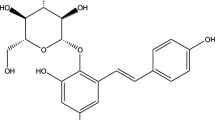
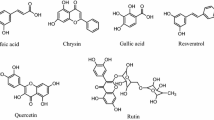
Abstract
Purpose. Extracts made from berries, herbs, and various plant materials, which might possess a range of activities, are used as health promoting products. Because little is known about their effects on the absorption of co-administered drugs, the effects of some food supplements, Finnish berries, and herbs were studied on the permeability of some commonly used drugs.
Methods. The permeabilities of verapamil, metoprolol, ketoprofen, paracetamol, and furosemide were studied across Caco-2 cell monolayers with contemporaneously administered extracts from flax seed, purple loosestrife, and Scots pine bark; bilberries, cowberries, and raspberries; oregano, rosemary, and sage. Toxicological tests were conducted to determine cellular damage.
Results. The effects of extracts on drug permeabilities were generally minor. Flax seed decreased the permeability of all drugs except verapamil. Purple loosestrife and pine decreased verapamil and metoprolol permeability. Changes caused by berries were mainly pH-related. Rosemary and oregano enhanced furosemide permeability.
Conclusions. Ingestion of extracts of herbs and berries studied are not expected to markedly change the permeabilities of highly permeable drugs. Harmful effects at sites of or during absorption are unlikely. However, if high doses of extracts are administered with low permeable drugs in vitro,effects on drug permeabilities could not be excluded. Use of such extracts should therefore be evaluated during continuous medication.
Similar content being viewed by others
REFERENCES
G. Wilkinson. The effects of diet, aging and disease states on presystemic elimination and oral drug bioavailability in humans. Adv. Drug Deliv. Rev. 27:129–159 (1997).
K. E. Anderson, A. H. Cooney, and A. Kappas. Nutrition and oxidative drug metabolism in man: relative influence of dietary lipids, carbohydrate and protein. Clin. Pharmacol. Ther. 26:493– 501 (1979).
T. C. Fagan, T. Walle, M. J. Oexmann, U. K. Walle, S. A. Bai, and T. E. Gaffney. Increased clearance of propranolol and theophyl-line by high-protein compared with high-carbohydrate diet. Clin. Pharmacol. Ther. 41:402–406 (1987).
E. J. Pantuck, C. B. Pantuck, A. Kappas, A. H. Conney, and K. E. Anderson. Effects of protein and carbohydrate content of diet on drug conjugation. Clin. Pharmacol. Ther. 50:254–258 (1991).
P. G. Welling. Effect of food on drug absorption. Annu. Rev. Nutr. 16:383–415 (1996).
A. Wallace and G. Amsden. Is it really OK to take this with food? Old interactions with a new twist. J. Clin. Pharmacol. 42:435–441 (2002).
W. Abebe. Herbal Medication: potential for adverse interactions with analgesic drugs. J. Clin. Pharm. Ther. 27:391–401 (2002).
R. Walgren, K. Walle, and T. Walle. Transport of quercetin and its glucosides across human intestinal epithelial Caco-2 cells. Bio-chem. Pharmacol. 55:1721–1727 (1998).
R. Walgren, K. Karnaky, G. Lindenmayer, and T. Walle. Efflux of dietary flavonoid quercetin 4--β--glucoside across human intes-tinal Caco-2 cell monolayers by apical multidrug resistance-associated protein-2. J. Pharmacol. Exp. Ther. 294:830–836 (2000).
R. Walgren, J.-T. Lin, R. Kinne, and T. Walle. Cellular uptake of dietary flavonoid quercetin 4--β--glucoside by sodium-dependent glucose transporter SGLT1. J. Pharmacol. Exp. Ther. 294:837– 843 (2000).
P. Artursson. Epithelial transport of drugs I. A model for study-ing the transport of drugs (_-blocking agents) over an intestinal epithelial cell line (Caco-2). J. Pharm. Sci. 79:476–482 (1990).
