Abstract
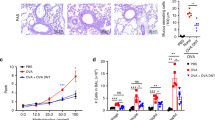
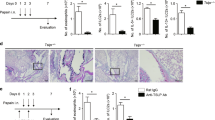
Programmed death one homolog (PD-1H) is a cell surface molecule of the B7/CD28 immune modulatory gene family. Although PD-1H has been shown to function as a coinhibitory receptor on T cells to limit naive T-cell activation and proliferation, its role in the regulation of the T-cell response to allergens is unknown. We report here that genetic ablation or blockade of PD-1H drastically promotes pulmonary inflammation with massive accumulation of eosinophils in a mouse model of experimental asthma, indicating a suppressive function of PD-1H in allergic inflammation. The loss of PD-1H led to elevated production of both innate cytokines (IL-6, MCP-1 and TNFα) and Th2 cytokines (IL-5 and IL-13) in the lung, indicating a critical role of PD-1H in suppressing the production of airway inflammatory cytokines. In addition, the loss of PD-1H also impaired the expansion of systemic and pulmonary regulatory T cells during asthma induction. These findings support a critical role of intrinsic PD-1H in the regulation of inflammatory responses to allergens. Finally, we showed that treatment with a PD-1H agonistic monoclonal antibody reduced the severity of asthma, which was accompanied by suppressed lung inflammation. Our findings support PD-1H as a potential target and suggest a possible strategy for the treatment of allergic asthma in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
23 May 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41423-023-01038-5
References
Flies DB, Higuchi T, Chen L. Mechanistic assessment of PD-1H coinhibitory receptor-induced T cell tolerance to allogeneic antigens. J Immunol 2015; 194: 5294–5304.
Flies DB, Wang S, Xu H, Chen L. Cutting edge: a monoclonal antibody specific for the programmed death one homolog prevents graft-versus-host disease in mouse models. J Immunol 2011; 187: 1537–1541.
Flies DB, Han X, Higuchi T, Zheng L, Sun J, Ye JJ et al. Coinhibitory receptor PD-1H preferentially suppresses CD4+ T cell-mediated immunity. J Clin Invest 2014; 124: 1966–1975.
Wang L, Rubinstein R, Lines JL, Wasiuk A, Ahonen C, Guo Y et al. VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J Exp Med 2011; 208: 577–592.
Yoon KW, Byun S, Kwon E, Hwang SY, Chu K, Hiraki M et al. Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53. Science 2015; 349: 1261669.
Ceeraz S, Sergent PA, Plummer SF, Schned AR, Pechenick D, Burns CM et al. VISTA deficiency accelerates the development of fatal murine lupus nephritis. Arthritis Rheumatol, 2016 epubdate ahead of print.
Wang L, Le Mercier I, Putra J, Chen W, Liu J, Schenk AD et al. Disruption of the immune-checkpoint VISTA gene imparts a proinflammatory phenotype with predisposition to the development of autoimmunity. Proc Natl Acad Sci USA 2014; 111: 14846–14851.
Lambrecht BN, Hammad H. Allergens and the airway epithelium response: gateway to allergic sensitization. J Allergy Clin Immunol 2014; 134: 499–507.
Veres TZ, Kopcsanyi T, van Panhuys N, Gerner MY, Liu Z, Rantakari P et al. Allergen-induced CD4+ T cell cytokine production within airway mucosal dendritic cell-T cell clusters drives the local recruitment of myeloid effector cells. J Immunol 2017; 198: 895–907.
May RD, Fung M. Strategies targeting the IL-4/IL-13 axes in disease. Cytokine 2015; 75: 89–116.
Ogasawara K, Yoshinaga SK, Lanier LL. Inducible costimulator costimulates cytotoxic activity and IFN-gamma production in activated murine NK cells. J Immunol 2002; 169: 3676–3685.
Paczesny S, Choi SW, Ferrara JL. Acute graft-versus-host disease:new treatment strategies. Curr Opin Hematol 2009; 16: 427–436.
Azuma T, Zhu G, Xu H, Rietz AC, Drake CG, Matteson EL et al. Potential role of decoy B7-H4 in the pathogenesis of rheumatoid arthritis: a mouse model informed by clinical data. PLoS Med 2009; 6: e1000166.
