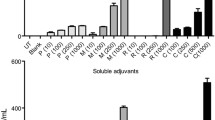
Abstract
The use of adjuvants for immunopotentiation has been investigated since the 1920s and a number of comprehensive reviews and monographs have been published on this subject. A recent trend in immunopotentiation has been the use of delivery systems which allow for sustained or controlled release of antigens and which induce prolonged immunity following a single dose. This concept has been termed either single-step or single-shot immunization. The delivery system has been modulated to potentiate the immune response either by delivering the antigen (and perhaps an adjuvant or adjuvants) either over a prolonged period of time or in a predetermined sequence or by incorporating substances with immunoadjuvant properties (e.g., lecithin and certain biodegradable polymers) as carriers within the delivery system. This Review focuses on the progress made in the design of delivery systems for immunopotentiation. Particular emphasis is given to delivery systems designed to achieve single-step immunization.
Similar content being viewed by others
REFERENCES
D. J. Stewart, B. L. Clark, J. E. Peterson, D. A. Griffiths, E. F. Smith, and I. J. O'donnell. Effect of pilus dose and type of Freund's adjuvant on the antibody and protective responses of vaccinated sheep to Bacteroides nodosus. Res. Vet. Sci. 35:130–137 (1983).
S. Lukes. Dependency of resistance of mice immunised against Taenia crassiceps on the dose of soluble antigen. Folia Parasitol. 34:329–333 (1987).
T. W. F. Pay and P. J. Hingley. Correlation of 140S antigen dose with the serum neutralising antibody response and the level of protection induced in cattle by foot-and-mouth disease vaccines. Vaccine 5:60–64 (1987).
J.-G. Hu, A. Ide, T. Yokoyama, and T. Kitagawa. Studies on the optimal immunisation schedule of the mouse as an experimental animal. The effect of antigen dose and adjuvant type. Chem. Pharm. Bull. 37:3042–3046 (1989).
J.-G. Hu, T. Yokoyama, and T. Kitagawa. Studies on the optimal immunization schedule of experimental animals. V. The effects of the route of injection, the content of Mycobacteria in Freund's adjuvant and the emulsifying antigen. Chem. Pharm. Bull. 38:1961–1965 (1990).
D. L. Wise, D. J. Trantolo, R. T. Marino, and J. P. Kitchell. Opportunities and challenges in the design of implantable biodegradable polymeric systems for the delivery of antimicrobial agents and vaccines. Adv. Drug Deliv. Rev. 1:19–39 (1987).
I. Nakashima, F. Ohta, T. Kobayashi, O. Kato, and N. Kato. Effect of antigen doses and time intervals between antigen injections on secondary, tertiary and quaternary antibody responses. Immunology 26:443–454 (1974).
M. Murray, P. B. Robinson, C. Grierson, and R. A. Crawford. Immunisation against Nippostrongylus brasiliensis in the rat. Acta Trop. 36:297–322 (1979).
R. C. H. Lau. Detection of diphtheria toxin antibodies in human sera in New Zealand by ELISA. J. Hyg Cambr. 96:415–418 (1986).
J. A. Reynolds, D. G. Harrington, C. L. Crabbs, C. J. Peters, and N. R. Di Luzio. Adjuvant activity of a novel metabolizable lipid emulsion with inactivated viral vaccines. Infect. Immun. 28:937–943 (1980).
M. Brugh, H. D. Stone, and H. W. Lupton. Comparison of inactivated Newcastle disease viral vaccines containing different emulsion adjuvants. Am. J. Vet. Res. 44:72–75 (1983).
H. S. Warren, F. R. Vogel, and L. A. Chedid, Current status of immunological adjuvants. Annu. Rev. Immunol. 4:369–388 (1986).
