Abstract
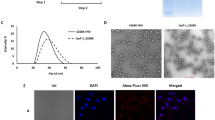
A nonviral gene delivery vector has been developed in our laboratory based on the cationic polymer, poly(2-(dimethylethylamino)ethyl methacrylate) (p(DMAEMA)). p(DMAEMA)-based polyplexes have been successfully used for the transfection of OVCAR-3 cells in vitro. However, these polyplexes were unable to transfect OVCAR-3 cells growing in the peritoneal cavity of nude mice after intraperitoneal administration, which could be ascribed to inactivation by components (including hyaluronic acid) present in the tumor ascitic fluid. The present work aimed at (a) protecting p(DMAEMA)-based polyplexes against destabilization or inactivation by polyanions such as hyaluronic acid present in tumor ascitic fluid and (b) enhancing cellular uptake of the protected p(DMAEMA)-based polyplexes by targeting with antibody Fab′ fragments. To fulfill these requirements, we have developed a detergent removal method to coat polyplexes with anionic lipids. With this method, spherical particles of ∼125 nm, which were protected from destabilization by polyanions, were obtained. More importantly, the transfection efficiency of lipopolyplexes was unaffected in the presence of hyaluronic acid, indicating that lipid coating of polyplexes protects against destabilization by hyaluronic acid. By conjugating antibody Fab′ fragments directed against the epithelial glycoprotein-2 to the lipidic surface of these lipopolyplexes, target cell–specific transfection of OVCAR-3 cells could be obtained in vitro. Cancer Gene Therapy (2001) 8, 405–413
Similar content being viewed by others
Article PDF
References
Robertson MW, III, Barnes MN, Rancourt C, et al. . Gene therapy for ovarian carcinoma Semin Oncol 1998 25: 397–406
Gomez-Navarro J, Siegal GP, Alvarez RD, et al . Gene therapy: ovarian carcinoma as the paradigm Am J Clin Pathol 1998 109: 444–467
Cristiano RJ . Viral and nonviral vectors for cancer gene therapy Anticancer Res 1998 18: 3241–3245
Robbins PD, Ghivizzani SC . Viral vectors for gene therapy Pharmacol Ther 1998 80: 35–47
Luo D, Saltzman WM . Synthetic DNA delivery systems Nat Biotechnol 2000 18: 33–37
Crystal RG . Transfer of genes to humans: early lessons and obstacles to success Science 1995 270: 404–410
Cristiano RJ . Targeted, nonviral gene delivery for cancer gene therapy Front Biosci 1998 3: D1161–D1170
van de Wetering P, Cherng J-Y, Talsma H, et al . Relation between transfection efficiency and cytotoxicity of poly(2-(dimethylamino)ethyl methacrylate)/plasmid complexes J Controlled Release 1997 49: 59–69
Arigita C, Zuidam NJ, Crommelin DJ, et al . Association and dissociation characteristics of polymer/DNA complexes used for gene delivery Pharm Res 1999 16: 1534–1541
De Smedt SC, Demeester J, Hennink WE . Cationic polymer-based gene delivery systems Pharm Res 2000 17: 113–126
van de Wetering P, Schuurmans-Nieuwenbroek NME, Hennink WE, et al . Comparative transfection studies of human ovarian carcinoma cells in vitro, ex vivo, and in vivo with poly(2-(dimethylamino)ethyl methacrylate)-based polyplexes J Gen Med 1999 1: 156–165
Ruponen M, Yla-Herttuala S, Urtti A . Interactions of polymeric and liposomal gene delivery systems with extracellular glycosaminoglycans: physicochemical and transfection studies Biochim Biophys Acta 1999 1415: 331–341
Zuidam NJ, Posthuma G, de Vries ET, et al . Effects of physicochemical characteristics of poly(2-(dimethylamino)ethyl methacrylate)-based polyplexes on cellular association and internalization J Drug Target 2000 8: 51–66
