Abstract
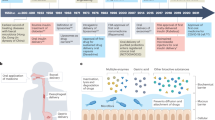
Biologically adhesive delivery systems offer important advantages1–5 over conventional drug delivery systems6. Here we show that engineered polymer microspheres made of biologically erodable polymers, which display strong adhesive interactions with gastrointestinal mucus and cellular linings, can traverse both the mucosal absorptive epithelium and the follicle-associated epithelium covering the lymphoid tissue of Peyer's patches. The polymers maintain contact with intestinal epithelium for extended periods of time and actually penetrate it, through and between cells. Thus, once loaded with compounds of pharmacological interest, the microspheres could be developed as delivery systems to transfer biologically active molecules to the circulation. We show that these microspheres increase the absorption of three model substances of widely different molecular size: dicumarol, insulin and plasmid DNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lenaerts, V., Couvreur, P., Grislain, L. & Maincent, P. in Bioadhesive Drug Delivery Systems (eds Lenaerts, V. & Gurny, R.) 93–104 (CRC Press, Boca Raton, FL, 1990).
Peppas, N. A. Hydrogels in Medicine and Pharmacy (CRC Press, Boca Raton, FL, 1987).
Ponchel, G. & Duchêne, D. in High Performance Biomaterials (ed. Szycher, M.) 231–242 (Technomic, Lancaster, PA, 1991).
Davis, S. S. & Illum, L. Biomaterials 9, 111–115 (1988).
Frisbie, C. D., Lawrence, F., Rozsnyai, A. N., Wrighton, M. S. & Charles, M. L. Science 265, 2071–2073 (1994).
Langer, R. & Chasin, M. Biodegradable Polymers as Drug Delivery Systems (Marcel Decker, New York, 1990).
Chickering, D. E., Jacob, J. S. & Mathiowitz, E. React. Polym. 25, 189–206 (1995).
Mathiowitz, E., Jacob, J., Chickering, D. & Pekarek, K. Macromolecules 26, 6756–6765 (1993).
Chickering, D. E. & Mathiowitz, E. J. Control. Release 34, 251–261 (1995).
Jacob, J., Santos, C., Carino, G., Chickering, D. & Mathiowitz, E. Proc. 22nd Int. Symp. Controlled Release of Bioactive Materials, 312–313 (Controlled Release Society, Inc., Deerfield, IL, 1995).
Chickering, D. E. III, Jacob, J. S. & Mathiowitz, E. Biotechnol. Bioeng. 52, 96–101 (1996).
Carino, G., Santos, C., Chen, C. J. & Mathiowitz, E. Proc. 22nd Int. Symp. Controlled Release of Bioactive Materials, 314–315 (Controlled Release Society, Inc., Deerfield, IL, 1995).
Sanders, E. & Ashworth, C. T. Exp. Cell Res. 22, 137–145 (1961).
Ermak, T. H., Dougherty, E. P., Bhagat, H. R., Kabok, Z. & Pappo, J. Cell Tissue Res. 279, 433–436 (1995).
Florence, A. & Jani, P. U. Drug Safety 10, 233–266 (1994).
Florence, A. T., Hillery, A. M., Hussain, N. & Jani, P. U. J. Control. Release 36, 39–46 (1995).
LeFevre, M. E., Olivo, R., Vanderhoff, J. W. & Joel, D. D. Proc. Soc. Exp. Biol. Med. 159, 298–302 (1978).
Lefevre, M. E., Joel, D. D. & Schidlovsky, G. Proc. Soc. Exp. Biol. Med. 179, 522–528 (1985).
Jani, P., Halbert, G. W., Langridge, J. & Florence, A. T. J. Pharm. Pharmacol. 41, 809–812 (1989).
Jani, P., Halbert, G. W., Langridge, J. & Florence, A. T. J. Pharm. Pharmacol. 42, 821–826 (1990).
Hillery, A. M., Jani, P. U. & Florence, A. T. J. Drug Target. 2, 151–156 (1994).
Bockman, D. E. & Cooper, M. D. Am. J. Anat. 136, 455–478 (1973).
Akers, M. J., Lach, J. L. & Fischer, L. J. J. Pharma. Sci. 62, 391–395 (1973).
Goodman, L. S. & Gilman, A. The Pharmacological Basis of Therapeutics (Macmillan, New York, 1975).
Tutwiler, G. F., Kirsch, T. & Bridi, G. Diabetes 27, 856–867 (1978).
Wolff, J. A. et al. Biotechniques 11, 474–485 (1991).
Lau, C. et al. Hum. Gene Ther. 6, 1145–1151 (1995).
Domb, A., Mathiowitz, E., Ron, E., Giannos, S. & Langer, R. J. Polym. Sci. 29, 571–579 (1991).
Wolff, J. A. et al. Science 247, 1465–1468 (1990).
Lee, S. W., Trapnell, B. C., Rade, J. J., Virmani, R. & Dichek, D. A. Circ. Res. 73, 797–807 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mathiowitz, E., Jacob, J., Jong, Y. et al. Biologically erodable microspheres as potential oral drug delivery systems. Nature 386, 410–414 (1997). https://doi.org/10.1038/386410a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/386410a0
This article is cited by
-
Bioinspired oral delivery devices
Nature Reviews Bioengineering (2023)
-
Stereocomplexed microparticles loaded with Salvia cadmica Boiss. extracts for enhancement of immune response towards Helicobacter pylori
Scientific Reports (2023)
-
Mucosa-interfacing electronics
Nature Reviews Materials (2022)
-
Foundations of gastrointestinal-based drug delivery and future developments
Nature Reviews Gastroenterology & Hepatology (2022)
-
Doxorubicin Release from Bovine Serum Albumin Microparticles
Pharmaceutical Chemistry Journal (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.