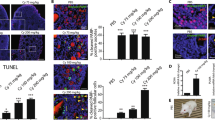
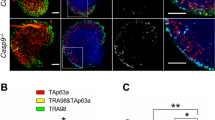
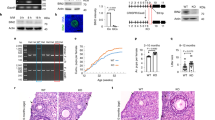
Abstract
Female sterility resulting from oocyte destruction is an unfortunate, and in many cases inevitable, consequence of chemotherapy. We show that unfertilized mouse oocytes exposed to therapeutic levels of the antitumor drug, doxorubicin (DXR), undergo apoptosis; however, fertilized oocytes do not initiate apoptosis, but enter cell-cycle arrest, when treated with DXR. Apoptosis induced by DXR in oocytes is blocked by sphingosine-1-phosphate, an inhibitor of ceramide-promoted cell death. Oocytes from Bax-deficient, but not p53-null, female mice display complete resistance to DXR-induced apoptosis in vivo and in vitro. Pretreatment of oocytes with a specific peptide inhibitor of caspases also abrogates the apoptotic response to DXR. These findings indicate that oocyte destruction caused by chemotherapy can be prevented by manipulation of apoptosis-associated signaling pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Waxman, J. Chemotherapy and the adult gonad: A review. J. R. Soc. Med. 76, 144–148 (1983).
Familiari, C. et al. Ultrastructure of human ovarian primordial follicles after combination chemotherapy for Hodgkin's disease. Hum. Reprod. 8, 2080–2087 (1993).
Ried, H.L., Jaffe, N. Rediation-induced changes in long-term survivors of childhood cancer after treatment with radiation therapy. Semin. Roentgenol. 29, 6–14 (1994).
Reichman, B.S. & Green, K.B. Breast cancer in young women: Effect of chemotherapy on ovarian function, fertility and birth defects. Monogr. Natl. Cancer Inst. 16, 125–129 (1994).
Gougeon, A. Regulation of ovarian follicular development in primates: Facts and hypotheses. Endocr. Rev. 17, 121–155 (1996).
Tucker, M.J., Morton, P.C., Sweitzer, C.L. & Wright, G. Cryopreservation of human embryos and oocytes. Curr. Opin. Obstet. Gynecol. 7, 188–192 (1995).
Tilly, J.L. & Ratts, V.S. Biological and clinical importance of ovarian cell death. Contemp. Obstet. Cynecol. 41, 59–86 (1996).
Tilly, J.L. Apoptosis and ovarian function. Rev. Reprod. 1, 162–172 (1996).
Tilly, J.L., Tilly, K.I. & Perez, G.I. The genes of cell death and cellular susceptibility to apoptosis in the ovary: A hypothesis. Cell Death Differ. 4, 180–187 (1997).
Reed, J.C. Bcl-2 and the regulation of programmed cell death J. Cell Biol. 124, 1–6 (1994).
Wyllie, A.H. The genetic regulation of apoptosis. Curr. Opin. Genet. Dev. 5, 97–104 (1995).
Yang, E. & Korsmeyer, S.J. Molecular thanatopsis: A discourse on the BCL-2 family and cell death. Blood 88, 386–401 (1996).
Tilly, K.I., Banerjee, S., Banerjee, P.P. & Tilly, J.L. Expression of the p53 and Wilms' tumor suppressor genes in the rat ovary: Gonadotropin repression in vivo and immunohistochemical localization of nuclear p53 protein to apoptotic granulosa cells of atretic follicles. Endocrinology 136, 1394–1402 (1995).
Keren-Tal, I. et al. Involvement of p53 expression in cAMP-mediated apoptosis in immortalized granulosa cells. Cxp. Cell Res. 218, 283–295 (1995).
Tilly, J.L., Tilly, K.I., Kenton, M.L. & Johnson, A.L. Expression of members of the bcl-2 gene family in the immature rat ovary: Equine chorionic gonadotropin-mediated inhibition of apoptosis is associated with decreased box and constitutive bcl-2 and bcl-x long messenger ribonucleic acid levels. Endocrinology 136, 232–241 (1995).
Ratts, V.S., Flaws, J.A., Kolp, R., Sorenson, C.M.S., Tilly, J.L. Ablation of bcl-2 gene expression decreases the number of oocytes and primordial follicles established in the post-natal female mouse gonad. Endocrinology 136, 3665–3668 (1995).
Knudson, C.M., Tung, K.S.K., Tourtellote, W.G., Brown, G.A.J. & Korsmeyer, S.J. Bax-deficient mice with lymphoid hyperplasia and male germ cell death. Science 270, 96–99 (1995).
Kugu, K. et al. Analysis of apoptosis and expression of bcl-2 gene family members in the human and baboon ovary. Cell Death Differ. (in the press).
Alnemri, E.S. et al. Human ICE/CED-3 protease nomenclature. Cell 87, 171 (1996).
Flaws, J.A. et al. lnterleukin-1β-converting enzyme-related proteases (IRPs) and mammalian cell death: Dissociation of IRP-induced oligonucleosomal endonuclease activity from morphological apoptosis in granulosa cells of the ovarian follicle. Endocrinology 136, 5042–5053 (1995).
Maravei, D.V. et al. Cleavage of cytoskeletal proteins by caspases during ovarian cell death: Evidence that cell-free systems do not always mimic apoptotic events in intact cells. Cell Death Differ. (in the press).
Spiegel, S., Foster, D. & Kolesnick, R.N. Signal transduction through lipid second messengers. Curr. Opin. Cell Biol. 8, 159–167 (1996).
Hannun, Y.A. Function of ceramide in coordinating cellular responses to stress. Science 274, 1855–1859 (1996).
