Abstract
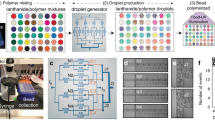
Bead-based assays on very large numbers of molecules in gene expression studies, drug screening and clinical diagnostics1, require the encoding of each of the microspheres according to the particular ligand bound to its surface2,3. This allows mixing the uniquely encoded microspheres and subjecting them to an assay simultaneously. When a particular microsphere gives a positive reaction, the substance on its surface can be identified by reading the code. Previously reported techniques for colour encoding polymer microspheres4,5,6,7,8,9,10 only allow for a limited number of unique codes. Graphical encoding methods use metallic particles11,12, which are rather uncommon in screening applications. Here, we demonstrate a new approach to encode polymer microspheres that are commonly used in screening applications, such as polystyrene microspheres, with a method that provides a virtually unlimited number of unique codes. Patterns can be written in fluorescently dyed microspheres by 'spatial selective photobleaching' and can be identified by confocal microscopy. Such encoded microparticles can find broad application in the collection and analysis of genetic information, high-throughput screening, medical diagnostics and combinatorial chemistry, and can also be used for labelling of consumer goods or as security labels to prevent counterfeiting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Meza, M.B. Bead-based HTS applications in drug discovery. Drug Discov. Today 1 (HTS Suppl.), 38–41 (2000).
Braeckmans, K. et al. Carrying the code. Mod. Drug Disc. (in the press).
Braeckmans, K., De Smedt, S.C., Leblans, M., Pauwels, R. & Demeester, J. Encoding microcarriers: present and future technologies. Nat. Rev. Drug Disc. 1, 447–456 (2002).
Egner, B.J. et al. Tagging in combinatorial chemistry: the use of coloured and fluorescent beads. Chem. Commun. 8, 735–736 (1997).
Kettman, J.R., Davies, T., Chandler, D., Oliver, K.G. & Fulton, R.J. Classification and properties of 64 multiplexed microsphere sets. Cytometry 33, 234–243 (1998).
Walt, D.R. Bead-based fiber-optic arrays. Science 287, 451–452 (2000).
Trau, M. & Battersby, B.J. Novel colloidal materials for high-throughput screening applications in drug discovery and genomics. Adv. Mater. 13, 975–979 (2001).
Keij, J.F. & Steinkamp, J.A. Flow cytometric characterization and classification of multiple dual-colour fluorescent microspheres using fluorescence lifetime. Cytometry 33, 318–323 (1998).
Kürner, J.M., Klimant, I., Krause, C., Pringsheim, E. & Wolfbeis, O.S. A new type of phosphorescent nanospheres for use in advanced time-resolved multiplexed bioassays. Anal. Biochem. 297, 32–41 (2001).
Han, M., Gao, X.H., Su, J.Z. & Nie S. Quantum-dot-tagged microbeads for multiplexed optical coding of biomolecules. Nat. Biotechnol. 19, 631–635 (2001).
Dames, A., England, J. & Colby, E. Bio-assay technique. World patent 00/16893 (2000).
Nicewarner-Peña, S.R. et al. Submicrometer metallic barcodes. Science 294, 137–141 (2001).
Pawley, J.B. Handbook of Biological Confocal Microscopy 2nd edn (Plenum, New York, 1995).
Hell, S., Reiner, G., Cremer, C. & Stelzer, E.H.K. Aberrations in confocal fluorescence microscopy induced by mismatches in refractive index. J. Microsc. 169, 391–405 (1993).
Wang, N., Butler, J.P. & Ingber, D.E. Mechanotransduction across the cell-surface and through the cytoskeleton. Science 260, 1124–1127 (1993).
Prabhakar, U., Eirikis, E. & Davis, H.M. Simultaneous quantification of proinflammetary cytokines in human plasma using the LabMAP (TM) assay. J. Immunol. Methods 260, 207–218 (2002).
Martins, T.B. Development of internal controls for the Luminex instrument as part of a multiplex seven-analyte viral respiratory antibody profile. Clin. Diagn. Lab. Immun. 9, 41–45 (2002).