P. Artursson, K. Palm, and K. Luthman. Caco-2 monolayers in experimental and theoretical predictions of drug transport. Adv. Drug Deliv. Rev. 22:67–84 (1996).
L.-S. Gan and D. Thakker. Applications of the Caco-2 model in the design and development of orally active drugs: elucidation of biochemical and physical barriers posed by the intestinal epithe-lium. Adv. Drug Deliv. Rev. 23:77–98 (1997).
G. L. Amidon, H. Lennernäs, V. P. Shah, and J. R. Crison. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bio-availability. Pharm. Res. 12:413–420 (1995).
M. Markowska, S. Oberle, C.-P. Juzwin, M. Hsu, M. Gryszk-iewicz, and A. J. Streeter. Optimizing Caco-2 cell monolayers to increase throughput in drug intestinal absorption analysis. J. Pharmacol. Toxicol. Methods 46:51–55 (2001).
C. Tannergren, P. Langguth, and K.-J. Hoffmann. Compound mixtures in Caco-2 cell permeability screens as a means to in-crease screening capacity. Pharmazie 56:337–342 (2001).
L. Laitinen, H. Kangas, A. M. Kaukonen, K. Hakala, T. Kotiaho, R. Kostiainen, and J. Hirvonen. N-in-one permeability studies of heterogeneous sets of compounds across Caco-2 cell monolayers. Pharm. Res. 20:187–197 (2003).
A. Palou, F. Serra, and C. Pico. General aspects of the assessment of functional foods in the European Union. Eur. J. Clin. Nutr. 57:S12–S17 (2003).
J.-P. Rauha, J.-L. Wolfender, J.-P. Salminen, K. Pihlaja, K. Hostettmann, and H. Vuorela. Characterization of the polyphe-nolic composition of purple loosestrife (Lythrum salicaria). Zeitschr. Naturforsch. 56C:13–20 (2001).
H. J. D. Dorman, A. Peltoketo, R. Hiltunen, and M. J. Tikkanen. Laitinen et al. 1914.Characterisation of the antioxidant properties of de-odourised aqueous extracts from selected Lamiaceaeherbs. Food Chem. 83:255–262 (2003).
V. L. Singleton, R. Orthofer, and R. M. Lamuela-Raventós. Analysis of total phenols and other oxidants by means of Folin-Ciocalteu reagent. Meth. Enzym. 299:152–178 (1999).
T. Mosman. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immun. Meth. 65:55–63 (1983).
M. P. Kahkönen, A. Hopia, and M. Heinonen. Berry phenolics and their antioxidant activity. J. Agric. Food Chem. 49:4076–4082 (2001).
R. Puupponen-Pimia, L. Nohynek, C. Meier, M. Kahkönen, M. Heinonen, A. Hopia, and K.-M. Oksman-Caldentey. Antimicrobial properties of phenolic compounds from berries. J. Appl. Mi-crobiol. 90:494–507 (2001).
M. Kähkönen, A. Hopia, H. Vuorela, J.-P. Rauha, K. Pihlaja, T. Kujala, and M. Heinonen. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem. 47:3954– 3962 (1999).
S. Häkkinen, M. Heinonen, S. Kärenlampi, H. Mykkänen, J. Ruuskanen, and R. Törrönen. Screening of selected flavonoids and phenolic acids in 19 berries. Food Res. Int. 32:345–353 (1999).
H. Takanaga, I. Tamai, and A. Tsuji. pH-dependent and carrier-mediated transport of salicylic acid across Caco-2 cells. J. Pharm. Pharmacol. 46:567–570 (1994).
T. Tsuruo, H. Iida, S. Tsukagoshi, and Y. Sakurai. Overcoming of vincristine resistance in P388 leukemia in vivo and in vitro through enhanced cytotoxicity of vincristine and vinblastine by verapamil. Cancer Res. 41:1967–1972 (1981).
S. Orlowski, L. Mir, J. Belehradek, and M. Garrigos. Effects of steroids and verapamil on P-glycoprotein ATPase activity: pro-gesterone, desoxycorticosterone corticosterone and verapamil are mutually non-exclusive modulators. Biochem. J. 317:515–522 (1996).
