Abstract
Purpose: PulmoSphere™ particles are specifically engineered for delivery by the pulmonary route with a hollow and porous morphology, physical diameters < 5 μm, and low tap densities (circa 0.1 g.cm-3). Deposition of PulmoSphere particles in the human respiratory tract delivered by pressurized metered dose inhaler (pMDI) was compared with deposition of a conventional micronized drug pMDI formulation.
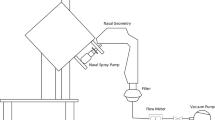
Methods: Nine healthy nonsmoking subjects (5 male, 4 female) completed a two-way crossover gamma scintigraphic study, assessing the lung and oropharyngeal depositions of albuterol sulfate, formulated as 99mTc-radiolabeled PulmoSphere particles or micronized particles (Ventolin EvohalerTM, GlaxoSmithKline, Ltd.) suspended in HFA-134a propellant.
Results: Mean (standard deviation) lung deposition, (% ex-valve dose) was doubled for the PulmoSphere formulation compared with Evohaler pMDI (28.5 (11.3) % vs. 14.5 (8.1) %, P < 0.01), whereas oropharyngeal deposition was reduced (42.6 (9.0) % vs. 72.0 (8.0) %, P < 0.01). Both PulmoSphere and Evohaler pMDIs gave uniform deposition patterns within the lungs.
Conclusions: These data provided “proof of concept” in vivo for the PulmoSphere technology as a method of improving targeting of drugs to the lower respiratory tract from pMDIs, and suggested that the PulmoSphere technology may also be suitable for the delivery of systemically acting molecules absorbed via the lung.
Similar content being viewed by others
REFERENCES
C. B. Lalor and A. J. Hickey. Pharmaceutical aerosols for delivery of drugs to the lungs. In I. Colbeck (ed.), Physical and Chemical Properties of Aerosols, Blackie Academic and Professional, London, United Kingdom, 1998, pp. 391–428.
A. L. Adjei and P. K. Gupta. Inhalation Delivery of Therapeutic Peptides and Proteins. Marcel Dekker, New York, 1997.
M. Hindle, D. A. G. Newton, and H. Chrystyn. Investigations of an optimal inhaler technique with the use of urinary salbutamol excretion as a measure of relative bioavailability to the lung. Thorax 48:607–610 (1993).
S. J. Farr, A. M. Rowe, R. Rubsamen, and G. Taylor. Aerosol deposition in the human lung following administration from a microprocesor controlled metered dose inhaler. Thorax 60:639–644 (1995).
S. P. Newman. Devices for inhaling medications. In T. J. H. Clark, T. Lee, S. Godfrey, and N. C. Thomson (eds.), Asthma, 4th ed., Edward Arnold Limited, London, United Kingdom, 2000, pp. 329–354.
R. Melchor, M. F. Biddiscombe, V. H. F. Mak, M. D. Short, and S. G. Spiro. Lung deposition patterns of directly labeled salbutamol in normal subjects and in patients with reversible airways obstruction. Thorax 48:506–511 (1993).
S. P. Newman, K. P. Steed, G. Hooper, J. I. Jones, and F. C. Upchurch. Improved targeting of beclomethasone dipropionate (250 ?g metered dose inhaler) to the lungs of asthmatics with the Spacehaler. Respir. Med. 93:424–431 (1999).
A. R. Clark. Medical aerosol inhalers: past, present and future. Aerosol Sci. Technol. 22: 374–391 (1995).
M. B. Dolovich. New propellant-free technologies under investigation. J. Aerosol Med. 12(suppl. 1):9–17 (1999).
D. A. Edwards, J. Hanes, G. Caponetti, J. Hrkach, A. Ben-Jebrai, M. L. Eskew, J. Mintzes, D. Deaver, N. Lotan, and R. Langer. Large porous particles for pulmonary drug delivery. Science 276: 1868–1871 (1997).
T. E. Tarara, J. Weers, and L. Dellamary. Engineered powders for inhalation. In R. N. Dalby, P. R. Byron, S. J. Farr, and J. Peart (eds.), Respiratory Drug Delivery VII, Serentec Press, Raleigh, North Carolina, 2000, pp. 413–416.
L. A. Dellamary, T. E. Tarara, D. J. Smith, C. H. Woelk, A. Adractas, M. L. Costello, H. Gill, and J. G. Weers. Hollow porous particles in metered dose inhalers. Pharm. Res. 17:168–174 (2000).
J. G. Weers, T. E. Tarara, H. Gill, B. S. English, and L. A. Dellamary. Homodispersion technology for HFA suspensions: particle engineering to reduce dosing variance. In R. N. Dalby, P. R. Byron, S. J. Farr, and J. Peart (eds), Respiratory Drug Delivery VII, Serentec Press, Raleigh, North Carolina, 2000, pp. 91–97.
United States Pharmacopeia. General tests and assays. Pharm. Forum 22:3048–3108 (1996).
S. P. Newman, A. R. Clark, N. Talaee, and S. W. Clarke. Pressurised aerosol deposition in the human lung with and without an 'open' spacer. Thorax 44:706–710 (1989).
Q. A. Summers, A. R. Clark, A. Hollingworth, J. Fleming, and S. T. Holgate. The preparation of a radiolabeled aerosol of nedoocromil sodium for administration by metered dose inhaler that accurately preserves particle size distribution of the drug. Drug Invest. 2:90–98 (1990).
P. H. Quanjer, G. J. Tammeling, J. E. Cotes, O. F. Pedersen, R. Peslin, and J. C. Yernault. Lung volumes and forced ventilatory flows. Eur. Respir. J. 6(suppl. 16):5–40 (1993).
G. R. Pitcairn and S. P. Newman. Tissue attenuation corrections in gamma scintigraphy. J. Aerosol Med. 3:187–198 (1997).
B. J. Meakin, D. A. Lewis, D. Ganderton, and G. Brambilla. In Vivo Lung Deposition of Hollow Porous Particles 263 Countering challenges posed by mimicry of CFC performance using HFA systems. In R. N. Dalby, P. R. Byron, S. J. Farr, and J. Peart (eds.), Respiratory Drug Delivery VII, Serentec Press, Raleigh, North Carolina, 2000, pp. 97–100.
C. L. Leach, P. J. Davidson, and R. J. Boudreau. Improved airway targeting with the CFC-free HFA-beclomethasone metereddose inhaler compared with CFC-beclomethasone. Eur. Respir. J. 12:1346–1353 (1998).
S. P. Newman. How well do in vitro particle size measurements predict drug delivery in vivo? J. Aerosol Med. 11(suppl. 1):97–104 (1998).
S. J. Farr, S. J. Warren, P. Lloyd, J. K. Okikawa, J. A. Schuster, A. M. Rowe, R. M. Rubsamen, and G. Taylor. Comparison of in vitro and in vivo efficiencies of a novel unit-dose aerosol generator and a pressurized metered dose inhaler. Int. J. Pharm. 198: 63–70 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirst, P.H., Pitcairn, G.R., Weers, J.G. et al. In Vivo Lung Deposition of Hollow Porous Particles from a Pressurized Metered Dose Inhaler. Pharm Res 19, 258–264 (2002). https://doi.org/10.1023/A:1014482615914
Issue Date:
DOI: https://doi.org/10.1023/A:1014482615914