Abstract
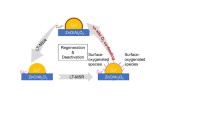
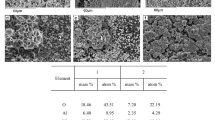
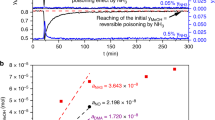
The reforming of methanol is acknowledged as a convenient means to generate hydrogen for a PEFC due to the low temperature (<280 °C) at which the reaction occurs and the low CO content of the reformate. However, the catalyst is prone to deactivation in the upper range of its operating temperatures. This paper presents a method of analysis in which relative changes in site concentrations are deduced from a series of long-duration rate measurements. The sites involved in the reforming reaction are shown to evolve independently of each other. In particular, concentration of sites for hydrogen adsorption decline at a greater rate than those responsible for the adsorption of oxygenated species. A key process in the deactivation of the catalyst is the decrease in its capacity to adsorb and dissociate hydrogen.
Similar content being viewed by others
References
J.C. Amphlett, R.F. Mann, B.A. Peppley and C.P. Thurgood, Catalyst Deactivation (2001), Vol. 139, J. Spivey (ed.) 950 pp.
B.A. Peppley, J.C. Amphlett, L.M. Kearns and R.F. Mann, Appl. Catal. A 179 (1999) 21.
B.A. Peppley, J.C. Amphlett, L.M. Kearns and R.F. Mann, Appl. Catal. A 179 (1999) 31.
J. Skrzypek, J. Sloczynski and S. Ledakowicz, Methanol Synthesis (Polish Scientific Publishers, Warsaw, 1994).
K.C. Waugh, Catal. Today 15 (1992) 51.
W.C. Conner and J.L. Falconer, Chem. Rev. 95 (1995) 759.
C.V. Ovesen, P. Stolze, J.K. Norskov and C.T. Campbell, J. Catal. 134 (1992) 445.
J. Hoogshagen and P.J. Zwietering, J. Chem. Phys. 21 (1953) 2224.
G.C. Chinchen, R.H. Logan and M.S. Spencer, Appl. Catal. 12 (1984) 89.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thurgood, C., Amphlett, J., Mann, R. et al. Deactivation of Cu/ZnO/Al2O3 Catalyst: Evolution of Site Concentrations with Time. Topics in Catalysis 22, 253–259 (2003). https://doi.org/10.1023/A:1023680121028
Issue Date:
DOI: https://doi.org/10.1023/A:1023680121028