Ye, F. et al. Fluorescent microsphere-based readout technology for multiplexed human single nucleotide polymorphism analysis and bacterial identification. Hum. Mutat. 17, 305–316 (2001).
Taylor, J.D. et al. Flow cytometric platform for high-throughput single nucleotide polymorphism analysis. Biotechniques 30, 661–675 (2001).
Yang, L., Tran, D.K. & Wang, X. BADGE, BeadsArray for the detection of gene expression, a high-throughput diagnostic bioassay. Genome Res. 11, 1888–1898 (2001).
Dunbar, S.A. & Jacobson, J.W. Application of the Luminex LabMAP in rapid screening for mutations in the cystic fibrosis transmembrane conductance regulator gene: A pilot study. Clin. Chem. 46, 1498–1500 (2000).
Vignali, D.A.A. Multiplexed particle-based flow cytometric assays. J. Immunol. Methods 243, 243–255 (2000).
Gordon, R.F. & MCDade, R.L. Multiplexed quantification of human IgG, IgA, and IgM with the FlowMetrix(TM) system. Clin. Chem. 43, 1799–1801 (1997).
WalkerPeach, C.R., Smith, P.L., DuBois, D.B. & Fulton, R.J. A novel rapid multiplexed assay for herpes simplex virus DNA using the FlowMetrix(TM) cytometric microsphere technology. Clin. Chem. 43, 21–21 (1997).
Smith, P.L., WalkerPeach, C.R., Fulton, R.J. & DuBois, D.B. A rapid, sensitive, multiplexed assay for detection of viral nucleic acids using the FlowMetrix system. Clin. Chem. 44, 2054–2056 (1998).
Oliver, K.G., Kettman, J.R. & Fulton, R.J. Multiplexed analysis of human cytokines by use of the FlowMetrix system. Clin. Chem. 44, 2057–2060 (1998).
Bellisario, R., Colinas, R.J. & Pass, K.A. Simultaneous measurement of thyroxine and thyrotropin from newborn dried blood-spot specimens using a multiplexed fluorescent microsphere immunoassay. Clin. Chem. 46, 1422–1424 (2000).
Ferguson, J.A., Steemers, F.J. & Walt, D.R. High-density fiber-optic DNA random microsphere array. Anal. Chem. 72, 5618–5624 (2000).
Ferguson, J.A., Boles, T.C., Adams, C.P. & Walt, D.R. A fiber-optic DNA biosensor microarray for the analysis of gene expression. Nat. Biotechnol. 14, 1681–1684 (1996).
Wedekind, P., Kubitscheck, U., Heinrich, O. & Peters, R. Line-scanning microphotolysis for diffraction-limited measurements of lateral diffusion. Biophys. J. 71, 1621–1632 (1996).
Acknowledgements
The financial support of the IWT and Tibotec is acknowledged with gratitude. Ghent University (BOF, the FWO-Vlaanderen ('krediet aan navorsers') and the IWT (Industrial Basic Research Project) are acknowledged for their support through instrumentation credits. We also wish to thank Emmanuel Gustin for useful discussions about the code design, Tom Meyvis for his help with the bleaching experiments and Patrick Van Oostveldt for many fruitful discussions about confocal microscopy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Braeckmans, K., De Smedt, S., Roelant, C. et al. Encoding microcarriers by spatial selective photobleaching. Nature Mater 2, 169–173 (2003). https://doi.org/10.1038/nmat828
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat828
This article is cited by
-
Construction of anti-counterfeiting pattern on the cellulose film by in-situ regulation strategies
Cellulose (2022)
-
Microsphere-based interferometric optical probe
Nature Communications (2018)
-
Direct Identification of On-Bead Peptides Using Surface-Enhanced Raman Spectroscopic Barcoding System for High-Throughput Bioanalysis
Scientific Reports (2015)
-
One-step pipetting and assembly of encoded chemical-laden microparticles for high-throughput multiplexed bioassays
Nature Communications (2014)
-
Universal process-inert encoding architecture for polymer microparticles
Nature Materials (2014)