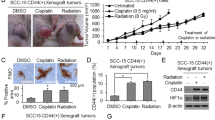
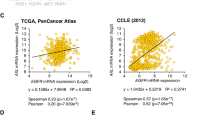
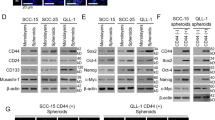
Abstract
The unique characteristic of head and neck squamous cell carcinoma (HNSCC) is that local invasion rather than distant metastasis is the major route for dissemination. Therefore, targeting the locally invasive cancer cells is more important than preventing systemic metastasis in HNSCC and other invasive-predominant cancers. We previously demonstrate a specific mechanism for HNSCC local invasion: the epithelial–mesenchymal transition (EMT) regulator Twist1 represses microRNA let-7i expression, leading to the activation of the small GTPase Rac1 and engendering the mesenchymal-mode movement in three-dimensional (3D) culture. However, targeting the EMT regulator is relatively difficult because of its transcription factor nature and the strategy for confining HNSCC invasion to facilitate local treatment is limited. Imipramine blue (IB) is a newly identified anti-invasive compound that effectively inhibits glioma invasion. Here we demonstrate that in HNSCC cells, a noncytotoxic dose of IB represses mesenchymal-mode migration in two-and-a-half-dimensional/3D culture system. IB suppresses EMT and stemness of HNSCC cells through inhibition of Twist1-mediated let-7i downregulation and Rac1 activation and the EMT signalling. Mechanistically, IB inhibits reactive oxygen species-induced nuclear factor-κB pathway activation. Importantly, IB promotes degradation of the EMT inducer Twist1 by enhancing F-box and leucine-rich repeat protein 14 (FBXL14)-mediated polyubiquitination of Twist1. Together, this study demonstrates the potent anti-invasion and EMT-inhibition effect of IB, suggesting the potential of IB in treating local invasion-predominant cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E . Cancer statistics, 2010. CA Cancer J Clin 2010; 60: 277–300.
Garavello W, Ciardo A, Spreafico R, Gaini RM . Risk factors for distant metastases in head and neck squamous cell carcinoma. Arch Otolaryngol Head Neck Surg 2006; 132: 762–766.
Pentenero M, Gandolfo S, Carrozzo M . Importance of tumor thickness and depth of invasion in nodal involvement and prognosis of oral squamous cell carcinoma: a review of the literature. Head Neck 2005; 27: 1080–1091.
Forastiere AA, Zhang Q, Weber RS, Maor MH, Goepfert H, Pajak TF et al. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol 2013; 31: 845–852.
Lorch JH, Goloubeva O, Haddad RI, Cullen K, Sarlis N, Tishler R et al. Induction chemotherapy with cisplatin and fluorouracil alone or in combination with docetaxel in locally advanced squamous-cell cancer of the head and neck: long-term results of the TAX 324 randomised phase 3 trial. Lancet Oncol 2011; 12: 153–159.
Friedl P . Prespecification and plasticity: shifting mechanisms of cell migration. Curr Opin Cell Biol 2004; 16: 14–23.
Sanz-Moreno V, Gadea G, Ahn J, Paterson H, Marra P, Pinner S et al. Rac activation and inactivation control plasticity of tumor cell movement. Cell 2008; 135: 510–523.
Sanz-Moreno V, Marshall CJ . The plasticity of cytoskeletal dynamics underlying neoplastic cell migration. Curr Opin Cell Biol 2010; 22: 690–696.
Wyckoff JB, Pinner SE, Gschmeissner S, Condeelis JS, Sahai E . ROCK- and myosin-dependent matrix deformation enables protease-independent tumor-cell invasion in vivo. Curr Biol 2006; 16: 1515–1523.
Panková K, Rösel D, Novotný M, Brábek J . The molecular mechanisms of transition between mesenchymal and amoeboid invasiveness in tumor cells. Cell Mol Life Sci 2010; 67: 63–71.
Madsen CD, Sahai E . Cancer dissemination-lessons from leukocytes. Dev Cell 2010; 19: 13–26.
Thiery JP, Acloque H, Huang RY, Nieto MA . Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871–890.
Nieto MA . Epithelial plasticity: a common theme in embryonic and cancer cells. Science 2013; 342: 1234850.
Tsai JH, Yang J . Epithelial-mesenchymal plasticity in carcinoma metastasis. Genes Dev 2013; 27: 2192–2206.
Yang WH, Lan HY, Huang CH, Tai SK, Tzeng CH, Kao SY et al. RAC1 activation mediates Twist1-induced cancer cell migration. Nat Cell Biol 2012; 14: 366–374.
Yang WH, Lan HY, Tai SK, Yang MH . Repression of bone morphogenetic protein 4 by let-7i attenuates mesenchymal migration of head and neck cancer cells. Biochem Biophys Res Commun 2013; 433: 24–30.
Di C, Mattox AK, Harward S, Adamson C . Emerging therapeutic targets and agents for glioblastoma migrating cells. Anticancer Agents Med Chem 2010; 10: 543–555.
