Abstract
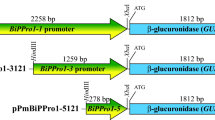
Sporamin, a tuberous storage protein of sweet potato, was systemically expressed in leaves and stems by wound stimulation. In an effort to demonstrate the regulatory mechanism of wound response on the sporamin gene, a 1.25 kb sporamin promoter was isolated for studying the wound-induced signal transduction. Two wound response-like elements, a G box-like element and a GCC core-like sequence were found in this promoter. A construct containing the sporamin promoter fused to a β-glucuronidase (GUS) gene was transferred into tobacco plants by Agrobacterium-mediated transformation. The wound-induced high level of GUS activity was observed in stems and leaves of transgenic tobacco, but not in roots. This expression pattern was similar to that of the sporamin gene in sweet potatoes. Exogenous application of methyl jasmonate (MeJA) activated the sporamin promoter in leaves and stems of sweet potato and transgenic tobacco plants. A competitive inhibitor of ethylene (2,5-norbornadiene; NBD) down-regulated the effect of MeJA on sporamin gene expression. In contrast, salicylic acid (SA), an inhibitor of the octadecanoid pathway, strongly suppressed the sporamin promoter function that was stimulated by wound and MeJA treatments. In conclusion, wound-response expression of the sporamin gene in aerial parts of plants is regulated by the octadecanoid signal pathway.
Similar content being viewed by others
References
An, G., Costa, M.A. and Ha, S.B. 1990. Nopaline synthase promoter is wound inducible and auxin inducible. Plant Cell 2: 225–233.
Bell, E. and Mullet, J.E. 1993. Characterization of the Arabidopsis lipoxygenase gene responsive to methyl-jasmonate and wounding. Plant Physiol. 103: 1133–1137.
Birkenmeier, G.F. and Ryan, C.A. 1998. Wound signaling in tomato plants. Plant Physiol. 117: 687–693.
Bradford, M.M. 1976. A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Chen, J.C., Chen, Y.M. and Yeh, K.W. 1997. Isolation and characterization of new sporamin gene members from sweet potato (Ipomoea batatas). Taiwania 42 (1): 34–42.
Conconi, A., Miquel, M. and Ryan, C.A. 1996. Changes in the intracellular lipid composition and free fatty acids of tomato leaves in response to wounding. Plant Physiol. 111: 717–803.
Creelman, R.A. and Mullet, J.E. 1995. Jasmonic acid distribution and action in plants: regulation during development and response to biotic and abiotic stress. Proc. Natl. Acad. Sci. USA 92: 4114–4119.
Creelman, R.A., Tierney, M.L. and Mullet, J.E. 1992. Jasmonic acid/methyl jasmonate accumulate in wounded soybean hypocotyls and modulate wound gene expression. Proc. Natl. Acad. Sci. USA 89: 4938–4941.
Devereux, J., Haeberli, P. and Smithies, O. 1984. A comprehensive set of sequence analysis programs for the VAX. Nucl. Acids Res. 12: 195–387.
Ding, L.C., Hu, C.Y., Yeh, K.W. and Wang, P.J. 1998. Development of insect-resistant transgenic cauliflower plants expressing the trypsin inhibitor gene isolated from local sweet potato. Plant Cell Rep. 17: 854–860.
Doares, S.H., Narvaez-Vasquez, J., Conconi, A. and Ryan, C.A. 1995. Salicylic acid inhibits synthesis of proteinase inhibitors in tomato leaves induced by systemin and jasmonic acid. Plant Physiol. 108: 1741–1746.
Farmer, E.E. and Ryan, C.A. 1992. Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell 4: 129–134.
Hattori, T., Nakagawa, S. and Nakamura, K. 1990. High-level expression of tuberous root storage protein genes of sweet potato in stems of plantlets grown in vitro on sucrose medium. Plant Mol. Biol. 14: 595–604.
Hattori, T., Yoshida, N. and Nakamura, K. 1989. Structural relationship among the members of multigene family coding for the sweet potato tuberous roots storage proteins. Plant Mol. Biol. 13: 563–572.
Hörsch, R.B., Fry, J.E., Hoffmann, N.L., Eichholtz, D., Rogers, S.G. and Fraley, R.T. 1985. A simple and general method for transferring genes into plants. Science 227: 1229–1231.
Imanishi, S., Kito-Nakamura, K., Matsuoka, K., Morikami, A. and Nakamura, K. 1997. A major jasmonate-inducible protein of sweet potato, ipomoelin, is an ABA-independent woundinducible protein. Plant Cell Physiol. 38: 643–652.
Jefferson, R.A., Kavanagh, T.A. and Bevan, M.W. 1987. GUS fusion: ?-glucuronidase as a sensitive and versatile gene fusion marker. EMBO J. 6: 3901–3908.