B. A. Vanselow. The application of adjuvants to veterinary medicine. Vet. Bull. 57:881–896 (1987).
R. Edelman. Vaccine adjuvants. Rev. Infect. Dis. 2:370–383 (1980).
S. Wright and L. Huang. Antibody-directed liposomes as drug delivery vehicles. Adv. Drug Deliv. Rev. 3:343–389 (1989).
N. van Rooijen. Liposomes as carrier and immunoadjuvant of vaccine antigens. Adv. Biotechnol. Process. 13:255–279 (1990).
C. R. Alving. Liposomes as carriers of antigens and adjuvants. J. Immunol. Methods 140:1–13 (1991).
C. Langhein and J. F. E. Newman. Antibody response to bacterial antigens covalently bound to biodegradable polymerized serum albumin beads. J. Appl. Bacteriol. 63:443–448 (1987).
P. M. Callahan, A. L. Shorter, and S. L. Hem. The importance of surface charge in the optimisation of antigen-adjuvant interactions. Pharm. Res. 8:851–858 (1991).
G. Ramon. Procédés pour accroitre la production des antitoxines. Ann. Inst. Pasteur 40:1–10 (1926).
J. H. Eldridge, J. K. Staas, J. A. Meulbroek, T. R. Tice, and R. M. Gilley. Biodegradable and biocompatible poly (D,L-lactide-co-glycolide) microspheres as an adjuvant for staphylococcal enterotoxin B toxoid which enhances the level of toxinneutralizing antibodies. Infect. Immun. 59:2978–2986 (1991).
A. K. Lascelles, G. Eagleson, K. J. Beh, and D. L. Watson. Significance of Freund's adjuvant/antigen injection granuloma in the maintenance of serum antibody response. Vet. Immunol. Immunopathol. 22:15–27 (1989).
R. Hunter, M. Oslen, and S. Buynitzky. Adjuvant activity of non-ionic block copolymers. IV. Effect of molecular weight and formation on titre and isotype of antibody. Vaccine 9:250–255 (1991).
A. C. Allison and G. Gregoriadis. Liposomes as immunological adjuvants. Nature 252:252 (1974).
W. Wagner, K. Beckman, M. G. Edinger, S. Deodhar, and J. R. Battisto. Immunotherapy with allergen sequestered in liposomes. J. Allergy Clin. Immunol. 73:118 (abstr. 39) (1984).
N. Arora and S. V. Gangal. Allergen entrapped in liposomes reduce allergenicity and induce immunogenicity on repeated injections in mice. Int. Arch. Allergy Appl. Immunol. 91:22–29 (1990).
R. J. Genco, R. Linzer, and R. T. Evans. Effect of adjuvants on orally administered antigens. Ann. N.Y. Acad. Sci. 409:650–668 (1983).
N. Latif and B. K. Bachhawat. Liposomes in immunology. J. Biosci. 6:491–502 (1984).
C. R. Alving. Liposomes as carriers for vaccines. In M. J. Ostro (ed.), Liposomes; From Biophysics to Therapeutics, Marcel Dekker, New York, 1987, pp. 195–218.
G. Gregoriadis. Immunological adjuvants: A role for liposomes. Immunol. Today 11:89–97 (1990).
N. van Rooijen, R. van Nieuwmegen, N. Kors, and P. Eikelenboom. The development of specific antibody-containing cells in the spleen of rabbits during the secondary immune response against free or liposome-associated albumin antigen. Anat. Rec. 208:579–587 (1984).
D. T. O'Hagan, D. Rahman, J. P. McGee, H. Jeffery, M. C. Davies, P. Williams, S. S. Davies, and S. J. Challacombe. Biodegradable microparticles as controlled release antigen delivery systems. Immunology 73:239–242 (1991).
M. Singh, A. Singh, and G. P. Talwar. Controlled delivery of diphtheria toxoid using biodegradable poly (D,L-lactide) microcapsules. Pharm. Res. 8:958–961 (1991).
