Abstract
Purpose. The purpose of this study was to design sustained release system which provides basal insulin release over a week by one injection in diabetic animals. For an effective injectable formulation and controlled release of insulin, a water soluble biodegradable triblock copolymer of PLGA-PEG-PLGA was used.
Methods. For in vitro release, samples were analyzed by reversed-phase high performance liquid chromatography. Animal studies using ZDF rats have been conducted to demonstrate the bioactivity of the released insulin. Insulin formulation was injected subcutaneously. At designated times, the blood glucose levels and insulin levels of the ZDF rats were measured.
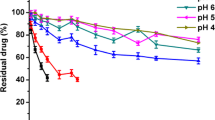
Results. The in vitro release of zinc-complexed insulin showed no initial burst and demonstrated constant release rate with the duration of 14 days. Constant steady state plasma levels of exogenous insulin were detected for nearly two weeks indicating constant rate of insulin release in vivo upon single subcutaneous injection.
Conclusions. We conclude that it is feasible to achieve basal insulin levels over a week by a single injection of ReGelTM formulation. This will provide various advantages, including depot formation without surgery, easy sterilization, straightforward drug loading, simple dose adjustment, system biocompatibility with no inflammatory reaction, and no requirement of using organic solvents.
Similar content being viewed by others
REFERENCES
A. H. Mokdad, E. S. Ford, B. A. Bowman, D. E. Nelson, M. M. Engelgau, F. Vinicor, and J. S. Marks. Diabetes trends in the U.S.: 1990-1998. Diabetes Care 23:1278-1283 (2000).
A. F. Amos, D. J. McCarty, and P. Zimmet. Diabet. Med. 14:S5-S85 (1997).
Diabetes. A cost and technology-driven disease management market. MedPro month, Medical Data International, CA, 1996.
R. Kawamori. Practical concept for insulin therapy. Intern. Med. 73:236-243 (1994).
M. Hinchcliffe and L. Illum. Intranasal insulin delivery and therapy. Adv. Drug Deliv. Rev. 35:199-234 (1999).
M. A. F. Goosen, Y. F. Leung, G. M. O'Shea, S. Chou, and A. M. Sun. Slow release of insulin from a biodegradable matrix implanted in diabetic rats. Diabetes 32:478-481 (1983).
S.-Y. Lin, L.-T. Ho, and H.-L. Chiou. Insulin controlled release microcapsules to prolong the hypoglycemic effect in diabetic rats. Biomater. Artif. Cells Artifi. Org. 16:815-828 (1988).
M. Takenaga, Y. Yamaguchi, A. Kitagawa, Y. Ogawa, Y. Mizushima, and R. Igarashi. A novel sustained-release formulation of insulin with dramatic reduction in initial rapid release. J. Cont. Rel. 79:81-91 (2002).
B. Jeong, Y. H. Bae, D. S. Lee, and S. W. Kim. Biodegradable block copolymers as injectable drug-delivery systems. Nature 388:860-862 (1997).
B. Jeong, D. S. Lee, J. Shon, Y. H. Bae, and S. W. Kim. Thermoreversible Gelation of Poly(Ethylene Oxide) Biodegradable Polyester Block Copolymers. J Polym Sci: Part A: Polym Chem. 37:751-780 (1999).
Y. J. Kim, S. Choi, J. J. Koh, M. Lee, K. S. Ko, and S. W. Kim. Controlled release of insulin from injectable biodegradable triblock copolymer. Pharm. Res. 18:548-550 (2001).
G. M. Zentner, R. Rathi, C. Shih, J. C. McRea, M.-H. Seo, H. Oh, B. G. Rhee, J. Mestecky, Z. Moldoveanu, M. Morgan, and S. Weitman. Biodegradable block copolymers for delivery of proteins and water-insoluble drugs. J. Cont. Rel. 72:203-215 (2001).
J. Brange and L. Langkær. Insulin structure and stability. In: Y. J. Wang, R. Pearlman (eds.), Stability and Characterization of Protein and Peptide Drugs: Case Histories, Plenum, New York, 1993, pp. 315-350.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, S., Kim, S.W. Controlled Release of Insulin from Injectable Biodegradable Triblock Copolymer Depot in ZDF Rats. Pharm Res 20, 2008–2010 (2003). https://doi.org/10.1023/B:PHAM.0000008050.99985.5c
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000008050.99985.5c