Abstract
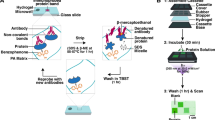
The protein microarray is a crucial biomaterial for the rapid and high-throughput assay of many biological events where proteins are involved. In contrast to the DNA microarray, it has not been sufficiently established because of protein instability under the conventional dry conditions. Here we report a novel semi-wet peptide/protein microarray using a supramolecular hydrogel composed of glycosylated amino acetate. The spontaneous gel-formation and amphiphilic properties of this supramolecular hydrogel have been applied to a new type of peptide/protein gel array that is compatible with enzyme assays. Aqueous cavities created in the gel matrix are a suitable semi-wet reaction medium for enzymes, whereas the hydrophobic domains of the fibre are useful as a unique site for monitoring the reaction. This array system overcomes several drawbacks of conventional protein chips, and thus can have potential applications in pharmaceutical research and diagnosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Shena, M., Shalon, D., Davis, R.W. & Brown, P.O. Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science 270, 467–470 (1995).
Winzeler, E.A. Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285, 901–906 (1999).
The chipping forecast. Nature Genet. 21 (suppl.), 1–60 (1999).
Macbeath, G. & Schreiber, S.L. Printing proteins as microarrays for high-throughput function determination. Science 289, 1760–1763 (2000).
Zhu, H. et al. Analysis of yeast protein kinases using protein chips. Nature Genet. 26, 283–289 (2000).
Zhu, H. et al. Global analysis of protein activities using proteome chips. Science 293, 2101–2105 (2001).
Arenkov, P. et al. Protein microchips: use for immunoassay and enzymatic reactions. Anal. Biochem. 278, 123–131 (2000).
Schild, H.G. Poly (N-isopropylacrylamide): experiment, theory and application. Prog. Polym. Sci. 17, 163–249 (1992).
Pelton, R. Temperature-sensitive aqueous microgels. Adv. Colloid Interface Sci. 85, 1–33 (2000).
Lee, K.Y. & Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 101, 1869–1879 (2001).
Lendlein, A. & Kelch, S. Shape-memory polymers. Angew. Chem. Int. Edn 41, 2034–2057 (2002).
Terech, P. & Weiss, R.G. Low molecular mass gelators of organic liquids and the properties of their gels. Chem. Rev. 97, 3133–3159 (1997).
Hanabusa, K., Yamada, M., Kimura, M. & Shirai, H. Prominent gelation and chiral aggregation of alkylamides derived from trans-1,2-diaminocyclohexane. Angew. Chem. Int. Edn Engl. 35, 1949–1951 (1996).
Murata, K. et al. Thermal and light control of the sol-gel phase transition in cholesterol-based organic gels. Novel helical aggregation modes as detected by circular dichroism and electron microscopic observation. J. Am. Chem. Soc. 116, 6665–6676 (1994).
Hamachi, I., Kiyonaka, S. & Shinkai, S. Solid-phase lipid synthesis (SPLS)-2: incidental discovery of organogelators based on artificial glycolipids. Tetrahedron Lett. 42, 6141–6145 (2001).
Kobayashi, H. et al. Molecular design of super hydrogelators: understanding the gelation process of azobenzene-based sugar derivatives in water. Org. Lett. 4, 1423–1427 (2002).
Jung, J.-H. et al. Self-assembly of a sugar-based gelator in water: its remarkable diversity in gelation ability and aggregate structure. Langmuir 17, 7229–7234 (2001).
Menger, F.M. & Caran, K.L. Anatomy of a gel: amino acid derivatives that rigidify water at submilimolar concentrations. J. Am. Chem. Soc. 122, 11679–11690 (2000).
Maitra, U., Mukhopadhyay, S., Sarkar, A., Rao, P. & Indi, S.S. Hydrophobic pockets in a nonpolymeric aqueous gel: observation of such a gelation process by color change. Angew. Chem. Int. Edn 40, 2281–2283 (2001).
Estroff, L.A. & Hamilton, A.D. Effective gelation of water using a series of bis-urea dicarboxylic acids. Angew. Chem. Int. Edn 39, 3447–3449 (2000).
Bhattacharya, S. & Acharya, S.N.G. Pronounced hydrogel formation by the self-assembled aggregates of N-alkyl dissacharide amphiphiles. Chem. Mater. 11, 3504–3511 (1999).
Oda, R., Huc, I. & Candau, S.J. Gemini surfactant as new, low molecular weight gelators of organic solvents and water. Angew. Chem. Int. Edn 37, 2689–2691 (1998).
Makarevic, J. et al. Bis(amino acid) oxalyl amides as ambidextrous gelators of water and organic solvents: supramolecular gels with temperature dependent assembly/dissolution equilibrium. Chem. Eur. J. 7, 3328–3341 (2001).
Kiyonaka, S., Sugiyasu, K., Shinkai, S. & Hamachi, I. First thermally responsive supramolecular polymer based on glycosylated amino acid. J. Am. Chem. Soc. 124, 10954–10955 (2002).
Kiyonaka, S., Shinkai, S. & Hamachi, I. Combinatorial library of low molecular-weight organo- and hydrogelators based on glycosylated amino acid derivatives by solid-phase synthesis. Chem. Eur. J. 9, 976–984 (2003).
Kiyonaka, S., Zhou, S.-L. & Hamachi, I. pH-responsive phase transition of supramolecular hydrogel consisting of glycosylated amino acetate and carboxylic acid derivative. Supramol. Chem. 15, 521–528 (2003).