P. Artursson, P. Edman, and I. Sjöholm. Biodegradable microspheres. II. Immune response to a heterogeneous and an autologous protein entrapped in polyacryl starch microparticles. J. Pharmacol. Exp. Ther. 234:255–260 (1985).
J. B. Dewar, D. A. Hendry, and J. F. E. Newmann. Biodegradable serum albumin polymers for the sustained release of virus antigens in vaccines. S. Afr. Med. J. 65:564–565 (1984).
R. Langer and J. Folkman. Polymers for the sustained release of proteins and other macromolecules. Nature 263:797–800 (1976).
I. Preis and R. S. Langer. A single step immunization by sustained antigen release. J. Immunol. Methods 28:193–197 (1979).
R. Langer. Polymers for the sustained release of macromolecules: Their use in a single-step method of immunization. Methods Enzymol. 73:57–75 (1981).
J. Kohn, S. M. Niemi, E. C. Albert, J. C. Murphy, R. Langer, and J. G. Fox. Single-step immunisation using a controlled release, biodegradable polymer with sustained adjuvant activity. J. Immunol. Methods 95:31–38 (1986).
J. B. Kohn, R. S. Langer, S. M. Niemi, and J. G. Fox. Biodegradable polymeric drug delivery system with adjuvant activity. International Patent Publication No. WO 88/02262 (1988).
R. L. Beck. Mammal immunisation. U.S. Patent No. 4 919 929 (1990).
J. P. Opdebeeck and I. G. Tucker. A cholesterol implant used as a delivery system to immunize mice with bovine serum albumin. J. Control. Release 23:271–279 (1993).
M. Z. I. Khan, I. G. Tucker, and J. P. Opdebeeck. Cholesterol and lecithin implants for sustained release of antigen: Release in vitro and antibody response in mice. Int. J. Pharm. 76:161–170 (1991).
M. Z. I. Khan, I. G. Tucker, and J. P. Opdebeeck. Evaluation of cholesterol-lecithin implants for sustained delivery of antigen: Release in vivo and single-step immunisation of mice. Int. J. Pharm. 90:255–262 (1993).
P. Y. Wang. Prolonged release of insulin by cholesterol-matrix implant. Diabetes 36:1068–1072 (1987).
P. Y. Wang. Implant preparations for delivery of bioactive materials. European Patent No. 0246 540 (1987).
L. F. Fries, D. M. Gordon, R. L. Richards, J. E. Egan, M. R. Hollingdale, M. Gross, C. Silverman, and C. R. Alving. Liposomal malaria vaccine in humans: A safe and potent adjuvant strategy. Proc. Natl. Acad. Sci. USA 89:358–362 (1992).
R. L. Richards, G. M. Swartz, Jr., C. Schultz, M. D. Hayre, G. S. Ward, W. R. Ballou, J. D. Chulay, W. T. Hockmeyer, S. L. Berman, and C. R. Alving. Immunogenicity of liposomal malaria sporozite antigen in monkeys: Adjuvant effects of aluminium hydroxide and non-pyrogenic liposomal lipid A. Vaccine 7:506–512 (1989).
S. Cohen, H. Bernstein, C. Hewes, M. Chow, and R. Langer. The pharmacokinetics of, and humoral responses to, antigen delivered by microencapsulated liposomes. Proc. Natl. Acad. Sci. USA 88:10440–10444 (1991).
T. M. S. Chang. Biodegradable semipermeable microcapsules containing enzymes, hormones, vaccines, and other biologicals. J. Bioeng. 1:25–32 (1976).
J. H. Eldridge, J. K. Staas, J. A. Meulbroek, J. R. McGhee, T. R. Tice, and R. M. Gilley. Biodegradable microspheres as a vaccine delivery system. Mol. Immunol. 28:287–294 (1991).
