Abstract
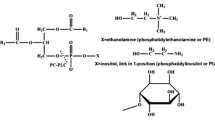
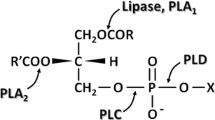
Octadecylphospho-l-serine (OPS), belonging to the family of alkylphosphate esters with anticancer activity, was synthesized by transesterification of octadecylphosphocholine using phospholipase d (PLD). With respect to the yield of product, PLD from cabbage proved to be superior to PLD from Streptomyces spp. Although the addition of n-hexane/2-octanol is advantageous to suppress the hydrolytic byproduct, a purely aqueous reaction medium was preferred because of better recovery of the product.
Similar content being viewed by others
References
Aurich I, Dürrschmidt P, Hirche F, Ulbrich-Hofmann R (1997) Transesterification of alkylphosphate esters by phospholipase D. Biotechnol. Lett. 19: 875-879.
Aurich I, Hirche F, Ulbrich-Hofmann R (1999) The determination of phospholipase D activity in emulsion systems. Anal. Biochem. 268: 337-342.
Brachwitz H, Langen P, Dube G, Schildt J, Paltauf F, Hermetter A (1990) Halo lipids. 10. Synthesis and cytostatic activity of Oalkylglycerophospho-L-serine analogs. Chem. Phys. Lipids 54: 89-98.
Brachwitz H, Ölke M., Bergmann J, Langen P (1997) Alkylphospho-L-serine analogues: synthesis of cytostatically active alkylphosphono derivatives Bioorg. Med. Chem. Lett. 7: 1739-1742.
Brachwitz H, Schönfeld R, Langen P, Paltauf F, Hermetter A (1989) New alkylphosphono-and phosphoserines, method for preparing them, and pharmaceutical substances containing them.World Patent WO 89/10370.
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248-254.
Hilgard P, Klenner T, Stekar J, Unger C (1993) Alkylphosphocholines: a new class of membrane-active anticancer agents. Cancer Chemother. Pharmacol. 23: 90-95.
Hirche F, Ulbrich-Hofmann R (1999) The interfacial pressure is an important parameter for the rate of phospholipase D catalyzed reactions in emulsion systems. Biochim. Biophys. Acta 1436: 383-389.
Hirche F, Ulbrich-Hofmann R (2000) The interdependence of solvent, acceptor alcohol and enzyme source in transphosphatidylation by phospholipase D. Biocatal. Biotrans. 18: 343-353.
Hirche F, Koch, MHJ, König S, Wadewitz T, Ulbrich-Hofmann R (1997) The influence of organic solvents on phospholipid transformations by phospholipase D in emulsion systems. Enzyme Microb. Technol. 20: 453-461.
Juneja LR, Kazuoka T, Yamane T, Shimizu S (1988) Kinetic evaluation of conversion of phosphatidylcholine to phosphatidylethanolamine by phospholipase D from different sources. Biochim. Biophys. Acta 960: 334-341.
Langen P, Maurer, HR, Brachwitz H, Eckert K, Veit A, Vollgraf C (1992) Cytostatic effects of various alkyl phospholipid analogues on different cells in vitro. Anticancer Res. 12: 2109-2112.
Maly K, Ñberall F, Schubert C, Kindler E, Stekar J, Brachwitz H, Grunicke HH (1995) Interference of new alkylphospholipid analogues with mitogenic signal transduction. Anti-Cancer Drug Des. 10: 411-425.
Servi S (1999) Phospholipases as synthetic catalysts. In: Fessner D, ed. Topics in Current Chemistry (Biocatalysis from Discovery to Application), Vol. 200. Berlin: Springer-Verlag, pp. 127-158.
Stekar J, Hilgard P, Voegeli R, Maurer HR, Engel J, Kutscher B, Nößner G, Schumacher W (1993) Antineoplastic activity and tolerability of a novel heterocyclic alkylphospholipid, D-20133. Cancer Chemother. Pharmacol. 32: 437-444.
Ulbrich-Hofmann R (2000) Phospholipases used in lipid transformations. In: Bornscheuer UT, ed. Enzymes in Lipid Modification. Weinheim: Wiley-VCH, pp. 219-262.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aurich, I., Dürrschmidt, P., Schierhorn, A. et al. Production of octadecylphospho-l-serine by phospholipase d. Biotechnology Letters 24, 585–590 (2002). https://doi.org/10.1023/A:1014876914801
Issue Date:
DOI: https://doi.org/10.1023/A:1014876914801