S. Doppenschmitt, H. Spahn-Langguth, C. G. Regårdh, and P. Langguth. Role of P-glycoprotein-mediated secretion in absorp-tive drug permeability: an approach using passive membrane per-meability and affinity to P-glycoprotein. J. Pharm. Sci. 88:1067– 1072 (1999).
B. Nare, R. K. Prichard, and E. Georges. Characterization of rhodamine-123 binding to P-glycoprotein in human multidrug-resistant cells. Mol. Pharmacol. 45:1145–1152 (1994).
S. D. Flanagan, L. H. Takahashi, X. Liu, and L. Z. Benet. Con-tributions of saturable active secretion, passive transcellular, and paracellular diffusion to the overall transport of furosemide across adenocarcinoma (Caco-2) cells. J. Pharm. Sci. 91:1169– 1177 (2002).
B. D. Oomah and G. Mazza. Effect of dehulling on chemical composition and physical properties of flax seed. Lebensm.-wiss. u.-Technol. 30:135–140 (1997).
V. Klein, V. Chajès, E. Germain, G. Schulgen, M. Pinault, D. Malvy, T. Lefrancq, A. Fignon, O. Le Floch, C. Lhuillery, and P. Bougnoux. Low alpha-linolenic acid content of adipose breast tissue is associated with increased risk of breast cancer. Eur. J. Cancer 36:335–340 (2000).
J. Bruneton. Pharmacognosy, Phytochemistry, Medicinal Plants. Lavoisier Publishing, Paris, France, pp. 105–106 (1995).
J.-P. Rauha, S. Remes, M. Heinonen, A. Hopia, M. Kähkönen, T. Kujala, K. Pihlaja, H. Vuorela, and P. Vuorela. Antimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds. Int. J. Food Microbiol. 56:3–12 (2000).
A. Saleem, H. Kivela, and K. Pihlaja. Antioxidant activity of pine bark constituents. Zeitschr. Naturforsch 58C:351–354 (2003).
L. Packer, G. Rimbach, and F. Virgili. Antioxidant acitivity and biologic properties of a procyanidin-rich extract from pine (Pinus marittima) bark, pycnogenol. Free Radic. Biol. Med. 27:704–724 (1999).
M. Nardini, C. Scaccini, L. Packer, and F. Virgili. In vitro inhi-bition of the activity of phosphorylase kinase, protein kinase C and protein kinase A by caffeic acid and a procyanidin-rich pine bark (Pinus marittima) extract. Biochim. Biophys. Acta 1474:219– 225 (2000).
S. Neuhoff, A.-L. Ungell, I. Zamora, and P. Artursson. pH-dependent bi-directional transport of weakly basic drugs across Caco-2 monolayers: implications for drug-drug interactions. Pharm. Res. 20:1141–1148 (2003).
M. Sakai, A. B. J. Noach, M. C. M. Blom-Roosemalen, A. G. deBoer, and D. D. Breimer. Absorption enhancement of hydro-philic compounds by verapamil in Caco-2 cell monolayers. Bio-chem. Pharmacol. 48:1199–1210 (1994).
M. Ollanketo, A. Peltoketo, K. Hartonen, R. Hiltunen, and M.-L. Riekkola. Extraction of sage (Salvia officinalisL.) by pressurized hot water and conventional methods: antioxidant activity of the extracts. Eur. Food Res. Technol. 215:158–163 (2002).
Y. Liu and M. Hu. Absorption and metabolism of flavonoids in the Caco-2 cell culture model and a perfused rat intestinal model. Drug Metab. Disp. 30:370–377 (2002).
K. Murota, S. Shimizu, S. Miyamoto, T. Izumi, A. Obata, and M. Kikuchi. and J. Terao. Unique uptake and transport of isoflavone aglycones by human intestinal Caco-2 cells: comparison of isofla-vonoids and flavonoids. J. Nutr. 132:1956–1961 (2002).