Maeshima, M., Sasaki, T. and Asahi, T. 1985. Characterization of major proteins in sweet potato tuberous roots. Phytochemistry 24: 1899–1902.
Mason, H.S., DeWald, D.B. and Mullet, J.E. 1993. Identification of a methyl jasmonate-responsive domain in the soybean vspB promoter. Plant Cell 5: 241–251.
Menke, F.L.H., Champion, A., Kijne, J.W. and Memelink J. 1999. A novel jasmonate-and elicitor-responsive element in the periwinkle secondary metabolite biosynthetic gene Str interacts with a jasmonate-and elicitor-inducible AP2-domain transcription factor, ORCA2. EMBO J. 18: 4455–4463.
Murashige, T. and Skoog F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497
O'Donnel, P.J., Calvert, C., Atzorn, R., Wasternack, C., Leyser, H.M.O. and Bowles, D.J. 1996. Ethylene as a signal mediating the wound response of tomato plants. Science 274: 1914–1917.
Ohta, S., Hattori, T., Morikami, A. and Nakamura, K. 1991. Highlevel expression of a sweet potato sporamin gene promoter: ?-glucuronidase (GUS) fusion gene in the stems of transgenic tobacco plants is conferred bymultiple cell type-specific regulatory elements. Mol. Gen. Genet. 225: 369–378.
Ohto, M., Nakamura-Kito, K. and Nakamura, K. 1992. Induction of expression of genes coding for sporamin and ?-amylase by polygalacturonic acid in leaf-petiole cuttings of sweet potato. Plant Physiol. 99: 422–427.
Palm, C.J., Costa, M.A., An, G. and Ryan, C.A. 1990. Woundinducible nuclear protein binds DNA fragments that regulate a proteinase inhibitor II gene from potato. Proc. Natl. Acad. Sci. USA 87: 603–607.
Peña-Cortés, H., Fisahn, J. and Willmitzer, L. 1995. Signals involved in wound-induced proteinase inhibitor II gene expression in tomato and potato plants. Proc. Natl. Acad. Sci. USA 92: 4106–4113.
Siebert, P.D., Chenchik, A., Kellogg, D.E., Lukyanov, K.A. and Lukyanov, S.A. 1995. An improved PCR method for walking in uncloned genomic DNA. Nucl. Acids Res. 23: 1087–1088.
Takeda, S., Kowyama, Y., Takeuchi, Y., Matsuoka, K., Nishimura, M. and Nakamura, K. 1995. Spatial patterns of sucrose-inducible and polygalacturonic acid-inducible expression of genes that encode sporamin and ß-amylase in sweet potato. Plant Cell Physiol. 36: 321–333.
Wang, S.J., Lin, C.T., Ho, K.C., Chen, Y.M. and Yeh, K.W. 1995. Nucleotide sequence of a sporamin gene in sweet potato. Plant Physiol. 108: 829–830.
Weiss, C. and Bevan, M. 1991. Ethylene and wound signal modulate local and systemic transcription of win2 genes in transgenic potato plants. Plant Physiol. 96: 943–951.
Williams, M.E., Foster, R. and Chua, N.H. 1992. Sequences flanking the hexameric G-box core CACGTG affect the specificity of protein binding. Plant Cell 4: 485–496.
Xu, Y., Chang, P.F.L., Liu, D., Narasimhan, M.L., Raghothama, K.G., Hasegawa, P.M. and Bressan, R.A. 1994. Plant defense genes are synergistically induced by ethylene and methyl jasmonate. Plant Cell 4: 1077–1085.
Yeh, K.W., Chen, J.C., Lin, M.I., Chen Y.M. and Lin, C.Y. 1997a. Functional activity of sporamin from sweet potato (Ipomoea batatas): a tuberous storage protein with trypsin inhibitor activity. Plant Mol. Biol. 33: 565–570.
Yeh, K.W., Juan, R.H. and Su, J.C. 1991. A rapid and efficient method for RNA isolation from plants with high carbohydrate content. Focus 13: 102–103.
Yeh, K.W., Lin, M.I., Tunan, S.J., Chen, Y.M., Lin, C.Y. and Kao, S.S. 1997b. Sweet potato (Ipomoea batatas) trypsin inhibitors expressed in transgenic tobacco plants confer resistance against Spodoptera litura. Plant Cell Rep. 16: 692–696.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, SJ., Lan, YC., Chen, SF. et al. Wound-response regulation of the sweet potato sporamin gene promoter region. Plant Mol Biol 48, 223–231 (2002). https://doi.org/10.1023/A:1013359227041
Issue Date:
DOI: https://doi.org/10.1023/A:1013359227041