Abstract
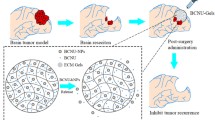
Minocycline, a tetracycline derivative, has been shown to inhibit tumor angiogenesis through inhibitory effects on matrix metalloproteinases. Previous studies have shown this agent to be effective against a rodent brain tumor model when delivered intracranially and to potentiate the efficacy of standard chemotherapeutic agents. In the present study, the in vivo efficacy of intracranial minocycline delivered by a biodegradable controlled-release polymer against rat intracranial 9L gliosarcoma was investigated to determine whether it potentiates the effects of systemic 1,3-bis (2-chloroethyl)-1-nitrosourea (BCNU). Minocycline was incorporated into the biodegradable polymer polyanhydride poly[bis(p-carboxyphenoxy)propane-sebacic acid] (pCPP:SA) at a ratio of 50:50 by weight. The release kinetics of minocycline from the polymer were assessed. For the efficacy studies, female Fischer 344 rats were implanted with 9L glioma. Treatment with minocycline delivered by the pCPP:SA polymer at the time of tumor implantation resulted in 100% survival in contrast to untreated control animals that died within 21 days. Treatment with the minocycline-polymer 5 days after tumor implantation provided only modest increases in survival. The combination of intracranial minocycline and systemic BCNU extended median survival by 82% compared to BCNU alone (p < 0.0001) and 200% compared to no treatment (p < 0.004). We conclude that local intracranial delivery of minocycline from biodegradable controlled-release polymers inhibits tumor growth and may have clinical utility when combined with a chemotherapeutic agent.
Similar content being viewed by others
References
Cheng SY, Huang HJ, Nagane M, Ji XD, Wang D, Shih CC, Arap W, Huang CM, Cavenee WK: Suppression of glioblastoma angiogenicity and tumorigenicity by inhibition of endogenous expression of vascular endothelial growth factor. Proc Natl Acad Sci USA 93: 8502–8507, 1996
Wesseling P, Ruiter DJ, Burger PC: Angiogenesis in brain tumors; pathobiological and clinical aspects. J Neuro-Oncol 32: 253–265, 1997
Sorensen DR, Read TA, Porwol T, Olsen BR, Timpl R, Sasaki T, Iversen PO, Benestad HB, Sim BK, Bjerkvig R: Endostatin reduces vascularization, blood flow, and growth in a rat gliosarcoma. Neuro-Oncology 4: 1–8, 2002
Short SC, Traish D, Dowe A, Hines F, Gore M, Brada M: Thalidomide as an anti-angiogenic agent in relapsed gliomas. J Neuro-Oncol 51: 41–45, 2001
Mentlein R, Eichler O, Forstreuter F, Held-Feindt J: Somatostatin inhibits the production of vascular endothelial growth factor in human glioma cells. Int J Cancer 92: 545–550, 2001
Sipos EP, Brem H: Local anti-angiogenic brain tumor therapies. J Neuro-Oncol 50: 181–188, 2000
Puduvalli VK, Sawaya R: Antiangiogenesis – therapeutic strategies and clinical implications for brain tumors. J Neuro-Oncol 50: 189–200, 2000
Kirsch M, Schackert G, Black PM: Anti-angiogenic treatment strategies for malignant brain tumors. J Neuro-Oncol 50: 149–163, 2000
Folkman J: Tumor angiogenesis: therapeutic implications. N Engl J Med 285: 1182–1186, 1971
Folkman J, Merler E, Abernathy C, Williams G: Isolation of a tumor factor responsible for angiogenesis. J Exp Med 133: 275–288, 1971
Folkman J: Tumor angiogenesis. In: Holland J, Frei E, Bast R et al. (eds) Cancer Medicine, 3rd edn. Lea & Febiger, Philadelphia, 1993, 153–170
Folkman J: What is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 82: 4–6, 1990
Folkman J: Angiogenic factors. Science 235: 442–447, 1987
Folkman J: Antiangiogenesis. In: DeVita VT, Hellman S, Rosenberg SA (eds) Biologic Therapy of Cancer. JP Lippincott, Philadelphia, 1991, pp 743–753
Brem S, Cotran R, Folkman J: Tumor angiogenesis: a quantitative method for histologic grading. J Natl Cancer Inst 48: 347–356, 1972
