Abstract
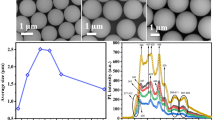
Formation mechanism of dye-doped silica particles less than 1 µm in diameters by the Stöber method was investigated. With increasing the content of dyes such as water-soluble porphyrin (TTMAPP) and laser dye (Nile blue), these dye-doped silica particles showed the similar tendency to the following characters: (1) Particle size increased, passed through the maximum at the dye content corresponds to the isoelectric point, and then followed by decrease; (2) Ratio of dimer and monomer of the incorporated dye decreased. In aqueous solution positively charged dyes showed good affinity with negatively charged SiO2 primary particles from the early stage of the formation. Zeta potential of doped-silica particles was measured against nominal dye content: from non-doped to highly doped particles, their surface charge changed from negative to positive. The similar influence caused by positively charged dyes on the particle formation was also observed by Ca2+ doping.
Similar content being viewed by others
References
E.J.A. Pope, Mat. Res. Soc. Symp. Proc. 372, 253 (1995).
S. Shibata, M. Yamane, K. Kamada, K. Ohta, K. Sasaki, and H. Masuhara, 8th Int. Workshop on Glasses and Ceramics from Gels, Faro, Portugal, 1995. J. Sol-Gel Science and Technology 8, 959 (1997).
D. Levy, M. Ocana, and C.J. Serna, J. Sol-Gel Sci. and Tech. 2, 761 (1994); M. Ocana, D. Levy, and C.J. Serna, J. Non-Cryst. Solids 147&148, 621 (1992).
E.J.A. Pope, Sol-Gel Optics II, SPIE 1758, 360 (1992).
S. Shibata, T. Taniguchi, T. Yano, A. Yasumori, and M. Yamane, J. Sol-Gel Sci. and Tech. 2, 755 (1994).
W. Stöber, A. Fink, and E. Bohn, J. Colloid and Interface Sci. 26, 62 (1968).
T. Shimohira and H. Ishijima, The Chem. Soc. Jpn. (9), 1503 (1981).
C.J. Brinker and G.W. Scherer, Particulate Sols and Gels, Sol-Gel Science (Academic Press, Inc., 1990), Chap. 4.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shibata, S., Taniguchi, T., Yano, T. et al. Formation of Water-Soluble Dye-Doped Silica Particles. Journal of Sol-Gel Science and Technology 10, 263–268 (1997). https://doi.org/10.1023/A:1018369200282
Issue Date:
DOI: https://doi.org/10.1023/A:1018369200282