U. K. Walle, K. L. French, R. A. Walgren, and T. Walle. Trans-port of genistein-7-glucoside by human intestinal Caco-2 cells: potential role for MRP2. Res. Comm. Mol. Pathol. Pharmacol. 103:45–56 (1999).
A. F. Castro and G. A. Altenberg. Inhibition of drug transport by genistein in multidrug-resistant cells expressing P-glycoprotein. Biochem. Pharmacol. 53:89–93 (1997).
C. Pascaud, M. Garrigos, and S. Orlowski. Multidrug resistance transporter P-glycoprotein has distinct but interacting binding sites for cytotoxic drugs and reversing agents. Biochem. J. 333:351–358 (1998).
D. G. Bailey, J. Malcolm, O. Arnold, and J. D. Spence. Grape-fruit juice-drug interactions. Br. J. Clin. Pharmacol. 46:101–110 (1998).
V. A. Eagling, L. Profit, and D. J. Back. Inhibition of the CYP3A4-mediated metabolism and P-glycoprotein-mediated transport of the HIV-1 protease inhibitor saquinavir by grape-fruit juice components. Br. J. Clin. Pharmacol. 48:543–552 (1999).
C. A. Plouzek, H. P. Ciolino, R. Clarke, and G. C. Yeh. Inhibition of P-glycoprotein activity and reversal of multidrug resistance in vitro by rosemary extract. Eur. J. Cancer 35:1541–1545 (1999).
P. Knekt, R. Järvinen, and A. Reunanen. and J. Maatela. Flavo-noid intake and coronary mortality in Finland: a cohort study. BMJ 312:478–481 (1996).
L. Le Marchand, S. P. Murphy, J. H. Hankin, L. R. Wilkens, and L. N. Kolonel. Intake of flavonoids and lung cancer. J. Natl. Cancer Inst. 92:154–160 (2000).
C. Manach, C. Morand, O. Texier, M. L. Favier, G. Agullo, C. Demigne, F. Regerat, and C. Remesy. Quercetin metabolites in plasma of rats fed diets containing rutin or quercetin. J. Nutr. 125:1911–1922 (1995).
I. Erlund, T. Kosonen, G. Alfthan, J. Maenpaa, K. Perttunen, J. Kenraali, J. Parantainen, and A. Aro. Pharmacokinetics of quer-cetin from quercetin aglycone and rutin in healthy volunteers. Eur. J. Clin. Pharmacol. 56:545–553 (2000).
J. Vaidyanathan and T. Walle. Transport and metabolism of the tea flavonoid (-)-epicathecin by the human intestinal cell line Caco-2. Pharm. Res. 18:1420–1425 (2001).
K. J. Dabrowski and F. W. Sosulski. Composition of free and hydrolysable phenolic acids in defatted flours of ten oilseeds. J. Agric. Food Chem. 32:128–331 (1984).
R. K. Harris and W. J. Haggerty. Assays for potentially anticar-cinogenic phytochemicals in flax seed. Cereal Foods World 38:147–151 (1993).
T. Kraushofer and G. Sonntag. Determination of some phenolic compounds in flax seed and nettle roots by HPLC with coulo-metric electrode array detection. Eur. Food Res. Technol. 215:529–533 (2002).
W. H. Morrison and D. E. Akin. Chemical composition of components comprising bast tissue in flax. J. Agric. Food Chem. 49:2333–2338 (2001).
B. D. Oomah, G. Mazza, and E. O. Kenaschuk. Cyanogenic com-pounds in flaxseed. J. Agric. Food Chem. 40:1346–1348 (1992).
R. R. Paris and M. Paris. Sur les pigments anthocyaniques de la Salicaire (Lythrum salicariaL.). Compt. Rend 258:361–364 (1967).
