Abstract
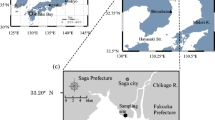
The chemical compositions of ground water and organic matter in sediments were investigated at a sandy shore of Tokyo Bay, Japan to determine the fate of ground water NO3 −. On the basis of Cl− distribution in ground water, the beach was classified into freshwater (FR)-, transition (TR)-, and seawater (SW)-zones from the land toward the shoreline. The NO3 − and N2O did not behave conservatively with respect to Cl− during subsurface mixing of freshwater and seawater, suggesting NO3 − consumption and N2O production in the TR-zone. Absence of beach vegetation indicated that NO3 − assimilation by higher plants was not as important as NO3 − sink. Low NH4 + concentrations in ground water revealed little reduction of NO3 − to NH4 +. These facts implied that microbial denitrification and assimilation were the likely sinks for ground water NO3 −. The potential activity and number of denitrifiers in water-saturated sediment were highest in the low-chlorinity part of the TR-zone. The location of the highest potential denitrification activity (DN-zone) overlapped with that of the highest NO3 − concentration. The C/N ratio and carbon isotope ratio (δ13C) of organic matter in sediment (< 100 -μm) varied from 12.0 to 22.5 and from −22.5 to −25.5‰, respectively. The δ13C value was inversely related to the C/N ratio (r 2 = 0.968, n = 11), which was explained by the mixing of organic matters of terrestrial and marine origins. In the DN-zone, the fine sediments were rich in organic matters with high C/N ratios and low δ13C values, implying that dissolved organic matters of terrestrial origin might have been immobilized under slightly saline conditions. A concurrent supply of NO3 − and organic matter to the TR-zone by ground water discharge probably generates favorable conditions for denitrifiers. Ground water NO3 − discharged to the beach is thus partially denitrified and fixed as microbial biomass before it enters the sea. Further studies are necessary to determine the relative contribution of these processes for NO3 − removal.
Similar content being viewed by others
References
APHA-AWWA-WPCF 1980. Standard methods for the examination of water and wastewater 15th edn.
Brock T.D., Lee D.R., Janes D. and Winek D. 1982. Groundwater seepage as a nutrient source to a drainage lake; Lake Mendota, Wisconsin. Water Res. 16: 1255-1263.
Brown A.C. and MacLachlen A. 1990. Ecology of Sandy Shores. Elsevier, 328pp.
Capone D.G. and Bautista M.F. 1985. A groundwater source of nitrate in nearshore marine sediments. Nature 313: 214-216.
Cherry J.A., Giliham R.W., Anderson E.G. and Johnson P.E. 1983. Migration of contaminants groundwater at a landfill: A case study 2. Groundwater monitoring devices. J. Hydrol. 63: 31-49.
Christensen J.P., Murray J.M., Devol A. and Codispoti L.A. 1987. Denitrification in continental shelf sediment has major impact on the oceanic nitrogen budget. Global Biogeochem. Cycles 1: 97-116.
Cifuentes L.A., Sharp J.H. and Vogel M.L. 1988. Stable carbon and nitrogen isotope biogeochemistry in the Delaware estuary. Limnol. Oceanogr. 33: 1102-1115.
Comly H.H. 1945. Cyanosis in infants caused by nitrate in well water. J. Am. Med. Assoc. 129: 112- 116.
Degens E.T., Guillard R.R.L., Sacket W.N. and Hellwbust J.A. 1986. Metabolic fractionation of carbon isotopes in marine plankton - I. Temperature and respiration experiments. Deep-Sea Res. 19: 1-9.
D'Elia C.F., Webb K.L. and Porter J.W. 1981. Nitrate-rich groundwater inputs to Discovery Bay, Jamaica: A significant source of N to local coral reefs? Bull. Mar. Sci. 31: 903-910.
Frape S.K. and Patterson P.J. 1981. Chemistry of pore-water and bottom sediments as indicators of seepage patterns in Perch Lake, Chalk River, Ontario. Limnol. Oceanogr. 26: 500-517.
