Abstract
Purpose. The objective of this study was to evaluate the extent and mechanism of uptake of fluorescent chitosan nanoparticles by the A549 cells, a human cell line derived from the respiratory epithelium.
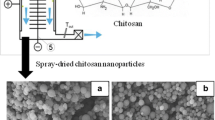
Methods. Covalent conjugation with fluorescein-5-isothiocyanate yielded stably labeled chitosan molecules, which were successfully formulated into nanoparticles by ionotropic gelation. Uptake of fluorescein-5-isothiocyanate-chitosan nanoparticles and chitosan molecules by confluent A549 cells was quantified by fluorometry.
Results. Cellular uptake of chitosan nanoparticles was concentration and temperature dependent, having Km and Vmax of 3.84 μM and 58.14 μg/mg protein/h, respectively. Uptake of chitosan nanoparticles was up to 1.8-fold higher than that of chitosan molecules alone and was not inhibited by excess unlabeled chitosan molecules. Hyperosmolarity, chlorpromazine and K+ depletion inhibited by 65, 34, and 54%, respectively, the uptake of chitosan nanoparticles at 37°C, but filipin had no influence on the uptake. Confocal imaging confirmed the internalization of the chitosan nanoparticles by the A549 cells at 37°C.
Conclusions. Formulation of chitosan into nanoparticles significantly improved its uptake by the A549 cells. Internalization of chitosan nanoparticles by the cells seems to occur predominantly by adsorptive endocytosis initiated by nonspecific interactions between nanoparticles and cell membranes, and was in part mediated by clathrin-mediated process.
Similar content being viewed by others
REFERENCES
N. G. M. Schipper, S. Olsson, J. A. Hoogstraate, A. G. deBoer, K. M. Vårum, and P. Artursson. Chitosans as absorption enhancers for poorly absorbable drugs 2: Mechanism of absorption enhancement. Pharm.Res. 14:923-929 (1997).
N. G. M. Schipper, K. M. Vårum, P. Stenberg, G. Ocklind, H. Lennernäs, and P. Artursson. Chitosans as absorption enhancers of poorly absorbable drugs. 3: Influence of mucus on absorption enhancement. Eur.J.Pharm.Sci. 8:335-343 (1999).
A. F. Kotzé, H. L. Lueβen, A. G. deBoer, J. C. Verhoef, and H. E. Junginger. Chitosan for enhanced intestinal permeability: Prospects for derivatives soluble in neutral and basic environments. Eur.J.Pharm.Sci. 7:145-151 (1998).
H.-Q. Mao, K. Roy, V. L. Troung-Le, and K. A. Janes, K. Y. Lin, Y. Wang, J. T. August, and K. W. Leong. Chitosan-DNA nanoparticles as gene carriers: Synthesis, characterization and transfection efficiency. J.Control.Release 70:399-421 (2001).
L. Illum, I. Jabbal-Gill, M. Hinchcliffe, A. N. Fisher, and S. S. Davis. Chitosan as a novel nasal delivery system for vaccines. Adv.Drug Deliv.Rev. 51:81-96 (2001).
K. A. Janes, M. P. Fresneau, A. Marazuela, A. Fabra, and M. J. Alonso. Chitosan nanoparticles as delivery systems for doxorubicin. J.Control.Release 73:255-267 (2001).
T. Jung, W. Kamm, A. Breitenbach, E. Kaiserling, J. X. Xiao, and T. Kissel. Biodegradable nanoparticles for oral delivery of peptides: Is there a role for polymers to affect mucosal uptake? Eur.J.Pharm.Biopharm. 50:147-160 (2000).
F. Delie. Evaluation of nano-and microparticle uptake by the gastrointestinal tract. Adv.Drug Deliv.Rev. 34:221-223 (1998).
K. Suh, B. Jeong, F. Liu, and S. W. Kim. Cellular uptake study of biodegradable nanoparticles in muscular smooth cells. Pharm.Res. 12:1495-1498 (1998).
B. Q. Roula and M. A. Mansoor. Synthesis of a fluorescent chitosan derivative and its application for the study of chitosanmucin interactions. Carbohydr.Polym. 38:99-107 (1999).
O. Hiraku and M. Yoshiharu. Biodegradation and distribution of water-soluble chitosan in mice. Biomaterials 20:175-182 (1999).
