Abstract
This paper is the first one of a research project aimed to find and optimize methods by which drug-excipient compatibility can be reliably and quickly assessed.
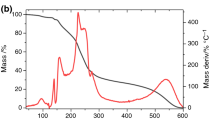
A number of experimental techniques (simultaneous TG-DSC, FT-IR spectroscopy, X-ray powder diffraction, scanning electron microscopy) have been used to investigate the compatibility between a novel tricyclic β-lactam antibiotic developed by GlaxoWellcome (now GlaxoSmithKline), GV118819x, and some commonly used excipients (poly(vinylpyrrolidone), magnesium stearate and α-lactose). Binary mixtures of two different compositions have been analyzed: drug:excipient=80:20 and 20:80 (mass/mass). Both qualitative and quantitative interaction indicators have been identified. It is shown that simultaneous thermal analysis is the best suited technique in the search of interaction indicators. With a proper selection of experimental conditions it is able to reveal the thermal changes brought about by the early stages of interaction, i.e. those occurring during the measurement on physical mixtures not previously annealed under stress conditions. Such an ability is discussed, in particular, with respect to the role of the water vapour, which has been found to be a critical parameter for all our systems.
Similar content being viewed by others
References
H. Nyqvist, Drug Dev. Ind. Pharm., 12 (1986) 953.
A. T. M. Serajuddin, A. B. Thakur, R. N. Ghoshal, M. G. Fakes, S. A. Ranadive, K. R. Morris and S. A. Varia, J. Pharm. Sci., 88 (1999) 696.
L. Gu, R. G. Strickley, L. Chi and Z. T. Chowan, Pharm. Res., 7 (1990) 379.
C. E. P. Malan, M. M. de Villiers and A. P. Lötter, Drug Dev. Ind. Pharm., 23 (1997) 533.
H. Jacobson and G. Reier, J. Pharm. Sci., 58 (1969) 631.
M. E. Brown, E. M. Antunes, B. D. Glass, M. Lebete and R. B. Walker, J. Therm. Anal. Cal., 56 (1999) 1317.
F. A. Chrazanowski, L. A. Ulissi, B. J. Fegely and A. C. Newman, Drug Dev. Ind. Pharm., 12 (1986) 783.
H. H. El-Shattawy, G. E. Peck and D. O. Kildsig, Drug Dev. Ind. Pharm., 8 (1982) 897.
S. A. Botha and A. P. Lötter, Drug Dev. Ind. Pharm., 16 (1990) 673.
Y. Matsunaga, R. Ohta, N. Bando, H. Yamada, H. Yuasa and Y. Kanaya, Chem. Pharm. Bull., 41 (1993) 720.
J. G. Kesavan and G. E. Peck, Drug Dev. Ind. Pharm., 22 (1996) 189.
R. Camarini, M. Panunzion, G. Bonanomi, D. Donati and A. Perboni, Tetrahedron Lett., 37 (1996) 2467.
B. Tamburini, A. Perboni, T. Rossi, D. Donati, D. Andreotti, G. Gaviraghi, R. Cerlesso and C. Bismara, European Patent Application EP 416, 953, 1991.
A. Perboni, B. Tamburini, T. Rossi, D. Donati, G. Gaviraghi and G. Tarzia, The Royal Society Chemistry, (1992) 21.
A. Marini, V. Berbenni, G. Bruni, C. Sinistri, A. Maggioni, A. Orlandi and M. Villa, J. Pharm. Sci., 89 (2000) 232.
C. F. Lerk, A. C. Andreae, A. H. de Boer, P. de Hoog, K. Kussendrager and J. Van Leverink, J. Pharm. Sci., 73 (1984) 856.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bruni, G., Amici, L., Berbenni, V. et al. Drug-Excipient Compatibility Studies. Search of interaction indicators. Journal of Thermal Analysis and Calorimetry 68, 561–573 (2002). https://doi.org/10.1023/A:1016052121973
Issue Date:
DOI: https://doi.org/10.1023/A:1016052121973