Abstract
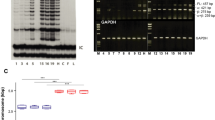
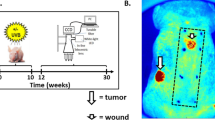
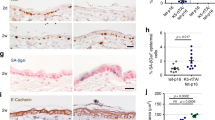
In chemical carcinogenesis, initiation is defined as the exposure of a tissue, generally mouse skin, to a limited dose of carcinogen which by itself does not induce tumours1. Subsequent treatments, usually by tumour promoters, are required to allow expression of initiation. In the absence of further treatment, the initiated cells are not recognizable by any known biochemical or biological criteria and the tissue itself functions normally. Much research has centred on elucidating the mechanism of initiation1–3. It seems likely that initiation involves a genetic change in the target cell as its expression after tumour promotion is a permanent characteristic of the tissue, even if many cell generations have elapsed between initiation and promotion4. Studies on tissues from patients with familial polyposis coli suggest that the occurrence of initiated cells in these individuals is due to an autosomal dominant gene5. Few studies have actually attempted to define the biological nature of the initiating alteration itself. We report here that cells resistant to terminal differentiation can be readily isolated from skin of BALB/c mice exposed to an initiating dose of carcinogen in vivo but not from control mouse skin. Also, we found that one strain of mouse (SENCAR) might contain a preexisting population of cells resistant to differentiation, and possibly initiated for tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yuspa, S. H. & Harris, C. C. in Cancer Epidemiology and Prevention (eds Schottenfeld, D. & Fraumeni, J.) (Saunders, Philadelphia, in the press).
Miller, E. C. Cancer Res. 38, 1479–1496 (1978).
Yuspa, S. H., Hennings, H. & Saffiotti, U. J. invest. Derm. 67, 199–208 (1976).
Van Duuren, B. L., Sivak, A., Katz, C., Seidman, I. & Melchionne, S. Cancer Res. 35, 502–505 (1975).
Kopelovitch, L. in Colorectal Cancer; Prevention, Etiology and Screening (eds Winawer, S., Schottenfeld, D. & Sherlock, P.) 97–108 (Raven, New York, 1981).
Kulesz-Martin, M., Koehler, B., Hennings, H. & Yuspa, S. H. Carcinogenesis 1, 995–1006 (1980).
Yuspa, S. H., Lichti, U., Morgan, D. & Hennings, H. in Biochemistry of Normal and Abnormal Epidermal Differentiation (eds Bernstein, I. A. & Seiji, M.) 171–188 (University of Tokyo Press, 1980).
Yuspa, S. H., Hennings, H., Kulesz-Martin, M. & Lichti, U. in Cocarcinogenesis and Biological Effects of Tumour Promoters (ed. Hecker, E.) (Raven, New York, in the press).
Yuspa, S. H., Hawley-Nelson, P., Koehler, B. & Stanley, J. R. Cancer Res. 40, 4694–4703 (1980).
Hennings, H. et al. Cell 19, 245–254 (1980).
Yuspa, S. H., Hawley-Nelson, P., Stanley, J. R. & Hennings, H. Transplantn Proc. 12, Suppl. 1, 114–122 (1980).
Hennings, H. et al. Cancer Res. 41, 773–779 (1981).
Yuspa, S. H., Ben, T. B., Hennings, H. & Lichti, U. Biochem. biophys. Res. Commun. 97, 700–708 (1980).
Thurnherr, N., Deschner, E., Stonehill, E. & Lipkin, M. Cancer Res. 33, 940–945 (1973).
Klein-Szanto, A. J. P., Nettsheim, P., Topping, D. C. & Olson, A. C. Carcinogenesis 1, 1007–1016 (1980).
Morson, B. & Bussey, H. Curr. Probl. Surg. 2, 1–52 (1970).
Kopelovich, L., Bias, N. E. & Helson, L. Nature 282, 619–621 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yuspa, S., Morgan, D. Mouse skin cells resistant to terminal differentiation associated with initiation of carcinogenesis. Nature 293, 72–74 (1981). https://doi.org/10.1038/293072a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/293072a0
This article is cited by
-
IL-13 from intraepithelial lymphocytes regulates tissue homeostasis and protects against carcinogenesis in the skin
Nature Communications (2016)
-
Milestones in Skin Carcinogenesis: The Biology of Multistage Carcinogenesis
Journal of Investigative Dermatology (2014)
-
Analysis of the peroxisome proliferator-activated receptor-β/δ (PPARβ/δ) cistrome reveals novel co-regulatory role of ATF4
BMC Genomics (2012)
-
Selective clonal expansion and microenvironmental permissiveness in tobacco carcinogenesis
Oncogene (2002)
-
IKKα controls formation of the epidermis independently of NF-κB
Nature (2001)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.