Zhang J, Dong Z, Zhou R, Luo D, Wei H, Tian Z. Isolation of lymphocytes and their innate immune characterizations from liver, intestine, lung and uterus. Cell Mol Immunol 2005; 2: 271–280.
Miller RA, Garcia G, Kirk CJ, Witkowski JM. Early activation defects in T lymphocytes from aged mice. Immunol Rev 1997; 160: 79–90.
Wynn TA. IL-13 effector functions. Annu Rev Immunol 2003; 21: 425–456.
Zhu Z, Homer RJ, Wang Z, Chen Q, Geba GP, Wang J et al. Pulmonary expression of interleukin-13 causes inflammation, mucus hypersecretion, subepithelial fibrosis, physiologic abnormalities, and eotaxin production. J Clin Invest 1999; 103: 779–788.
Lin JX, Migone TS, Tsang M, Friedmann M, Weatherbee JA, Zhou L et al. The role of shared receptor motifs and common Stat proteins in the generation of cytokine pleiotropy and redundancy by IL-2, IL-4, IL-7, IL-13, and IL-15. Immunity 1995; 2: 331–339.
Zurawski SM, Chomarat P, Djossou O, Bidaud C, McKenzie AN, Miossec P et al. The primary binding subunit of the human interleukin-4 receptor is also a component of the interleukin-13 receptor. J Biol Chem 1995; 270: 13869–13878.
Chomarat P, Banchereau J. Interleukin-4 and interleukin-13: their similarities and discrepancies. Int Rev Immunol 1998; 17: 1–52.
Duvernelle C, Freund V, Frossard N. Transforming growth factor-beta and its role in asthma. Pulm Pharmacol Ther 2003; 16: 181–196.
Rubtsov YP, Rasmussen JP, Chi EY, Fontenot J, Castelli L, Ye X et al. RegulatoryT cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 2008; 28: 546–558.
Palomares O, Yaman G, Azkur AK, Akkoc T, Akdis M, Akdis CA. Role of Treg in immune regulation of allergic diseases. Eur J Immunol 2010; 40: 1232–1240.
Acknowledgements
This work is supported in part by the 985 project grant from Sun Yat-Sen University, Guangdong Province Innovative Research Program Project 2011Y035, PRC; National Institutes of Health grants P50 CA196530 and P30 CA016359; and an endowment from the United Technologies Corporation, USA.
Author information
Authors and Affiliations
Contributions
HL and LC developed the concept, designed the experiments and wrote the manuscript. XL, LH and LL advised, and/or performed the experiments.
Corresponding author
Ethics declarations
Conflict of interest
LC serves as an advisor/board member for Pfizer, AstraZeneca, NextCure, GenomiCare and Vcanbio, and receives research support from Boehringer Ingelheim, Pfizer and NextCure. LC also serves as an uncompensated adjunct faculty member of Sun Yat-Sen University. The remaining authors declare no conflict of interest.
Additional information
The original online version of this article was revised: In the Acknowledgement section of this article, NIH grants numbered P50 CA196530 and P30 CA016359 were incorrectly attributed to author Lieping Chen and should not have been cited. The original article has been corrected.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, H., Li, X., Hu, L. et al. A crucial role of the PD-1H coinhibitory receptor in suppressing experimental asthma. Cell Mol Immunol 15, 838–845 (2018). https://doi.org/10.1038/cmi.2017.16
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2017.16
Keywords
This article is cited by
-
Repositioning baloxavir marboxil as VISTA agonist that ameliorates experimental asthma
Cell Biology and Toxicology (2024)
-
VISTA and its ligands: the next generation of promising therapeutic targets in immunotherapy
Cancer Cell International (2023)
-
VISTA regulates microglia homeostasis and myelin phagocytosis, and is associated with MS lesion pathology
Acta Neuropathologica Communications (2021)
-
Exploring the VISTA of microglia: immune checkpoints in CNS inflammation
Journal of Molecular Medicine (2020)
-
The structure, expression, and multifaceted role of immune-checkpoint protein VISTA as a critical regulator of anti-tumor immunity, autoimmunity, and inflammation
Cellular & Molecular Immunology (2018)