Abstract
This Review describes the work conducted by the TRANSBIG consortium in the development of the MINDACT (Microarray In Node negative Disease may Avoid ChemoTherapy) trial. The goal of the trial is to provide definitive evidence regarding the clinical relevance of the 70-gene prognosis signature, and to assess the performance of this signature compared with that of traditional prognostic indicators for assigning adjuvant chemotherapy to patients with node-negative breast cancer. We outline the background work and the key questions in node-negative early-stage breast cancer, and then focus on the MINDACT trial design and statistical considerations. The challenges inherent in this trial in terms of logistics, implementation and interpretation of the results are also discussed. We hope that this article will trigger further discussion about the difficulties of setting up and analyzing trials aimed at establishing the worth of new methods for better selection of patients for cancer treatment.
Key Points
-
In the past 20 years, little progress has been made regarding new prognostic markers that can assist oncologists in treatment decision-making for node-negative early-stage breast cancer
-
By using gene-expression profiling, the Netherlands Cancer Institute developed a 70-gene prognostic signature for node-negative breast cancer that has now been commercialized. This signature was developed as a dichotomous risk classification for the endpoint of distant metastasis within 5 years and has been retrospectively validated in two series of patients
-
The MINDACT clinical trial will prospectively validate the 70-gene prognostic signature
-
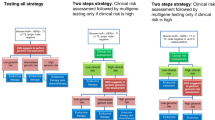
Validation of the risk of distant relapse will be assessed through clinicopathologic criteria, using Adjuvant! Online and MammaPrint® (Agendia). In case of discordant results, patients will be randomized to treatment decision-making based on clinicopathologic risk or genomic risk
-
Two further randomizations will be possible in the MINDACT trial: randomization to chemotherapy, based on anthracycline-based regimens, and randomization to endocrine therapy, based on the outcome of 2 years of tamoxifen followed by 5 years of letrozole, compared with 7 years of letrozole
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
van de Vijver MJ et al. (2002) A gene-expression signature as a predictor of survival in breast cancer. N Eng J Med 447: 1999–2009
Hutchins L et al. (1998) CMF versus CAF with and without Tamoxifen in high-risk node-negative breast cancer patients and a natural history follow-up study in low-risk node-negative patients: First results of Intergroup trial INT 0102 [abstract #2]. Proc Am Soc Clin Oncol 17: a1
McShane LM et al. (2005) REporting recommendations for tumour MARKer prognostic studies. Nat Clin Pract Oncol 2: 416–422
[No authors listed] (2001) The National Institutes of Health Consensus Development Conference: adjuvant therapy for breast cancer. Bethesda, Maryland, USA. November 1–3, 2000. Proceedings. J Natl Cancer Inst Monogr 30: 1–152
Goldhirsch A et al. (2005) Meeting highlights: international expert consensus on the primary therapy of early breast cancer 2005. Ann Oncol 16: 1569–1583
D'Eredita G et al. (2001) Prognostic factors in breast cancer: the predictive value of the Nottingham prognostic index in patients with a long-term follow-up that were treated in a single institution. Eur J Cancer 37: 591–596
van 't Veer LJ et al. (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415: 530–536
van de Vijver MJ et al. (2002) A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 347: 1999–2009
Piccart MJ et al. (2004) Multi-center external validation study of the Amsterdam 70-gene prognostic signature in node negative untreated breast cancer: are the results still outperforming the clinical-pathological criteria? [abstract] Breast Cancer Res Treat 88 (Suppl 1): aS38
Cardoso F et al. (2005) Multi-centre independent validation of a gene prognostic signature for patients with node-negative breast cancer (BC)—final results [abstract]. Breast Cancer Res Treat 95 (Suppl 1): aS301
Adjuvant! Online [http://www.adjuvantonline.com]