L. J. Silvestri and R. H. Pyle. Sustained release dosage forms. International Patent Publication No. WO 90/09166 (1990).
J. H. Eldridge, C. J. Hammond, J. A. Meulbroek, J. A. Staas, R. M. Gilley, and T. R. Tice. Controlled vaccine release in the gut-associated lymphoid tissues. I. Orally administered biodegradable microspheres target the Peyer's patches. J. Control. Release 11:205–214 (1990).
N. A. Mitchison. Dose, frequency and route of administration of antigen. In F. Borek (ed.), Immunogenicity, North-Holland, Amsterdam, 1972, pp. 87–111.
D. W. Dresser. Specific inhibition of antibody production. II. Paralysis induced in adult mice by small quantities of protein antigen. Immunology 5:378–388 (1962).
G. J. V. Nossal. Immunogenicity and immunopotentiation. In G. C. Woodrow and M. M. Levine (eds.), New Generation Vaccines, Marcel Dekker, New York, 1990, pp. 85–94.
E. Sercarz and A. H. Coons. Specific inhibition of antibody formation during immunological paralysis and unresponsiveness. Nature 184:1080–1082 (1959).
L. C. Sauerbronn Maia, N. M. Vaz, and E. M. Vaz. Effect of soluble antigen on IgE responses in the mouse. Int. Arch. Allergy 46:339–344 (1974).
A. K. Walduck and J. P. Opdebeeck. The effect of antigen delivery profile on the level, avidity and isotype of murine antibodies (submitted for publication) (1993).
A. A. Amkraut, S. I. Yum, and M. B. Rittenberg. Specific suppression of IgG plaque forming cells (PFC) by continuous administration of antigen from an implantable delivery system. Fed. Proc. 35:675 (abstr. 2584) (1976).
M. N. Abo-Shehada and I. V. Herbert. Investigation of effects of some immunoadjuvants and routes of antigen administration in raising antibodies in rabbits against metacestode cyst antigens of Taenia multiceps. J. Immunol. Methods 61:345–350 (1983).
P. N. Shek, B. Y. K. Yung, and N. Z. Stanacev. Comparison between multilamellar and unilamellar liposomes in enhancing antibody formation. Immunology 49:37–44 (1983).
P. N. Shek, B. Y. K. Yung, and N. Z. Stanacev. Physicochemical and immunological properties of albumin-associated dialkylether phosphatidylcholine liposomes. Biochim. Biophys. Acta 855:3–40 (1986).
D. Davis and G. Gregoriadis. Liposomes as adjuvants with immunopurified tetanus toxoid: Influence of liposomal characteristics. Immunology 61:229–234 (1987).
T. D. Heath, D. C. Edwards, and B. E. Ryman. The adjuvant properties of liposomes. Biochem. Soc. Trans. 4:129–133 (1976).
N. van Rooijen, and R. van Nieuwmegen. Liposomes in immunology: Multilamellar phosphatidylcholine liposomes as a simple, biodegradable and harmless adjuvant without any immunogenic activity of its own. Immunol. Commun. 9:243–256 (1980).
S. L. Snyder and W. E. Vannier. Immunologic response to protein immobilized on the surface of liposomes via covalent azobonding. Biochim. Biophys. Acta 772:288–294 (1984).
P. N. Shek and B. H. Sabiston. Immune response mediated by liposome-associated protein antigens. II. Comparison of the effectiveness of vesicle-entrapped and surface-associated antigen in immunopotentiation. Immunology 47:627–632 (1982).
P. Artursson, I.-L. Martensson, and I. Sjöholm. Biodegradable microspheres. III. Some immunological properties of polyacryl starch microparticles. J. Pharm. Sci. 75:697–701 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Khan, M.Z.I., Opdebeeck, J.P. & Tucker, I.G. Immunopotentiation and Delivery Systems for Antigens for Single-Step Immunization: Recent Trends and Progress. Pharm Res 11, 2–11 (1994). https://doi.org/10.1023/A:1018977107167
Issue Date:
DOI: https://doi.org/10.1023/A:1018977107167