expression of BDNF but not NT3 mRNA in rat brain after cortical impact injury J Neurosci Res 1996 44: 157–164
Yang K et al. Cationic lipid-mediated NGF gene transfection increases neurofilament phosphorylation Molec Brain Res 1996 43: 13–20
Pierre E et al. Insulin-like growth factor-I liposomal gene transfer and systemic growth hormone stimulate wound healing J Burn Care Rehab 1997 18: 287–291
Qiu YH et al. Activation of phosphatidylinositol 3-kinase by brain-derived neurotrophic factor gene transfection in septo-hippocampal cultures J Neurosci Res 1998 52: 192–200
Zou LL et al. Liposome-mediated NGF gene transfection following neuronal injury: potential therapeutic applications Gene Therapy 1999 6: 994–1005
Jeschke MG et al. IGF-I gene transfer in thermal trauma Gene Therapy 1999 6: 1015–1020
Jeschke MG et al. Effect of multiple gene transfers of insulin like growth factor I complementary DNA gene constructs in rats after thermal injury Arch Surg 1999 134: 1137–1141
Tao Z et al. Insulin-like growth factor-I cDNA gene transfer in vitro and in vivo Biochem Genet 2000 38: 41–55
Jeschke MG et al. Biodistribution and feasibility of nonviral IGF-I gene transfers in thermally injured skin Lab Invest 2000 80: 151–158
Yang K et al. In vitro studies of liposome-mediated gene transfection Perez-Polo JR (eds); Paradigms of Neural Injury; Methods of Neuroscience Academic Press 1996 290–297
Yang K et al. In vitro studies of liposome-mediated gene transfection
Vogel JC . Nonviral skin gene therapy Hum Gene Ther 2000 11: 2253–2259
Werner S et al. The function of KGF in morphogenesis and re-epithelialization of wounds Science 1994 266: 819–823
Werner S . Keratinocyte growth factor: a unique player in epithelial repair processes Cytok Growth Fact Rev 1998 9: 153–165
Rubin JS et al. Keratinocyte growth factor Cell Biol Int 1995 19: 399–411
Smola H, Thiekötter G, Fusenog NE . Mutual induction of growth factor gene expression by epidermal-dermal cell interaction J Cell Biol 1993 122: 417–429
Winkles JA et al. Differential expression of keratinocyte growth factor (KGF) and KGF receptor genes in human vascular smooth muscle cells and arteries J Cell Physiol 1997 173: 380–386
Finch PW et al. Human KGF is FGF-related with properties of paracrine effector of epithelial cells Science 1989 145: 752–755
Senaldi G et al. Keratinocyte growth factor protects murine hepatocytes from tumor necrosis-induced apoptosis in vivo and in vitro Hepatology 1998 27: 1584–1591
Marchese C et al. Modulation of keratinocyte growth factor receptor expression in human cultured keratinocytes Cell Growth Diff 1997 265: 989–997
Humbel RE . Insulin-like growth factor-I and factor-II Eur J Biochem 1990 190: 445–462
Baxter RC . Circulating levels and molecular distribution of the acid-labile (alpha) subunit of the high molecular weight insulin-like growth factor-binding protein complex J Clin Endocrinol Metab 1990 70: 1347–1353
Huang KF, Chung DH, Herndon DN . Insulin-like growth factor-1 (IGF-I) reduces gut atrophy and bacterial translocation after severe burn injury Arch Surg 1993 128: 47–54
Strock LL, Singh H, Abdullah A . The effect of insulin-like growth factor-1 on postburn hypermetabolism Surgery 1990 108: 161–164
Clemmons DR . Insulin-like growth factor-1 as an anabolic agent in catabolic states Ann Intern Med 1994 120: 596–597
Meyer NA, Barrow RE, Herndon DN . Combined insulin-like growth factor-1 and growth hormone improves weight loss and wound healing in burned rats J Trauma 1994 31: 1008–1012
Jeschke MG, Herndon DN, Barrow RE . Insulin-like growth factor-I plus insulin-like growth factor binding protein-3 affects the hepatic acute phase response and hepatic morphology in thermally injured rats Ann Surg 2000 231: 408–416
Martin P . Wound healing-aiming for perfect skin regeneration Science 1997 276: 75–81
Steenfos HH . Growth factors and wound healing Scand J Plast Reconstr Hand Surg 1994 28: 95–105
Alexander MY, Akhurst RJ . Liposome-mediated gene transfer and expression via the skin Hum Molec Genet 1995 4: 2279–2285
Felgner PL . Nonviral strategies for gene therapy Sci Am 1997 6: 102–106
Bangham AD . Physical structure and behavior of lipids and lipid enzymes Adv Lipid Res 1963 1: 65–104
Sharata HH, Katz KH . Liposomes Int J Dermatol 1996 35: 761–769
Felgner PL . Improvements in cationic liposomes for in vivo gene transfer Hum Gene Ther 1996 7: 1791–1793
Wheeler CJ, Felgner PL, Tsai YT . A novel cationic lipid greatly enhances plasmid DNA delivery and expression in mouse lung Proc Natl Acad Sci USA 1996 93: 11454–11459
Gao X, Huang L . Cationic liposome-mediated gene transfer Gene Therapy 1995 2: 71–72
Mahato RI et al. Biodistribution and gene/expression of lipid/plasmid complexes after systemic administration Hum Gene Ther 1998 9: 2083–2099
Lee ER, Marshall J, Siegel CS . Detailed analysis of structures and formulations of cationic lipids for efficient gene transfer to the lung Hum Gene Ther 1996 7: 1701–1717
Jeschke MG, Barrow RE, Perez-Polo JR, Herndon DN . Cholesterol-containing cationic liposomes attenuate type I acute phase proteins and pro-inflammatory cytokines in thermally injured rats Arch Surg 1999 134: 1098–1102
Hengge UR, Walker PS, Vogel JC . Expression of naked DNA in human, pig and mouse skin J Clin Invest 1996 97: 2911–2916
Sun L et al. Transfection with aFGF cDNA improves wound healing J Invest Dermatol 1997 108: 313–318
Singer AJ, Clark RAF . Cutaneous wound healing N Engl J Med 1999 341: 738–746
Herndon DN, Wilmore DW, Mason AD, JrPruitt BA, Jr . Development and analysis of a small animal model stimulating the human postburn hypermetabolic response J Surg Res 1978 25: 394–403
Herndon DN, Wilmore DW, Mason AD, JrPruitt BA, Jr . Development and analysis of a small animal model stimulating the human postburn hypermetabolic response
Acknowledgements
This study was supported by the Deutsche Forschungsgemeinschaft (DFG Je 233/1-2) and by the Clayton Foundation for Research.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jeschke, M., Richter, G., Herndon, D. et al. Therapeutic success and efficacy of nonviral liposomal cDNA gene transfer to the skin in vivo is dose dependent. Gene Ther 8, 1777–1784 (2001). https://doi.org/10.1038/sj.gt.3301589
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301589
Keywords
This article is cited by
-
Basic concepts, current evidence, and future potential for gene therapy in managing cutaneous wounds
Biotechnology Letters (2019)
-
Non-viral liposomal keratinocyte growth factor (KGF) cDNA gene transfer improves dermal and epidermal regeneration through stimulation of epithelial and mesenchymal factors
Gene Therapy (2002)