Abstract
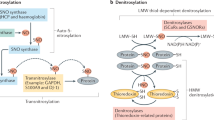
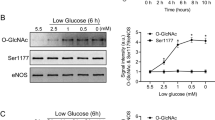
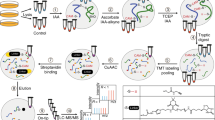
Nitric oxide (NO) has been linked to numerous physiological and pathophysiological events that are not readily explained by the well established effects of NO on soluble guanylyl cyclase. Exogenous NO S-nitrosylates cysteine residues in proteins, but whether this is an important function of endogenous NO is unclear. Here, using a new proteomic approach, we identify a population of proteins that are endogenously S-nitrosylated, and demonstrate the loss of this modification in mice harbouring a genomic deletion of neuronal NO synthase (nNOS). Targets of NO include metabolic, structural and signalling proteins that may be effectors for neuronally generated NO. These findings establish protein S-nitrosylation as a physiological signalling mechanism for nNOS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Denninger, J. W. & Marletta, M. A. Biochim. Biophys. Acta 1411, 334–350 (1999).
Stamler, J. S., Toone, E. J., Lipton, S. A. & Sucher, N. J. Neuron 18, 691–696 (1997).
Stamler, J. S. et al. Proc. Natl Acad. Sci. U. S. A. 89, 444–448 (1992).
Mayer, B. et al. J. Biol. Chem. 273, 3264–3270 (1998).
Lander, H. M., Ogiste, J. S., Pearce, S. F., Levi, R. & Novogrodsky, A. J. Biol. Chem. 270, 7017–7020 (1995).
Broillet, M. C. & Firestein, S. Neuron 16, 377–385 (1996).
Molina y Vedia, L. et al. J. Biol. Chem. 267, 24929–24932 (1992).
Kashiba-Iwatsuki, M. et al. J. Biochem. (Tokyo) 122, 1208–1214 (1997).
Dicks, A. P. & Williams, D. L. Chem. Biol. 3, 655–659 (1996).
Feelisch, M. Naunyn Schmiedebergs Arch. Pharmacol. 358, 113–122 (1998).
Jia, L., Bonaventura, C., Bonaventura, J. & Stamler, J. S. Nature 380, 221–226 (1996).
Xu, L., Eu, J. P., Meissner, G. & Stamler, J. S. Science 279, 234–237 (1998).
Mannick, J. B. et al. Science 284, 651–654 (1999).
Stamler, J. S. et al. Proc. Natl Acad. Sci. U. S. A. 89, 7674–7677 (1992).
Kenyon, G. L. & Bruice, T. W. Methods Enzymol. 47, 407–430 (1977).
Stuchbury, T. et al. Biochem. J. 151, 417–432 (1975).
Lander, H. M. et al. J. Biol. Chem. 272, 4323–4326 (1997).
Kluge, I., Gutteck-Amsler, U., Zollinger, M. & Do, K. Q. J. Neurochem. 69, 2599–2607 (1997).
Gaston, B. et al. Proc. Natl Acad. Sci. USA 90, 10957–10961 (1993).
Meyer, D. J., Kramer, H., Ozer, N., Coles, B. & Ketterer, B. FEBS Lett. 345, 177–180 (1994).
Lipton, S. A. Cell Death Differ. 6, 943–951 (1999).
Huang, P. L., Dawson, T. M., Bredt, D. S., Snyder, S. H. & Fishman, M. C. Cell 75, 1273–1286 (1993).
Choi, Y. B. et al. Nature Neurosci. 3, 15–21 (2000).
Beaumont, V. & Zucker, R. S. Nature Neurosci. 3, 133–141 (2000).
Ludwig, A., Zong, X., Jeglitsch, M., Hofmann, F. & Biel, M. Nature 393, 587–591 (1998).
Li, H. & Forstermann, U. J. Pathol. 190, 244–254 (2000).
Enikolopov, G., Banerji, J. & Kuzin, B. Cell Death Differ. 6, 956–963 (1999).
Quinn, C. C., Gray, G. E. & Hockfield, S. J. Neurobiol. 41, 158–164 (1999).
Inglis, F. M., Furia, F., Zuckerman, K. E., Strittmatter, S. M. & Kalb, R. G. J. Neurosci. 18, 10493–10501 (1998).
Kuzin, B., Roberts, I., Peunova, N. & Enikolopov, G. Cell 87, 639–649 (1996).
Kuzin, B. et al. Curr. Biol. 10, 459–462 (2000).
Nicotera, P., Leist, M. & Manzo, L. Trends Pharmacol. Sci. 20, 46–51 (1999).
Gross, W. L. et al. Proc. Natl Acad. Sci. USA 93, 5604–5609 (1996).
Wolosker, H., Panizzutti, R. & Engelender, S. FEBS Lett 392, 274–276 (1996).
Borgs, M. et al. Hepatology 23, 1564–1571 (1996).
Sato, T., Kamata, Y., Irifune, M. & Nishikawa, T. Brain Res. 704, 117–120 (1995).
Bonfoco, E. et al. J Neurochem. 67, 2484–2493 (1996).
Andrade, F. H., Reid, M. B., Allen, D. G. & Westerblad, H. J. Physiol. (Lond) 509, 577–586 (1998).
Colledge, M. & Scott, J. D. Trends Cell Biol. 9, 216–221 (1999).
Brenman, J. E. et al. Cell 84, 757–767 (1996).
Sattler, R. et al. Science 284, 1845–1848 (1999).
Jaffrey, S. R., Snowman, A. M., Eliasson, M. J., Cohen, N. A. & Snyder, S. H. Neuron 20, 115–124 (1998).
Smith, D. B. & Johnson, K. S. Gene 67, 31–40 (1988).
Huber, R. E. & Criddle, R. S. Arch. Biochem. Biophys. 122, 164–173 (1967).
Stadtman, T. C. Annu. Rev. Biochem. 49, 93–110 (1980).
Otero, G. et al. Mol. Cell 3, 109–118 (1999).
Wittschieben, B. O. et al. Mol. Cell 4, 123–128 (1999).
Acknowledgements
We thank A. Grewal and A. Nazarian for assistance with mass fingerprinting, A. Kashani for assistance during early stages of this project, and D. Guastella for genotyping the mice. This work was supported by USPHS grants (to S.H.S.), a grant from the National Cancer Institute (to P.T.), a Young Investigator Award from the National Alliance for Research on Schizophrenia and Affective Disorders (to S.R.J.), and a Howard Hughes Fellowship for Physicians (to C.D.F.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jaffrey, S., Erdjument-Bromage, H., Ferris, C. et al. Protein S-nitrosylation: a physiological signal for neuronal nitric oxide. Nat Cell Biol 3, 193–197 (2001). https://doi.org/10.1038/35055104
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35055104
This article is cited by
-
Pivotal role for S-nitrosylation of DNA methyltransferase 3B in epigenetic regulation of tumorigenesis
Nature Communications (2023)
-
S-Nitrosylation of cathepsin B affects autophagic flux and accumulation of protein aggregates in neurodegenerative disorders
Cell Death & Differentiation (2022)
-
A nitric oxide burst at the shoot apex triggers a heat-responsive pathway in Arabidopsis
Nature Plants (2022)
-
Arsenic alters nitric oxide signaling similar to autism spectrum disorder and Alzheimer’s disease-associated mutations
Translational Psychiatry (2022)
-
Protein S-nitrosation differentially modulates tomato responses to infection by hemi-biotrophic oomycetes of Phytophthora spp.
Horticulture Research (2021)