Abstract
Purpose. The effects of five different permeation enhancer systems on the transport properties of a peptidomimetic thrombin inhibitor, CRC 220, were investigated in monolayers of a human intestinal cell line (Caco-2).
Methods. The transepithelial transport rates and additionally the cytotoxic properties of these enhancers were characterized using the following tests: measurement of the transepithelial electrical resistance (TEER), the MTT-transformation, the protein content and the release of cytosolic lactate dehydrogenase (LDH), as well as FITC-phalloidin and propidium iodide staining.
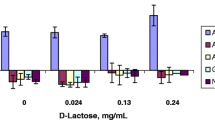
Results. All permeation enhancer systems showed concentration-dependent effects on cell permeability and toxicity. The most prominent effects on peptide transport were seen at the highest concentration (40 mM), yielding the rank order, NaTC > NaTC/Cholesterol > Solulan C24 > NaTC/Oleic acid > NaTC/PC18. Using the TEER after 120 min exposure as the most sensitive parameter describing cytotoxicity, the following order was obtained: Solulan C24 > NaTC > NaTC/ PC 18 = NaTC/Cholesterol > NaTC/Oleic acid > NaTC/PC. Generally, efficient enhancement of peptide transport was associated with a noticeable influence on cell viability under in-vitro conditions.
Conclusions. Taking into account permeation and cytotoxicity as a function of concentration, both NaTC at 15 mM and the mixed micellar system NaTC/oleic acid at 0.75 mM offer interesting enhancement properties, showing an 18-fold increase in CRC 220 transport rates. The effects on cell viability and cytotoxicity were comparatively low and of reversible nature.
Similar content being viewed by others
REFERENCES
P. L. Smith, D. A. Wall, C. H. Gochoco and G. Wilson. (D) Routes of delivery: Case studies. (5) Oral absorption of peptides and proteins. Adv. Drug Delivery Rev. 8:253–290 (1992).
V. H. L. Lee and A. Yamamoto. Penetration and enzymatic barriers to peptide and protein absorption. Adv. Drug Delivery Rev. 4:171–207 (1990).
S. Muranishi. Absorption enhancers. Critical Reviews in Therapeutic Drug Carrier Systems 7:1–33 (1990).
W. Stüber, R. Koschinsky, G. Dickneite and C. Kolar. Amidinophenylalanin-Derivate, Verfahren zu deren Herstellung, deren Verwendung und diese enthaltende Mittel als Antikoagulantien. EP. 0513543 (1992).
E. Walter, T. Kissel, M. Reers, G. Dickneite, D. Hoffmann and W. Stüber. Transepithelial transport properties of peptidomimetic thrombin inhibitors in monolayers of a human intestinal cell line (Caco-2) and their correlation to in vivo data. Pharm. Res. 12:360–365 (1995).
T. Ohtani, M. Murakami, A. Yamamoto, K. Takada and S. Muranishi. Effect of absorption enhancers on pulmonary absorption of fluorescein isothiocyanate dextrans with various molecular weights. Int. J. Pharm. 77:141–150 (1991).
U. Werner. Modelle der nasalen und intestinalen Mukosa für Transport-und Metabolismusuntersuchungen. PhD-thesis, Cuvillier Verlag Göttingen ISBN: 3-89588-403-0 (1995).
U. Werner and T. Kissel. Development of a human nasal epithelial cell culture model and its suitability for transport and metabolism studies under in-vitro conditions. Pharm. Res. 12:565–571 (1995).
C. S. Schasteen, M. G. Donovan and J. N. Cogburn. A novel in vitro screen to discover agents which increase the absorption of molecules across the intestinal epithelium. J. Control. Rel. 21:49–62 (1992).
O. H. Lowry, N. J. Rosenbrough, A. L. Farr and R. J. Randall. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193:265–275 (1951).
T. Mosmann. Rapid colorimetric assay for cellular growth and survial: Application to proliferation and cytotoxcity assays. J. Immunol. Methods 65:55–63 (1983).
B. J. Kroesen, G. Mesander, J. G. ter Haar, T. H. The and L. de Leij. Direct visualisation and quantification of cellular cytotoxicity using two colour fluorescence. J. Immunol. Meth. 156:47–52 (1992).
F. G. J. Poelma, J. J. Tukker and D. J. A. Crommelin. The role of bile salts in the intestinal absorption of drugs. Acta Pharm. Technol. 36:43–52 (1990).
E. K. Anderberg, C. Nyström and P. Artursson. Epithelial transport of drugs in cell culture. VII: Effects of pharmaceutical surfactant excipients and bile acids on transepithelial permeability in monolayers of human intestinal epithelial (Caco-2) cells. J. Pharm. Sci. 81:879–887 (1992).
A. L. M. Velardi, A. K. Groen, R. P. J. O. Elferink, R. van der Meer, G. Palasciano and G. N. J. Tytgat. Cell type-dependent effect of phospholipid and cholesterol on bile salt cytotoxicity. Gastroenterology 101:457–464 (1991).
M. Murakami, K. Takada and S. Muranishi. Further mechanistic study on intestinal absorption enhanced by unsaturated fatty acids: reversible effect by sulfhydryl modification. Biochim. Biophys. Acta 1117:83–89 (1992).
J. Drewe, G. Fricker, J. Vonderscher and C. Beglinger. Enteral absorption of octreotide: absorption enhancement by polyoxyethylene-24-cholesterol ether. Br. J. Pharmacol. 108:298–303 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Werner, U., Kissel, T. & Reers, M. Effects of Permeation Enhancers on the Transport of a Peptidomimetic Thrombin Inhibitor (CRC 220) in a Human Intestinal Cell Line (Caco-2). Pharm Res 13, 1219–1227 (1996). https://doi.org/10.1023/A:1016020505313
Issue Date:
DOI: https://doi.org/10.1023/A:1016020505313