Fry B. and Sherr E.B. 1984. ?13C measurements as indicators of carbon flow in marine and freshwater ecosystems. Contributions to Marine Science 27: 13-27.
Goolsby D.A. 2000. Mississippi Basin nitrogen flux believed to cause Gulf hypoxia. EOS 81: 321-327.
Giblin A.E. and Gaines A.G. 1990. Nitrogen inputs to a marine embayment: The importance of groundwater. Biogeochemistry 10: 309-328.
Hedges J.I., Clark W.A., Quay P.D., Richey J.E., Devol A.H. and Santos U.D.M. 1986. Compositions and fluxes of particulate organic material in the Amazon River. Limnol. Oceanogr. 31: 717-738.
Hynes R.K. and Knowles R. 1984. Production of nitrous oxide by Nitrosomonas europaea: effects of acetylene, pH and oxygen. Can. J. Microbiol. 30: 1397-1404.
Johannes R.E. 1980. The ecological significance of the submarine discharge of groundwater. Mar. Ecol. Prog. Ser. 3: 365-373.
Knowles R. 1982. Denitrification. Microbial. Rev. 46: 43-70.
Lee D.R., Cherry J.A. and Pickens J.F. 1980. Ground-water transport of a slat tracer through a sandy lake bed. Limnol. Oceanogr. 25: 45-61.
Lewis J.B. 1987. Measurements of groundwater seepage flux onto a coral reef: Spatial and temporal variations. Limnol. Oceanogr. 32: 1165-1169.
Matson E.A. and Brinston M.M. 1990. Stable carbon isotopes and the C:N ratio in the estuaries of the Pamlico and Neuse Rivers, North Carolina. Limnol. Oceanogr. 35: 1290-1300.
Matson E.A. 1993. Nutrient flux through soils and aquifers to the coastal zone of Guam (Mariana Islands). Limnol. Oceanogr. 38: 361-371.
Minagawa M., Winter D.A. and Kaplan I.R. 1994. Comparison of Kjeldahl and combustion methods for measurement of nitrogen stable isotope ratios in organic matter. Anal. Chem. 56: 1859-1861.
Murphy E.M., Davis S.N., Long A., Donahue D. and Jull A.J.T. 1989. Characterization and isotopic composition of organic and inorganic carbon in the Milk River Aquifer. Wat. Resour. Res. 25: 1893- 1905.
Ogawa H., Aoki N., Kon I. and Ogura N. 1994. Stable carbon isotope ratio of suspended particulate and sedimentary organic matter during the summer blooming in Tokyo Bay. Chikyu-Kagaku (Geochemistry) 28: 21-36.
Ogawa N. and Ogura N. 1997. Dynamics of particulate organic matter in the Tamagawa Estuary and inner Tokyo Bay. Estuarine, Coastal, Shelf Sci. 44: 263-273.
Ogura N., Kimura K., Sekigawa T., Yamada K. and Minagawa M. 1986. Stable isotope ratios of particulate organic carbon in surface water at inner part of Tokyo Bay. Chikyu-Kagaku (Geochemistry) 20: 13-19.
Paerl H.W. 1997. Coastal eutrification and harmful algal blooms: Importance of atmospheric deposition and groundwater as “new” nitrogen and other nutrient sources, Limnol. Oceanogr. 42: 1154-1156.
Power J.F. and Schepers J.S. 1989. Nitrate contamination of groundwater in North America. Agriculture, Ecosystem and Environment 26: 165-287.
Rosswall T. 1983. The nitrogen cycle. In: Bolin B. and Cook R.B. (eds), The Major Biogeochemical Cycles and Their Interactions. SCOPE 21. John Wiley and Sons, New York, pp. 46-50.
Redfield B.C. 1958. The biological control of chemical factors in the environment. Am. Sci. 46: 205- 221.