Ravdin PM et al. (2001) Computer program to assist in making decisions about adjuvant therapy for women with early breast cancer. J Clin Oncol 19: 980–991
Olivotto IA et al. (2005) Population-based validation of the prognostic model ADJUVANT! for early breast cancer. J Clin Oncol 23: 2716–2725
Simon R et al. (2003) Pitfalls in the use of DNA microarray data for diagnostic and prognostic classification. J Natl Cancer Inst 95: 14–18
Dudoit S et al. (2002) Comparison of discrimination methods for the classification of tumors using gene expression data. JASA 97: 77–87
De Vita Jr VT et al. (Eds; 2006) Cancer. Principles and Practice of Oncology. Philadelphia: Lippincott Williams & Wilkins
Cardoso F et al. (2005) Multi centre independent validation of a gene prognostic signature for patients with node negative breast cancer-final results [abstract]. Breast Cancer Res Treat 94 (Suppl 1): A301
Buyse M et al. (2006) Validation and clinical utility of a 70-gene prognostic signature for women with node-negative breast cancer. J Natl Cancer Inst 98: 1183–1192
Early Breast Cancer Trialists' Collaborative Group (EBCTCG) (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365: 1687–1717
Ross JS et al. (2003) The Her-2/neu gene and protein in breast cancer 2003: biomarker and target of therapy. Oncologist 8: 307–325
Smith IE et al. (2005) Neoadjuvant treatment of postmenopausal breast cancer with anastrozole, tamoxifen or both in combination: the Immmediate Preoperative Aastrozole, Tamoxifen, or Combined with Tamoxifen (IMPACT) multicenter double-blind randomized trial. J Clin Oncol 23: 5108–5116
[No authors listed] (1999) ICH Harmonised Tripartite Guideline: statistical principles for clinical trials. International Conference on Harmonisation E9 Expert Working Group. Stat Med 18: 1905–1942
O'Shaughnessy J et al. (2002) Superior survival with capecitabine plus docetaxel combination therapy in anthracycline-pretreated patients with advanced breast cancer: phase III trial results. J Clin Oncol 20: 2812–2823
Jones S et al. (2003) Three year results of a prospective randomized trial of adjuvant chemotherapy for patients (pts) with stage I-III operable, invasive breast cancer comparing 4 courses of doxorubicin/cyclophosphamide (AC) to 4 courses of docetaxel/ cyclophosphamide (TC) [abstract]. Proc Am Soc Clin Oncol a59
Goss PE et al. (2003) A randomized trial of letrozole in postmenopausal women after five years of tamoxifen therapy for early-stage breast cancer. N Engl J Med 349: 1793–1802
Coombes RC et al. (2004) A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 350: 1081–1092
Thurlimann BJ et al. (2005) BIG 1–98: randomized double-blind phase III study to evaluate letrozole (L) vs. tamoxifen (T) as adjuvant endocrine therapy for postmenopausal women with receptor-positive breast cancer [abstract #511]. Proc Am Soc Clin Oncol 23: 16S
Bonnefoi H et al. (2003) Locally advanced/inflammatory breast cancers treated with intensive epirubicin-based neoadjuvant chemotherapy: are there molecular markers in the primary tumour that predict for 5-year clinical outcome? Ann Oncol 14: 406–413
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L van't Veer is a named inventor on a patent application for the 70-gene signature and reports being employed by Agendia B.V., the commercial entity that markets the 70-gene signature as Mammaprint®. The other authors declared they have no competing interests.
Rights and permissions
About this article
Cite this article
Bogaerts, J., Cardoso, F., Buyse, M. et al. Gene signature evaluation as a prognostic tool: challenges in the design of the MINDACT trial. Nat Rev Clin Oncol 3, 540–551 (2006). https://doi.org/10.1038/ncponc0591
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc0591
This article is cited by
-
Controlling technical variation amongst 6693 patient microarrays of the randomized MINDACT trial
Communications Biology (2020)
-
Progress in adjuvant systemic therapy for breast cancer
Nature Reviews Clinical Oncology (2019)
-
The PROMISe to increase precision in adjuvant therapy for early breast cancer: To “Type” or to “Print”?
npj Breast Cancer (2018)
-
Patterns in target-directed breast cancer research
SpringerPlus (2016)
-
Multi-gene classifiers for prediction of recurrence in breast cancer patients
Breast Cancer (2016)