Abstract
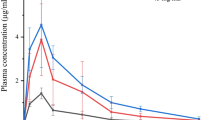
The absorption enhancing effect of α-, β-, and γ-cyclodextrin (CD), dimethyl-β-cyclodextrin (DMβCD), and hydroxypropyl-β-cyclodextrin (HPβCD) on intranasally administered insulin was investigated in rats. Coadministration of 5% (w/v) DMβCD to the insulin solution resulted in a high bioavailability, 108.9 ± 36.4% (mean ± SD, n = 6), compared to i.v. administration, and a strong decrease in blood glucose levels, to 25% of their initial values. Coadministration of 5% α-CD gave rise to an insulin bioavailability of 27.7 ± 11.5% (mean ± SD, n = 6) and a decrease in blood glucose to 50% of its initial value. The rate of insulin absorption and the concomitant hypoglycemic response were delayed for the α-CD-containing solution as compared to the DMβCD preparation. The other CDs, HPβCD (5%), β-CD (1.8%), and γ-CD (5%), did not have significant effects on nasal insulin absorption. DMβCD at a concentration of 5% (w/v) induces ciliostasis as measured on chicken embryo tracheal tissue in vitro, but this effect is reversible. In conclusion, DMβCD is a potent enhancer of nasal insulin absorption in rats.
Similar content being viewed by others
REFERENCE
M. J. Humphrey. The oral bioavailability of peptides and related drugs. In S. S. Davis, L. Illum, and E. Tomlinson (eds.), Delivery Systems for Peptide Drugs, Plenum Press, 1986, New York, pp. 139–151.
J. C. Verhoef, H. E. Boddé A. G. de Boer, J. A. Bouwstra, H. E. Junginger, F. W. H. M. Merkus, and D. D. Breimer. Transport of peptide and protein drugs across biological membranes. Eur. J. Drug Metab. Pharmacokin. 15:83–93 (1990).
Y. W. Chien, K. S. E. Su, and S. F. Chang. Nasal Systemic Drug Delivery, Marcel Dekker, New York, 1989.
S. Hirai, T. Yashiki, and H. Mima. Effects of surfactants on the nasal absorption of insulin in rats. Int. J. Pharm. 9:165–172 (1981).
G. S. Gordon, A. C. Moses, R. D. Silver, J. S. Flier, and M. C. Carey. Nasal absorption of insulin: Enhancement by hydrophobic bile salts. Proc. Natl. Acad. Sci. USA 82:7419–7423 (1985).
M. J. M. Deurloo, W. A. J. J. Hermens, S. G. Romeijn, J. Verhoef, and F. W. H. M. Merkus. Absorption enhancement of intranasally administered insulin by sodium taurodihydrofusidate (STDHF) in rabbits and rats. Pharm. Res. 6:853–856 (1989).
J. P. Longenecker, A. C. Moses, R. D. Silver, M. C. Carey, and E. J. Dubovi. Effects of sodium taurodihydrofusidate on nasal absorption of insulin in sheep. J. Pharm. Sci. 76:351–355 (1987).
M. Mishima, Y. Wakita, and M. Nakano. Studies on the promoting effects of medium chain fatty acid salts on the nasal absorption of insulin in rats. J. Pharmacobio-Dyn. 10:624–631 (1987).
L. Illum, N. F. Farraj, H. Critchley, B. R. Johansen, and S. S. Davis. Enhanced nasal absorption of insulin in rats using lysophosphatidylcholine. Int. J. Pharm. 57:49–54 (1989).
R. D. Ennis, L. Borden, and W. A. Lee. The effects of permeation enhancers on the surface morphology of the rat nasal mucosa: A scanning electron microscopy study. Pharm. Res. 7:468–475 (1990).
W. A. J. J. Hermens and F. W. H. M. Merkus. The influence of drugs on nasal ciliary movement. Pharm. Res. 4:445–449 (1987).
W. A. J. J. Hermens, M. J. M. Deurloo, S. G. Romeijn, J. Verhoef, and F. W. H. M. Merkus. Nasal absorption enhancement of 17β-estradiol by dimethyl-β-cyclodextrin in rabbits and rats. Pharm. Res. 7:500–503 (1990).
N. G. M. Schipper, W. A. J. J. Hermens, S. G. Romeijn, J. Verhoef, and F. W. H. M. Merkus. Nasal absorption of 17β-estradiol and progesterone from a dimethyl-β-cyclodextrin inclusion formulation in rats. Int. J. Pharm. 64:61–66 (1990).
K. Uekama and M. Otagiri. Cyclodextrins in drug carrier systems. CRC Crit. Rev. Ther. Drug Carrier Syst. 3:1–40 (1987).
Y. Otani, T. Ieri, K. Uekama, K. Fukunaga, and J. Pitha. Differential effects of α-, β-and γ-cyclodextrins on human erythrocytes. Eur. J. Biochem. 186:17–22 (1989).
S.-I. Hirai, H. Okada, T. Yashiki, and Y. Uda. European Patent Application No. 83302118.1 (1983).
H. J. M. van de Donk, J. Zuidema, and F. W. H. M. Merkus. The influence of the pH and osmotic pressure upon tracheal ciliary beat frequency as determined with a new photo-electric registration device. Rhinology 18:93–104 (1980).
W. A. J. J. Hermens, P. M. Hooymans, J. Verhoef, and F. W. H. M. Merkus. Effects of absorption enhancers on human nasal tissue ciliary movement in vitro. Pharm Res. 7:144–146 (1990).
E. Björk and P. Edman. Degradable starch microspheres as a nasal delivery system for insulin. Int. J. Pharm. 47:233–238 (1988).
N. F. Farraj, B. R. Johansen, S. S. Davis, and L. Illum. Nasal administration of insulin using bioadhesive microspheres as a delivery system. J. Control. Rel. 13:253–261 (1990).
H. J. M. van de Donk, J. Zuidema, and F. W. H. M. Merkus. Correlation between the sensitivity of the ciliary beat frequency of human adenoid tissue and chicken embryo tracheas for some drugs. Rhinology 20:81–87 (1982).
G. S. M. J. E. Duchateau, J. Zuidema, and F. W. H. M. Merkus. Bile salts and nasal drug absorption. Int. J. Pharm. 31:193–199 (1986).
W. A. J. J. Hermens, C. W. J. Belder, J. M. W. M. Merkus, P. M. Hooymans, J. Verhoef, and F. W. H. M. Merkus. Intranasal estradiol administration to oophorectomized women. Eur. J. Obstet. Gynaecol. Reprod. Biol. (in press).
A. Yoshida, H. Arima, K. Uekama, and J. Pitha. Pharmaceutical evaluation of hydroxyalkyl ethers of β-cyclodextrins. Int. J. Pharm. 46:217–222 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Merkus, F.W.H.M., Verhoef, J.C., Romeijn, S.G. et al. Absorption Enhancing Effect of Cyclodextrins on Intranasally Administered Insulin in Rats. Pharm Res 8, 588–592 (1991). https://doi.org/10.1023/A:1015896405389
Issue Date:
DOI: https://doi.org/10.1023/A:1015896405389