Hamachi, I., Kiyonaka, S. & Shinkai, S. Solid-phase lipid synthesis (SPLS) for construction of an artificial glycolipid library. Chem. Commun. 1281–1282 (2000).
Kunitake, T. Synthetic bilayer membranes: molecular design, self-organization, and application. Angew. Chem. Int. Edn Engl. 31, 709–726 (1992).
Stryer, L. The interaction of a naphthalene dye with apomyoglobin and apohemoglobin. A fluorescent probe of nonpolar binding sites. J. Mol. Biol. 13, 482–495 (1965).
Surewicz, W.K., Mantsch, H.H. & Chapman, D. Determination of protein secondary structure by fourier transform infrared spectroscopy: a critical assessment. Biochemistry 32, 389–394 (1993).
Masaki, T., Tanabe, M., Nakamura, K. & Soejima, M. Studies on a new proteilytic-enzyme from achromobactor-lyticus M497-1. 1. purification and some enzymatic-properties. Biochim. Biophys. Acta 660, 44–50 (1981).
Masaki, T., Fujihashi, T., Nakamura, K. & Soejima, M. Studies on a new proteilytic-enzyme from achromobactor-lyticus M497-1. 2. specificity and inhibitionstudies of achromobactor protein-1. Biochim. Biophys. Acta 660, 51–55 (1981).
Drapeau, G.R., Houmard, J. & Boily, Y. Purification and properties of an extracellular protease of staphylococcus aureus. J. Biol. Chem. 247, 6720–6726 (1972).
Lottenberg, R., Christensen, U., Jackson, C.M. & Coleman, P.L. Assay of coagulation proteases using peptide chromogenic and fluorogenic substrates. Meth. Enzymol. 80, 341–361 (1981).
Matayoshi, E.D., Wang, G.T., Krafft, G.A. & Erickson, J. Novel fluorogenic substrates for assaying retroviral proteases by resonance energy-transfer. Science 247, 954–958 (1990).
Collier, C.P. et al. [2]catenane-based solid state electronically reconfigurable switch. Science 289, 1172–1175 (2000).
Aggeli, A. et al. Responsive gels formed by the spontaneous self-assembly of peptides into polymeric beta-sheet tapes. Nature 386, 259–262 (1997).
Holmes, T.C. et al. Extensive neurite outgrowth and active synapse formation on self-assembling peptide scaffolds. Proc. Natl Acad. Sci. USA 97, 6728–6733 (2000).
Collier, J.H. et al. Thermally and photochemically triggered self-assembly of peptide hydrogels. J. Am. Chem. Soc. 123, 9463–9464 (2001).
Schneider, J.P. et al. Responsive hydrogels from the intramolecular folding and self-assembly of a designed peptide. J. Am. Chem. Soc. 124, 15030–15037 (2002).
Kisiday, J. et al. Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair. Proc. Natl Acad. Sci. USA 99, 9996–10001 (2002).
Petka, W.A., Harden, J.L., McGrath, K.P., Wirtz, D. & Tirrell, D.A. Reversible hydrogels from self-assembling artificial proteins. Science 281, 389–392 (1998).
Welsh, E.R. & Tirrell, D.A. Engineering the extracellular matrix: A novel approach to polymeric biomaterials. I. Control of the physical properties of artificial protein matrices designed to support adhesion of vascular endothelial cells. Biomacromolecules 1, 23–30 (2000).
Nowak, A.P. et al. Rapidly recovering hydrogel scaffolds from self-assembling diblock copolypeptide amphiphiles. Nature 417, 424–428 (2002).
Altomare, A., Cascarano, G., Giacovazzo, C. & Guagliardi, A. Completion and refinement of crystal structures with SIR92. J. Appl. Cryst. 26, 343–350 (1993).
Acknowledgements
This research is partially supported by JST (PRESTO, Synthesis and Control) and a Grant-in-Aid for the 21st COE Research Project on “Functional Innovation of Molecular Informatics” from the Ministry of Education, Science, Sports and Culture of Japan, and Tokuyama Science Foundation and Kurata Science Foundation. S.K. is a postdoctoral fellow of the Japan Society for the Promotion of Science (JSPS). We are grateful to H. Ueyama (Kyushu university) for valuable comments on construction of the supramolecular gel microarray.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information, Fig. S1
Supplementary Information, Fig. S2 (PDF 205 kb)
Supplementary Information, Fig. S3
Supplementary Information, Table S1
Rights and permissions
About this article
Cite this article
Kiyonaka, S., Sada, K., Yoshimura, I. et al. Semi-wet peptide/protein array using supramolecular hydrogel. Nature Mater 3, 58–64 (2004). https://doi.org/10.1038/nmat1034
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat1034
This article is cited by
-
Four distinct network patterns of supramolecular/polymer composite hydrogels controlled by formation kinetics and interfiber interactions
Nature Communications (2023)
-
Fluorescence microscopic visualization of functionalized hydrogels
NPG Asia Materials (2022)
-
A printable hydrogel microarray for drug screening avoids false positives associated with promiscuous aggregating inhibitors
Nature Communications (2018)
-
A bioinspired dual-crosslinked tough silk protein hydrogel as a protective biocatalytic matrix for carbon sequestration
NPG Asia Materials (2017)
-
Sulfated glycopeptide nanostructures for multipotent protein activation
Nature Nanotechnology (2017)