Tamargo RJ, Bok RA, Brem H: Angiogenesis inhibition by minocycline. Cancer Res 51: 672–675, 1991
Sotomayor EA, Teicher BA, Schwartz GN, Holden SA, Menon K, Herman TS, Frei E: Minocycline in combination with chemotherapy or radiological therapy in vitro and in vivo. Cancer Chemother Pharmacol 30: 377–384, 1992
Teicher BA, Sotomayor EA, Huang ZD: Antiangiogenic agents potentiate cytotoxic cancer therapies against primary and metastatic disease. Cancer Res 52: 6702–6704, 1992
Parangi S, O'Reilly M, Christofori G, Holmgren L, Grosfeld J, Folkman J, Hanahan D: Antiangiogenic therapy of transgenic mice impairs de novo tumor growth. Proc Natl Acad Sci USA 93: 2002–2007, 1996
Sasamura H, Takahashi A, Miyao N, Yanase M, Masumori N, Kitamura H, Itoh N, Tsukamoto T: Inhibitory effect on expression of angiogenic factors by antiangiogenic agents in renal cell carcinoma. Br J Cancer 86: 768–773, 2002
Teicher BA, Holden SA, Ara G, Northey D: Response of the FSaII fibrosarcoma to antiangiogenic modulators plus cytotoxic agents. Anticancer Res 13: 2101–2106, 1993
Guerin C, Laterra J, Masnyk T, Golub LM, Brem H: Selective endothelial growth inhibition by tetracyclines that inhibit collagenase. Biochem Biophys Res Commun 188: 740–745, 1992
Greenwald RA, Moak SA, Ramamurthy NS, Golub LM: Tetracyclines suppress matrix metalloproteinase activity in adjuvant arthritis and in combination with flurbiprofen, ameliorate bone damage. J Rheumatol 19: 927–938, 1992
Sadowski T, Steinmeyer J: Effects of tetracyclines on the production of matrix metalloproteinases and plasminogen activators as well as of their natural inhibitors, tissue inhibitor of metalloproteinases-1 and plasminogen activator inhibitor-1. Inflamm Res 50: 175–182, 2001
Kroon AM, Dontje BHJ, Holtrop M, Van den Bogert C: The mitochondrial genetic system as a target for chemotherapy: tetracyclines as cytostatics. Cancer Lett 25: 33–40, 1984
Weingart JD, Sipos EP, Brem H: The role of minocycline in the treatment of intracranial 9L glioma. J Neurosurg 82: 635–640, 1995
Grossman SA, Reinhard C, Colvin OM, Chasin M, Brundrett R, Tamargo RJ, Brem H: The intracerebral distribution of BCNU delivered by surgically implanted biodegradable polymers. J Neurosurg 76: 640–647, 1992
Tamargo RJ, Myseros JS, Epstein JI, Yang MB, Chasin M, Brem H: Interstitial chemotherapy of the 9L gliosarcoma: controlled release polymers for drug delivery in the brain. Cancer Res 53: 329–333, 1993
Yang MB, Tamargo RJ, Brem H: Controlled delivery of 1,3-bis (2-chloroethyl)-1-nitrosurea from ethylene-vinyl acetate copolymer. Cancer Res 48: 5103–5107, 1989
Langer R, Folkman J: Polymers for the sustained release of proteins and other macromolecules. Nature 263: 797–800, 1976
Brem H, Tamargo RJ, Olivi A, Pinn M, Weingart JD, Wharam M, Epstein JI: Biodegradable polymers for controlled delivery of chemotherapy with and without radiation therapy in the monkey brain. J Neurosurg 80: 283–290, 1994
Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, Black K, Sisti M, Brem S, Mohr G: Placebocontrolled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet 345: 1008–1012, 1995
Valtonen S, Timonen U, Toivanen P, Kalimo H, Kivipelto L, Heiskanen O, Unsgaard G, Kuurne T: Interstitial chemotherapy with carmustine-loaded polymers for highgrade gliomas: a randomized double-blind study. Neurosurgery 41: 44–48, 1997
van den Bogert C, van Kernebeek G, de Leij L, Kroon AM: Inhibition of mitochondrial protein synthesis leads to proliferation arrest in the G1-phase of the cell cycle. Cancer Lett 32: 41–51, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Frazier, J.L., Wang, P.P., Case, D. et al. Local Delivery of Minocycline and Systemic BCNU have Synergistic Activity in the Treatment of Intracranial Glioma. J Neurooncol 64, 203–209 (2003). https://doi.org/10.1023/A:1025695423097
Issue Date:
DOI: https://doi.org/10.1023/A:1025695423097