Schiff S.L., Aravena R., Trumbore S.E. and Dillon P.J. 1990. Dissolved organic carbon cycling in forested watersheds: A carbon isotope approach. Wat. Res. Res. 26: 2949-2957.
Seitzinger S.P. 1988. Denitrification in freshwater and coastal marine ecosystems: Ecological and geochemical significance. Limnol. Oceanogr. 33: 702-724.
Sewell P.L. 1982. Urban groundwater as a possible nutrient source for an estuarine benthic algal bloom. Est. Coastal Shelf Sci. 15: 569-576.
Simmons G.M. Jr 1992. Importance of submarine groundwater discharge (SGWD) and seawater cycling to material flux across sediment/water interfaces in marine environments. Mar. Ecol. Prog. Ser. 84: 173-184.
Slater J.M. and Capone D.G. 1987. Denitrification in aquifer soil and nearshore marine sediments in-fluenced by groundwater nitrate. Appl. Environ. Microbiol. 53: 1292-1297.
Smith R.L. and Duff J.H. 1988. Denitrification in a sand and gravel aquifer. Appl. Environ. Microbiol. 54: 1071-1078.
Smith R.L., Howes B.L. and Duff J.H. 1991. Denitrification in nitrate-contaminated groundwater: Occurrence in steep vertical geochemical gradients. Geochim. Cosmochim. Acta 55: 1815-1825.
Solorzano L. 1969. Determination of ammonia in natural waters by the phenolhypochlorite method. Limnol. Oceanogr. 14: 799-801.
Strebel O., Duynisveld W.H.M. and Bottcher J. 1989. Nitrate pollution of groundwater in western Europe. Agriculture, Ecosystem and Environment 26: 189-214.
Suzumura M., Ueda S. and Sumi E. 2000. Control of phosphate concentration through adsorption and desorption processes in groundwater and seawater mixing at sandy beaches in Tokyo Bay, Japan. J. Oceanogr. 56: 667-673.
Takahashi K., Wada E. and Sakamoto M. 1992. Carbon isotope ratio and photosynthetic activity of phytoplankton in the eutrophic Mikawa Bay, Japan. Ecol. Res. 7: 355-361.
Tan F.C. and Strain P.M. 1979. Organic carbon isotope ratios in recent sediments in the St. Lawrence Estuary and the Gulf of St. Lawrence. Estuarine, Coastal Mar. Sci. 8: 213-225.
Valiela I., Teal J.M., Volkman S., Shafer D. and Carpenter E.J. 1978. Nutrient and particulate fluxes in a salt marsh ecosystem: Tidal exchanges and inputs by precipitation and groundwater. Limnol. Oceanogr. 23: 798-812.
Virgil J., Hynes W.S. and Kaiser L. 1965. Nitrates in municipal water supply cause methemoglobinemia in infants. Public Health Report 80: 1119-1121.
Weiss R.E. and Price B.A. 1980. Nitrous oxide solubility in water and seawater. Mar. Chem. 8: 347- 359.
Wood E.D., Armstrong F.A. and Richards F.A. 1967. Determination of nitrate in seawater by cadmiumcopper reduction to nitrite. J. Mar. Biol. Assoc. UK. 47: 23-31.
Yoshinari T. and Knowles R. 1976. Acetylene inhibition of nitrous oxide reduction by denitrifying bacteria. Biochem. Biophys. Res. Commun. 69: 705-710.
Zimmermann C.F., Montgomery J.R. and Carlson P.R. 1985. Variability of dissolved reactive phosphate flux rates in nearshore estuarine sediments: Effects of groundwater flow. Estuaries 8: 228-236.
Rights and permissions
About this article
Cite this article
Ueda, S., Go, CS.U., Suzumura, M. et al. Denitrification in a seashore sandy deposit influenced by groundwater discharge. Biogeochemistry 63, 187–205 (2003). https://doi.org/10.1023/A:1023350227883
Issue Date:
DOI: https://doi.org/10.1023/A:1023350227883