Munson JM, Fried L, Rowson SA, Bonner MY, Karumbaiah L, Diaz B et al. Anti-invasive adjuvant therapy with imipramine blue enhances chemotherapeutic efficacy against glioma. Sci Transl Med 2012; 4: 127ra36.
Carlsson SK, Brothers SP, Wahlestedt C . Emerging treatment strategies for glioblastoma multiforme. EMBO Mol Med 2014; 6: 1359–1370.
Newton HB, Rosenblum MK, Walker RW . Extraneural metastases of infratentorial glioblastoma multiforme to the peritoneal cavity. Cancer 1992; 69: 2149–2153.
Evan-Ram S, Yamada KM . Cell migration in 3D matrix. Curr Opin Cell Biol 2005; 17: 524–532.
Slorach EM, Chou J, Werb Z . Zeppo1 is a metastasis promoter that represses E-cadherin expression and regulates p120-catenin isoform expression and localization. Genes Dev 2011; 25: 471–484.
Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY, Yang WH et al. Bmi1 is essential in Twist1-induced epithelial- mesenchymal transition. Nat Cell Biol 2010; 12: 982–992.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Prince ME, Sivanandan R, Kaczorowski A, Wolf GT, Kaplan MJ, Dalerba P et al. Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proc Natl Acad Sci USA 2007; 104: 973–978.
Morgan MJ, Liu ZG . Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res 2011; 21: 103–115.
Li CW, Xia W, Huo L, Lim SO, Wu Y, Hsu JL et al. Epithelial-mesenchymal transition induced by TNF-α requires NF-κB-mediated transcriptional upregulation of Twist1. Cancer Res 2012; 72: 1290–1300.
Cogswell JP, Godlevski MM, Wisely GB, Clay WC, Leesnitzer LM, Ways JP et al. NF-kappa B regulates IL-1 beta transcription through a consensus NF-kappa B binding site and a nonconsensus CRE-like site. J Immunol 1994; 153: 712–723.
Libermann TA, Baltimore D . Activation of interleukin-6 gene expression through the NF-KB transcription factor. Mol Cell Biol 1990; 10: 2327–2334.
Ravid T, Hochstrasser M . Diversity of degradation signals in the ubiquitin–proteasome system. Nat Rev Mol Cell Biol 2008; 9: 679–689.
Lander R, Nordin K, LaBonne C . The F-box protein Ppa is a common regulator of core EMT factors Twist, Snail, Slug, and Sip1. J Cell Biol 2011; 194: 17–25.
Zhong J, Ogura K, Wang Z, Inuzuka H . Degradation of the transcription factor Twist, an oncoprotein that promotes cancer metastasis. Discov Med 2013; 15: 7–15.
Ho MS, Tsai PI, Chien CT . F-box proteins: the key to protein degradation. J Biomed Sci 2006; 13: 181–191.
Viñas-Castells R, Beltran M, Valls G, Gómez I, García JM, Montserrat-Sentís B et al. The hypoxia-controlled FBXL14 ubiquitin ligase targets SNAIL1 for proteasome degradation. J Biol Chem 2010; 285: 3794–3805.
Hsu DS, Wang HJ, Tai SK, Chou CH, Hsieh CH, Chiu PH et al. Acetylation of Snail modulates the cytokinome of cancer cells to enhance the recruitment of macrophages. Cancer Cell 2014; 26: 534–548.
Acknowledgements
We thank Professor Víctor M Díaz (Universitat Pompeu Fabra, Barcelona, Spain) for the generous gifts of pcDNA3-myc-FBXL14 and pcDNA3-FBXL14ΔF plasmids. We thank Professor Kuo-Wei Chang (National Yang-Ming University, Taiwan) for providing OECM-1 cell line. This work was supported by Ministry of Science and Technology (103-2321-B-010-019, 103-2314-B-010-034, 103-2633-H-010-001, and 103-2314-B-010-035 to M-HY), Taipei Veterans General Hospital (V104-E8-001 to M-HY), a grant from Ministry of Education, Aim for the Top University Plan (to M-HY) and a grant from the Ministry of Health and Welfare, Center of Excellence for Cancer Research (MOHW104-TDU-B-211-124-001 to M-HY).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Emory University holds the intellectual property to imipramine blue and that Jack L Arbiser is the inventor. Emory University has licensed the intellectual property to ABBY Therapeutics.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Yang, WH., Su, YH., Hsu, WH. et al. Imipramine blue halts head and neck cancer invasion through promoting F-box and leucine-rich repeat protein 14-mediated Twist1 degradation. Oncogene 35, 2287–2298 (2016). https://doi.org/10.1038/onc.2015.291
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.291
This article is cited by
-
Tight junction protein 1 promotes vasculature remodeling via regulating USP2/TWIST1 in bladder cancer
Oncogene (2022)
-
Emerging role of F-box proteins in the regulation of epithelial-mesenchymal transition and stem cells in human cancers
Stem Cell Research & Therapy (2019)
-
Imipramine blue sensitively and selectively targets FLT3-ITD positive acute myeloid leukemia cells
Scientific Reports (2017)