M. T. Torrent Marti. Estudio farmacognóstico de Lythrum sali-caria L. Circ. Farm. 33:265–307 (1975).
X. Ma, C. Ji, Y. Wang, G. Zhang, and Y. Liu. New tannins from Lythrum salicariaL. J. Chin. Pharm. Sci. 5:225 (1996).
E. Fujita, Y. Saeki, M. Ochiai, and T. Inoue. Investigation of the neutral constituents of Lythrum salicaria. Bull. Inst. Chem. Res. Kyoto Univ. 50:327–331 (1972).
H. Pan and L. Lundgren. Phenolics from inner bark of Pinus Sylvestris. Phytochemistry 42:1185–1189 (1996).
J. Wilska-Jezka, J. Los, and M. Pawlak. Wild plant fruits as a source of catechins and proanthocyanidins. Bull. Liaison-Groupe Polyphenols 16:246–250 (1992).
S. Häkkinen and S. Auriola. High-performance liquid chroma-tography with electrospray ionization mass spectrometry and di-ode array ultraviolet detection in the identification of flavonol aglycones and glycosides in berries. J. Chromatogr. A. 829:91–100 (1998).
R. Huopalahti, E. P. Jarvenpaa, and K. Katina. A novel solid-phase extraction-HPLC method for the analysis of anthocyanin and organic acid composition of Finnish cranberry. J. Liq. Chrom. & Rel. Technol. 23:2695–2701 (2000).
Ø. M. Andersen. Chromatographic separation of anthocyanins in cowberry (lingon berry) Vaccinium vites-idaeaL. J. Food Sci. 50:1230–1232 (1985).
W. Henning. Flavonolglykoside der Erdbeeren (Fragaria x ananassaDuch.), Himbeeren (Rubus idaeusL.) und Brombeeren (Rubus fruticosusL.). Z. Lebensm. Unters. Forsch. 173:180–187 (1981).
I. C. W. Arts, B. van de Putte, and P. C. H. Hollman. Catechin contents of foods commonly consumed in the Netherlands. 1. Fruits, vegetables, staple foods and processed foods. J. Agric. Food Chem. 48:1746–1751 (2000).
I. P. Gerothanassis, V. Exarchou, V. Lagouri, A. Troganis, M. Tsimidou, and D. Boskou. Methodology for identification of phe-nolic acids in complex phenolic mixtures by high-resolution two-dimensional nuclear magnetic resonance. Application to metha-nolic extracts of two oregano species. J. Agric. Food Chem. 46:4185–4192 (1998).
M. Kosar, H. J. P. Dorman, O. Bachmayer, K. H. C. Basel, and R. Hiltunen. An improved on-line HPLC-DPPH* method for the screening of free radical scavenging compounds in water extracts of Lamiaceae plants. Chem. Nat. Comp. 39:161–166 (2003).
G. Zgórka and K. Glowniak. Variation of free phenolic acids in medicinal plants belonging to the Lamiaceaefamily. J. Pharm. Biomed. Anal. 26:79–87 (2001).
W. Zheng and S. Y. Wang. Antioxidant acitivity and phenolic compounds in selected herbs. J. Agric. Food Chem. 49:5165–5170 (2001).
P. B. Andrade, R. M. Seabra, P. Valentāo, and F. Areias. Simul-taneous determination of flavonoids, phenolic acids and couma-rins in seven medicinal species by HPLC/diode array detector. J. Liq. Chrom. & Rel. Technol. 21:2813–2820 (1998).
L. Pizzale, R. Bortolomeazzi, S. Vichi, E. Überegger, and L. S. Conte. Antioxidant activity of sage (Salvia officinalis and S fru-ticosa) and oregano (Origanum onitesand O indercedens) ex-tracts related to their phenolic compound content. J. Sci. Food Agric. 82:1645–1651 (2002).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Laitinen, L.A., Tammela, P.S.M., Galkin, A. et al. Effects of Extracts of Commonly Consumed Food Supplements and Food Fractions on the Permeability of Drugs Across Caco-2 Cell Monolayers. Pharm Res 21, 1904–1916 (2004). https://doi.org/10.1023/B:PHAM.0000045246.94064.ab
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000045246.94064.ab