Witty, J.P., Bridgham, J.T. & Johnson, A.L. Induction of apoptotic cell death in hen granulosa cells by ceramide. Endocrinology 137, 5269–5277 (1996).
Kaipia, A., Chun, S., Eisenhauer, K. & Hsueh, A.J.W. Tumor necrosis factor-α and its second messenger, ceramide, stimulate apoptosis in cultured ovarian follicles. Endocrinology 137, 4864–4870 (1996).
Martimbeau, S. & Tilly, J.L. Physiological cell death in endocrine-dependent tissues: An ovarian perspective. Clin. Endocrinol. 46, 241–254 (1997).
Alberts, D.S., Bachur, N.R. & Holtzman, J.L. The pharmokinetics of daunomycin in man. Clin. Pharmacol. Ther. 12, 96–104 (1971).
Speth, P.A., van Hoesel, Q.G. & Haanen, C. Clinical pharmokinetics of doxorubicin. Clin. Pharmokinetics 15, 15–31 (1988).
Bose, R. et al. Ceramide synthase mediates daunorubicin-induced apoptosis: An alternative mechanism for generating death signals. Cell 82, 405–414 (1995).
Jaffrezou, J.P. et al. Daunorubicin-induced apoptosis: Triggering of ceramide generation through sphingomyelin hydrolysis. EMBO J. 15, 2417–2424 (1996).
Wang, E., Norred, W.P., Bacon, C.W., Riley, R.T. & Merril, A.H. Inhibition of sphingolipid biosynthesis by fumonisins. J. Biol. Chem. 266, 14486–14490 (1991).
Cuvillier, O. et al. Suppression of ceramide-mediated programmed cell death by sphingosine-1-phosphate. Nature 381, 800–803 (1996).
Oltvai, Z.N., Milliman, C.L. & Korsmeyer, S.J. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74, 609–619 (1993).
Yin, C., Knudson, C.M., Korsmeyer, S.J. & Van Dyke, T. Bax suppresses tumorigenesis and stimulates apoptosis in vivo. Nature 385, 637–640 (1997).
McCurrach, M.E., Connor, T.M.F., Knudson, C.M., Korsmeyer, S.J. & Lowe, S.W. & Lowe, S.W., box-deficiency promotes drug resistance and oncogenic transformation by attenuating p53-dependent apoptosis. Proc. Natl. Acad. Sci. USA 94, 2345–2349 (1997).
Tchang, F., Gusse, M., Soussi, T. & Mechali, M. Stabilization and expression of high levels of p53 during early development in Xenopus laevis. Dev. Biol. 159, 163–172 (1993).
Lowe, S.W., Ruley, H.E., Jacks, T. & Housman, D.E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993).
Lane, D.P., Lu, X., Hupp, T. & Hall, P.A. The role of the p53 protein in the apoptotic response. Philos. Trans. R. Soc. Land. B. Biol. Sci. 345, 277–280 (1994).
Ko, L.J. & Prives, C. p53: Puzzle and paradigm. Cenes Dev. 10, 1054–1072 (1996).
Miyashita, T. & Reed, J.C. Tumor suppressor p53 is a direct transcriptional activator of the human box gene. Cell 80, 293–299 (1995).
Patel, T., Gores, G. & Kaufmann, S.H. The role of proteases during apoptosis. FASEB J 10, 587–597 (1996).
Nicholson, D.W. et al. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature 376, 37–43 (1995).
Jacobson, M.D., Weil, M. & Raff, M.C. Role of Ced-3/ICE-family proteases in staurosporine-induced programmed cell death. J. Cell. Biol. 133, 1041–1051 (1996).
Chinnaiyan, A.M. et al. Molecular ordering of the cell death pathway. Bcl-2 and Bcl-XL function upstream of the CED-3-like apoptotic proteases. J. Biol. Chem. 271, 4573–4584 (1996).
Xiang, J., Chao, D.T. & Korsmeyer, S.J. BAX-induced cell death may not require interleukin-1β-converting enzyme-like proteases. Proc. Natl. Acad. Sci. USA 93, 14559–14563 (1996).
Deveraux, Q.L., Takahashi, R., Salvesen, G.S. & Reed, J.C. X-linked IAP is a direct inhibitor of cell-death proteases. Nature 388, 300–304 (1997).
Jacobson, M.D., Burne, J.F. & Raff, M.C. Programmed cell death and Bcl-2 protection in the absence of a nucleus. EMBO J. 13, 1899–1910 (1994).
Schulze-Osthoff, K., Walczak, H., Droge, W. & Krammer, P.H. Cell nucleus and DNA fragmentation are not required for apoptosis. J. Cell Biol, 127, 15–20 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Perez, G., Knudson, C., Leykin, L. et al. Apoptosis-associated signaling pathways are required for chemotherapy-mediated female germ cell destruction. Nat Med 3, 1228–1232 (1997). https://doi.org/10.1038/nm1197-1228
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1197-1228
This article is cited by
-
Onco-fertility and personalized testing for potential for loss of ovarian reserve in patients undergoing chemotherapy: proposed next steps for development of genetic testing to predict changes in ovarian reserve
Fertility Research and Practice (2021)
-
The use of GnRH analogs in preserving ovarian function during chemotherapy
Middle East Fertility Society Journal (2021)
-
Individual-oocyte transcriptomic analysis shows that genotoxic chemotherapy depletes human primordial follicle reserve in vivo by triggering proapoptotic pathways without growth activation
Scientific Reports (2021)
-
The BCL-2 pathway preserves mammalian genome integrity by eliminating recombination-defective oocytes
Nature Communications (2020)
-
Apoptosis in the fetal testis eliminates developmentally defective germ cell clones
Nature Cell Biology (2020)