Abstract
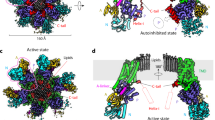
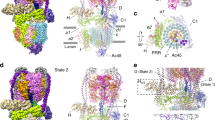
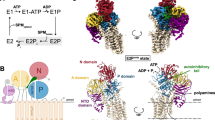
The H+-ATPase from the plasma membrane of Neurospora crassa is an integral membrane protein of relative molecular mass 100K, which belongs to the P-type ATPase family that includes the plasma membrane Na+/K+-ATPase and the sarcoplasmic reticulum Ca2+-ATPase. The H+-ATPase pumps protons across the cell's plasma membrane using ATP as an energy source, generating a membrane potential in excess of 200 mV (1–3). Despite the importance of P-type ATPases in controlling membrane potential and intracellular ion concentrations, little is known about the molecular mechanism they use for ion transport. This is largely due to the difficulty in growing well ordered crystals and the resulting lack of detail in the three-dimensional structure of these large membrane proteins. We have now obtained a three-dimensional map of the H+-ATPase by electron crystallography of two-dimensional crystals grown directly on electron microscope grids. At an in-plane resolution of 8 Å, this map reveals ten membrane-spanning α-helices in the membrane domain, and four major cytoplasmic domains in the open conformation of the enzyme without bound ligands.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Scarborough, G. A. in Handbook of Biological Physics Vol. 2(eds Konings, W. N., Kaback, H. R. & Lolkema, J. S.) 75–92 (Elsevier Science, Amsterdam, 1996).
Scarborough, G. A. in Molecular Aspects of Transport Proteins (ed. de Pont, J. J. H. H. M.) 117–134 (Elsevier, Amsterdam, 1992).
Nakamoto, R. K. & Slayman, C. W. Molecular properties of the fungal plasma-membrane H+-ATPase. J. Bioenerg. Biomemb. 21, 621–632 (1989).
Nakamoto, R. K., Rao, R. & Slayman, C. W. Transmembrane segments of the P-type cation-transporting ATPases. Ann. N. Y. Acad. Sci. 574, 165–179 (1989).
Mandala, S. M. & Slayman, C. W. The amino and carboxyl termini of the Neurospora plasma membrane H+-ATPase are cytoplasmically located. J. Biol. Chem. 264, 16276–16281 (1989).
Hennessey, J. P. J & Scarborough, G. A. Direct evidence for the cytoplasmic location of the NH2- and COOH-terminal ends of the Neurospora crassa plasma membrane H+-ATPase. J. Biol. Chem. 265, 532–537 (1990).
Scarborough, G. A. & Hennessey, J. P. J Identificaton of the major cytoplasmic regions of the Neurospora crassa plasma membrane H+-ATPase using protein chemical techniques. J. Biol. Chem. 265, 16145–16149 (1990).
Rao, U. S., Hennessey, J. P. J & Scarborough, G. A. Identification of the membrane-embedded regions of the Neurospora crassa plasma membrane H+-ATPase. J. Biol. Chem. 266, 14740–14746 (1991).
Rao, U. S., Bauzon, D. D. & Scarborough, G. A. Cytoplasmic location of amino acids 359–440 of the Neurospora crassa plasma membrane H+-ATPase. Biochim. Biophys. Acta 1108, 153–158 (1992).
Goormaghtigh, E., Chadwick, C. & Scarborough, G. A. Monomers of the Neurospora plasma membrane H+-ATPase catalyse efficient proton translocation. J. Biol. Chem. 261, 7466–7471 (1986).
Chadwick, C., Goormaghtigh, E. & Scarborough, G. A. Ahexameric form of the Neurospora crassa plasma membrane H+-ATPase. Arch. Biochem. Biophys. 252, 348–356 (1987).
Henderson, R. & Unwin, P. N. T. Three-dimensional model of purple membrane obtained by electron microscopy. Nature 257, 28–32 (1975).
Henderson, R. et al. Model for the structure of bacteriorhodopsin based on high-resolution electron cryo-microscopy. J. Mol. Biol. 213, 899–929 (1990).