W. Wang, S. Q. Bo, S. Q. Li, and W. Qin. Determination of the Mark-Houwink equation for chitosans with different degrees of deacetylation. Int.J.Biol.Macromol. 13:281-285 (1991).
S. C. Tan, E. Khor, T. K. Tan, and S. M. Wong. The degree of deacetylation of chitosan: Advocating the first derivative UV-spectrophotometry method of determination. Talanta 45:713-719 (1998).
P. Calvo, C. Remuñán-Lòpez, J. L. Vila-Jato, and M. J. Alonso. Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J.Appl.Polymer Sci. 63:125-132 (1997).
W. Lutz and R. Kumar. Hypertonic sucrose treatment enhances second messenger accumulation in vasopressin-sensitive cells. Am.J.Physiol. 264:F228-F233 (1993).
G. G. Pietra, L. Johns, W. Byrnes, and S. Villaschi. Inhibition of adsorptive endocytosis and transcytosis in pulmonary microvessels. Lab.Invest. 59:683-690 (1988).
R. Miñana, J. M. Duran, M. Tomas, J. Renau-Piqueras, and C. Guerri. Neural cell adhesion molecule is endocytosed via a clathrin-dependent pathway. Eur.J.Neurosci. 13:749-756 (2001).
I. H. Madshus, K. Sandvig, S. Olsnes, and B. V. Deurs. Effect of reduced endocytosis induced by hypotonic shock and potassium depletion on the infection of Hep 2 cells by picornaviruses. J.Cell Physiol. 131:14-22 (1987).
G. Daukas and S. H. Zigmond. Inhibition of receptor-mediated but not fluid-phase endocytosis in polymorphonuclear leukocytes. J.Cell Biol. 101:1673-1679 (1985).
P. A. Orlandi and P. H. Fishman. Filipin-dependent inhibition of cholera toxin: Evidence for toxin internalization and activation through caveolae-like domains. J.Cell Biol. 141:905-915 (1998).
C. M. Lehr. The transcytosis approach. In A.G. de Boer (ed.), Drug Absorption Enhancement-Concepts, Possibilities, Limitations and Trends, Harwood Academic, Switzerland, 1994, pp. 325-366.
B. J. Nichols and J. Lippincott-Schwartz. Endocytosis without clathrin coats. Trends Cell Biol. 11:406-412 (2001).
J. E. Schnitzer. Caveolae: From basic trafficking mechanisms to targeting transcytosis for tissue-specific drug and gene delivery in vivo. Adv.Drug Deliv.Rev 49:265-280 (2001).
L. H. Wang, K. G. Rothberg, and R. G. Anderson. Mis-assembly of clathrin lattices on endosomes reveals a regulatory switch for coated pit formation. J.Cell Biol. 123:1107-1117 (1993).
C. Racine, M. Belanger, H. Hirabayashi, M. Boucher, J. Chakir, and J. Couet. Reduction of caveolin 1 gene expression in lung carcinoma cell lines. Biochem.Biophys.Res.Commun. 255:580-586 (1999).
N. G. M. Schipper. K. M Vårum and P. Artursson. Chitosans as absorption enhancers for poorly absorbable drugs. 1: Influence of molecular weight and degree of acetylation on drug transport across human intestinal epithelial (Caco-2) cells. Pharm.Res. 13:1686-1692 (1996).
L. Illum, N. F. Farraj, and S. S. Davis. Chitosan as a novel nasal delivery system for peptide drugs. Pharm.Res. 11:1186-1189 (1994).
N. Haffejee, J. Du Plessis, D. G. Muller, C. Schultz, A. F. Kotze, and C. Goosen. Intranasal toxicity of selected absorption enhancers. Pharmazie 56:882-888 (2001).
J. A. Swanson and C. Watts. Macropinocytosis. Trends Cell Biol. 5:424-428 (1995).
R. C. Stearns, J. D. Paulauskis, and J. J. Godleski. Endocytosis of ultrafine particles by A549 cells. Am.J.Respir.Cell Mol.Biol. 24:108-115 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, M., Ma, Z., Khor, E. et al. Uptake of FITC-Chitosan Nanoparticles by A549 Cells. Pharm Res 19, 1488–1494 (2002). https://doi.org/10.1023/A:1020404615898
Issue Date:
DOI: https://doi.org/10.1023/A:1020404615898