Abstract
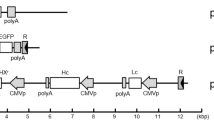
We have established cell lines that contain plasmid constructs carrying combinations of genes coding for human growth hormone (hGH) or the human hepatitis B virus surface antigen complex (HBsAg), placed under the expression control of a human heat shock promoter. In addition, neomycin resistance genes and oncogenes have been employed for selection of cell lines. These lines have been passed as tumours in rodents. After proteolytic dissociation of cells from excised tumours, up to 109 tumour cells were obtained in culture, per (20 mm diameter) tumour. These cells can produce, after heat induction, approximately similar quantities of hGH or HBsAg as the parental cells prior to passage as tumours in animals. In addition, we have cultivated one of these cell lines on spherical microcarriers (cytodex 1R) in 1 liter fermentors, using media with or without serum. Under these conditions, a daily production of mg quantities of hGH per tumour, per heat shock, was obtained. This procedure is applicable to the pilot scale production of natural proteins, or their protein-engineered variants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nover, L. 1987. Expression of heat shock genes in homologous and heterologous systems. Enzyme Microb. Technol. 9:130–144.
Amin, J., Mestril, R., Schiller, P., Dreano, M. and Voellmy, R. 1987. Organization of the Drosophila melanogaster hsp70 heat shock regulation unit. Mol. Cell. Biol. 7:1055–1062
Dreano, M., Brochot, J., Myers, A., Cheng-Mayer, C., Rungger, D., Voellmy, R. and Bromley, P. 1986. High-level, heat-regulated synthesis of proteins in eukaryotic cells. Gene 49:1–8
Dreano, M., Fouillet, X., Brochot, J., Vallet, J.-M., Michel, M.-L., Rungger, D. and Bromley, P. 1987. Heat-regulated expression of the hepatitis B virus surface antigen in the human Wish cell line. Virus Res. 8:43–59
Rancourt, D.E., Walker, V.K. and Davies, P.L. 1987. Flounder antifreeze protein synthesis under heat shock control in transgenic Drosophila melanogter. Mol. Cell. Biol. 7:2188–2195
Wu, B., Kingston, R.E. and Morimoto, R. 1986 Human HSP70 promoter contains at least two distinct regulatory domains. Proc. Nad. Acad. Sci. USA. 83:629–633
Wurm, F.M., Gwinn, K.A. and Kingston, R.E. 1986. Inducible overproduction of the mouse c-myc protein in mammalian cells. Proc. Natl. Acad. Sci. USA. 83:5414–5418
Xiao, H. and Lis, T.J. 1988. Germline transformation used to define key features of heat-shock response elements. Science 239:1139–1141.
Chessebeuf, M. and Padieu, P. 1984. Rat liver epithelial cell cultures in serum free medium. Primary cultures and derived cell lines expressing hepatic functions. In Vitro 20:780–795
Chessebeuf-Padieu, M., Mignot, G., Tsaconas, C., Fischbach, M., Padieu, P. and Mack, G. 1986. Serum free and protein free medium for the continuous culture of epithelial cell lines, and myeloma and hybridoma strains. Symposium “Quo Vadis”, Croupe Sanofi, Toulouse-Labége, France; MEDSI Publ., Paris, p. 385–397
Mignot, G., Dhainaut, F., Diez-Ibanez, M., Chessebeuf-Padieu, M. and Padieu, P. 1986. Culture de ligneées cellulaires épithéliales dans des milieux chimiquement définis sur microporteurs en biogénérateurs. C. R. Soc. Biol., Paris,180:460–472
Voellmy, R., Ahmed, A., Schiller, P., Bromley, P. and Rungger, D. 1985. Isolation and functional analysis of a human 70,000-Dalton heat shock protein gene segment. Proc. Natl. Acad. Sci. USA 82:4949–4953
Tabin, C.J., Bradley, S.M., Bargmann, C.I., Weinberg, R.A., Papageorge, A.G., Scolnick, E.M., Dhar, R., Lowy, D.R. and Chang, E.H. 1982. Mechanism of activation of a human oncogene. Nature 300:143–149.
Meneguzzi, G., Binetruy, B., Grisoni, M. and Cuzin, F. 1984. Plasmidal maintenance in rodent nbroblasts of a BPV1-pBR322 shuttle vector without immediately apparent oncogenic transformation of the recipient cells. EMBO J. 3:365–371
Rubin, H., Chu, B.M. and Arnstein, P. 1986. Dynamics of tumor growth and cellular adaptation after inoculation into nude mice of varying numbers of transformed 3T3 cells and of readaptation to culture of the tumor cells. Cancer Res. 46:2027–2034
Binetruy, B., Meneguzzi, G., Breathnach, R. and Cuzin, F. 1982. Recombinant DNA molecules comprising bovine papilloma virus type 1 DNA linked to plasmid DNA are maintained in a plasmidial state both in rodent nbroblasts and in bacterial cells. EMBO J. 1:621–628
Turek, L.P., Byrne, J.C., Lowy, D.R., Dvoretzky, I., Friedman, R.M. and Howley, P.M., 1982. Interferon induces morphologic reversion with elimination of extrachromosomal viral genomes in bovine papilloma virus-transformed mouse cells. Proc. Natl. Acad. Sci. USA 79:7914–7918
Lacey, M., Alpert, S. and Hanahan, D. 1986. Bovine papillomavirus genome elicits skin tumours in transgenic mice. Nature 322:609–612
Manniatis, T., Fritsch, E. and Sambrook, J. 1982. Molecular cloning: A laboratory manual. Cold Spring Harbor Laboratory Press, New York.
Graham, F.L. and Van Der EB, A.J. 1973. A new technique for assay of infectivity of human adenovirus 5 DNA. Virol. 52:456–467.
Southern, P. and Berg, P. 1982. Mammalian cell transformation with SV40 hybrid plasmid vectors, p. 41–45 In: Eucaryotic viral vectors. Y. Gluzman (Ed. ). Cold Spring Harbor Laboratory, Cold Spring Harbor, New York.
Danscher, G. and Norgaard, J.O.R. 1983. Light microscopic visualization of colloidal gold on resin-embedded tissue. J. Histochem. Cytochem. 31:1394–1398
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dreano, M., Fischbach, M., Montandon, F. et al. Production of Secretable Proteins Using the Passage In Vivo as Tumours of Cells Carrying Heat-Inducible Expression Constructs. Nat Biotechnol 6, 953–958 (1988). https://doi.org/10.1038/nbt0888-953
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0888-953