Kühlbrandt, W., Wang, D. N. & Fujiyoshi, Y. Atomic model of plant light-harvesting complex by electron crystallography. Nature 367, 614–621 (1994).
Walz, T. et al. The three-dimensional structure of aquaporin-1. Nature 387, 624–627 (1997).
Cheng, A., v. Hoek, A. N., Yeager, M., Verkman, A. S. & Mitra, A. Three-dimensional organisation of a human water channel. Nature 387, 627–630 (1997).
Li, H., Lee, S. & Jap, B. Molecular design of aquaporin-1 water channel as revealed by electron crystallography. Nature Struct. Biol. 4, 263–265 (1997).
Cyrklaff, M., Auer, M., Kühlbrandt, W. & Scarborough, G. A. 2-D structure of the Neurospora crassa plasma membrane ATPase as determined by electron cryomicroscopy. EMBO J. 14, 1854–1857 (1995).
Goormaghtigh, E., Vigneron, L., Scarborough, G. A. & Ruysschaert, J.-M. Tertiary conformational changes of the Neurospora crassa plasma membrane H+-ATPase monitored by hydrogen/deuterium exchange kinetics: a Fourier transform infrared spectroscopy approach. J. Biol. Chem. 269, 27409–27413 (1994).
Toyoshima, C., Sasabe, H. & Stokes, D. L. Three-dimensional cryo-electron microscopy of the calcium ion pump in the sarcoplasmic reticulum membrane. Nature 362, 469–471 (1993).
Serrano, R. Structure and function of proton translocating ATPase in plasma membranes of plants and fungi. Biochem. Biophys. Acta 947, 1–28 (1988).
Scarborough, G. A. Large single crystals of the Neurospora crassa plasma membrane H+-ATPase: An approach ot the crystallisation of integral membrane proteins. Acta Crystallogr. D 50, 643–649 (1994).
Dubochet, J. et al. Cryo-electron microscopy of vitrified specimens. Quart. Rev. Biophys. 21, 129–228 (1988).
Crowther, R. A., Henderson, R. & Smith, J. M. MRC image processing programs. J. Struct. Biol. 116, 9–16 (1996).
Collaborate Computational Project No. 4. The CCP4 suite: Programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methdos for building protein models in electron density maps. Acta Crystallogr. A 47, 110–119 (1991).
Sheehan, B., Fuller, S. D., Piqus, M. E. & Yeager, M. AVS software for visualisation in molecular biology. J. Struct. Biol. 116, 99–106 (1996).
Frank, J. et al. SPIDER and WEB: Processing and visualisation of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996).
Kühlbrandt, W. Discrimination of protein and nucleic acids by electron microscoy using contrast variation. Ultramicroscopy 7, 221–232 (1982).
Unwin, P. N. T. & Ennis, P. D. Two configurations of a channel-forming membrane protein. Nature 307, 609–613 (1984).
Acknowledgements
We thank M. Radermacher for help with the program SPIDER, D. Mills and J. Vonck for help with the JEOL 3000 SFF, and J. Postma for assistance with computer graphics. W.K. gratefully acknowledges financial support from the Deutsche Forschungsgemeinschaft. G.A.S. was supported by a grant from USPHS NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Auer, M., Scarborough, G. & Kühlbrandt, W. Three-dimensional map of the plasma membrane H+-ATPase in the open conformation. Nature 392, 840–843 (1998). https://doi.org/10.1038/33967
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/33967
This article is cited by
-
Structure and activation mechanism of the hexameric plasma membrane H+-ATPase
Nature Communications (2021)
-
Overexpression of PMA1 enhances tolerance to various types of stress and constitutively activates the SAPK pathways in Saccharomyces cerevisiae
Applied Microbiology and Biotechnology (2017)
-
The plant plasma membrane proton pump ATPase: a highly regulated P-type ATPase with multiple physiological roles
Pflügers Archiv - European Journal of Physiology (2009)
-
Protons and how they are transported by proton pumps
Pflügers Archiv - European Journal of Physiology (2009)
-
Crystal structure of the plasma membrane proton pump
Nature (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.