Martin FJ, Papahadjopoulos D . Irreversible coupling of immunoglobulin fragments to preformed vesicles. An improved method for liposome targeting J Biol Chem 1982 257: 286–288
Bout A, Valerio D, Scholte BJ . In vivo transfer and expression of the lacZ gene in the mouse lung Exp Lung Res 1993 19: 193–202
Hinrichs WL, Schuurmans-Nieuwenbroek NM, van de Wetering P, et al . Thermosensitive polymers as carriers for DNA delivery J Controlled Release 1999 60: 249–259
Hamilton TC, Young RC, McKoy WM, et al . Characterization of a human ovarian carcinoma cell line (NIH:OVCAR-3) with androgen and estrogen receptors Cancer Res 1983 43: 5379–5389
Cherng JY, van de Wetering P, Talsma H, et al . Effect of size and serum proteins on transfection efficiency of poly((dimethylamino)ethyl methacrylate)-plasmid nanoparticles Pharm Res 1996 13: 1038–1042
Levy D, Gulik A, Seigneuret M, et al . Phospholipid vesicle solubilization and reconstitution by detergents. Symmetrical analysis of the two processes using octaethylene glycol mono- n -dodecyl ether Biochemistry 1990 29: 9480–9488
Hansen CB, Kao GY, Moase EH, et al . Attachment of antibodies to sterically stabilized liposomes: evaluation, comparison, and optimization of coupling procedures Biochim Biophys Acta 1995 1239: 133–144
Fiske CH, Subbarow Y . The colorimetric determination of phosphorus J Biol Chem 1925 66: 375–400
Bligh EG, Dyer EJ . A rapid method of total lipid extraction and purification Can J Biochem Physiol 1959 37: 911–917
Katayose S, Kataoka K . Water-soluble polyion complex associates of DNA and poly(ethylene glycol)-poly( L-lysine) block copolymer Bioconjugate Chem 1997 8: 702–707
Edwards DP, Grzyb KT, Dressler LG, et al . Monoclonal antibody identification and characterization of a Mr 43,000 membrane glycoprotein associated with human breast cancer Cancer Res 1986 46: 1306–1317
Langmuir VK, Mendonca HL, Woo DV . Comparisons between two monoclonal antibodies that bind to the same antigen but have differing affinities: uptake kinetics and 125I-antibody therapy efficacy in multicell spheroids Cancer Res 1992 52: 4728–4734
Lee RJ, Huang L . Folate-targeted, anionic liposome-entrapped polylysine-condensed DNA for tumor cell–specific gene transfer J Biol Chem 1996 271: 8481–8487
Lee RJ, Huang L . Lipidic vector systems for gene transfer Crit Rev Ther Drug Carrier Syst 1997 14: 173–206
Haisma HJ, Pinedo HM, Rijswijk A, et al . Tumor-specific gene transfer via an adenoviral vector targeted to the pan-carcinoma antigen EpCAM Gene Ther 1999 6: 1469–1474
Fonseca MJ, Storm G, Hennink WE, et al . Cationic polymeric gene delivery of β-glucuronidase for doxorubicin prodrug therapy J Gen Med 1999 1: 407–414
Acknowledgements
The authors thank Trudy Riool for performing the electron microscopy studies, and Ferry Verbaan for synthesizing the p(DMAEMA) polymers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mastrobattista, E., Kapel, R., Eggenhuisen, M. et al. Lipid-coated polyplexes for targeted gene delivery to ovarian carcinoma cells. Cancer Gene Ther 8, 405–413 (2001). https://doi.org/10.1038/sj.cgt.7700311
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700311
Keywords
This article is cited by
-
Charge shielding effects on gene delivery of polyethylenimine/DNA complexes: PEGylation and phospholipid coating
Journal of Materials Science: Materials in Medicine (2012)
-
DNA vaccines: ready for prime time?
Nature Reviews Genetics (2008)
-
Lipid-based Nanoparticles for Nucleic Acid Delivery
Pharmaceutical Research (2007)
-
Artificial viruses: a nanotechnological approach to gene delivery
Nature Reviews Drug Discovery (2006)
-
A nonviral carrier for targeted gene delivery to tumor cells
Cancer Gene Therapy (2004)