Abstract
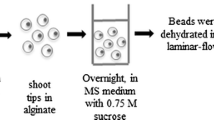
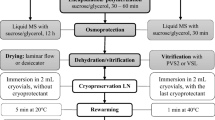
Embryonic axes of Persian lilac (Melia azedarach L.)encapsulated into calcium alginate beads with sucrose (0.75 M) and salicylicacid (0, 50 μM, 200 μM) were subjected to cryopreservationtechnique with dehydration and freezing in liquid nitrogen or to coldpreservation by stocking alginate beads in empty petri dishes for 4 monthsat 4 °C. In these two cases 200 μM salicylic acid enhancedsignificantly the percentage of viability of encapsulated embryonic axes andthe role of salicylic acid in increasing tolerance to dehydration is discussed.
Similar content being viewed by others
References
Blakesley, D., S. Al-Mazrooei & G.G. Henshaw, 1995. Cryopreservation of embryonic tissue of sweet potato (Ipomoea batatas): use of sucrose and dehydration for cryoprotection. Plant Cell Rep 15: 259–260.
Brodelius, P., L. Linse & K. Nilsson, 1982. Viability and biosynthetic capacity of immobilized plant cells. In: A. Fujiwara (Ed.), Plant Tissue Culture, p. 371. Proc 5th Int Cong of Plant Tissue and Cell Culture Mazuren, Tokyo.
Dereuddre, J., C. Scottez, Y. Arnaud & M. Duron, 1990. Resistance of alginate-coated axillary shoot tips of pear tree (Pyrus communis L., Beurre Hardy) in vitro plantlets to dehydration and subsequent freezing in liquid nitrogen: effect of previous cold hardening. C R Acad Sci Paris III310: 317–323.
Dumet, D., F. Engelmann, N. Chabrillange, Y. Duval & J. Dereuddre, 1993. Importance of sucrose for the acquisition of tolerance to dessication and cryopreservation of oil palm somatic embryos. Cryo-letters 14: 243–250.
Janeiro, L.V., M. Vieitez & A. Ballester, 1996. Cryopreservation of somatic embryos and embryonic axes of Camellia japonica L. Plant Cell Rep 15: 699–703.
Koster, K.L. & A.C. Leopold, 1988. Sugar and dessication tolerance in seeds. Plant physiol 88: 829–832.
Maruyama, E., I. Kinoshita, K. Ishii & K. Ohba, 1997. Germplasm conservation of the tropical forest trees, Cedrela odorata L., Guazuma crinita Mart. and Jacaranda mimosaefolia D. Don., by shoot tip encapsulation in calcium-alginate and storage at 12–25 ?C. Plant Cell Rep 16: 393–396.
Murashige, T. & F. Skoog, 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473–497.
Onay, A., C.E. Jeffree & M.M. Yeoman, 1996. Plant regeneration from encapsulated embryoids and an embryogenic mass of pistachio, Pistacia vera L. Plant Cell Rep 15: 723–726.
Radhamani, J. & K.P.S. Chandel, 1992. Cryopreservation of embryonic axes of trifoliate orange (Poncirus trifoliata (L.) RAF). Plant Cell Rep 11: 372–374.
Raskin, I., 1992. Role of salicylic acid in plants. Annual Rev Plant Physiol and Plant Mol Biol (USA) 43: 439–463.
Senaratna, T., B.D. McKersie & S.R. Bowley, 1989. Dessication tolerance of Alfafa (Medicago sativa L.) somatic embryos. Influence of abscisic acid, stress pretreatments and drying rates. Plant Sci 65: 253–259.
Seo, S., K. Ishizuka & Y. Ohashi, 1995. Induction of salicylic acid beta-glucosidase in tobacco (Nicotiana tabacum) leaves by exogenous salicylic acid. Plant and Cell Physiol 36(3): 447–453.
Shah, J. & D.F. Klessig, 1999. Salicylic acid: signal perception and transduction. In: P.P.J. Hooykaers, M.A. Hall & K.R. Libbenga (Eds.), Biochemistry and Molecular Biology of Plant Hormones, pp. 513–541. Elsevier Science, Amsterdam, Netherlands.
Uragami, A., A. Sakai & A. Nagai, 1990. Cryopreservation of dried axillary buds from plantlet of Asparagus officinalis L. grown in vitro. Plant Cell Rep 9: 328–331.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bernard, F., Shaker-Bazarnov, H. & Kaviani, B. Effects of salicylic acid on cold preservation and cryopreservation of encapsulated embryonic axes of Persian lilac (Melia azedarach L.). Euphytica 123, 85–88 (2002). https://doi.org/10.1023/A:1014416817303
Issue Date:
DOI: https://doi.org/10.1023/A:1014416817303